In the intricate study of organic chemistry, most transformations do not occur in a single, concerted step.
Instead, they proceed through the formation of fleeting, high-energy species known as reactive intermediates.
While much academic focus is often placed on the positive carbocation, a complete understanding of reaction pathways requires an in-depth look at Carbanions and free radicals.
These reactive species dictate the outcome of base-catalysed reactions and photochemical processes, serving as essential building blocks for complex molecular synthesis.
Carbanions and Free Radicals
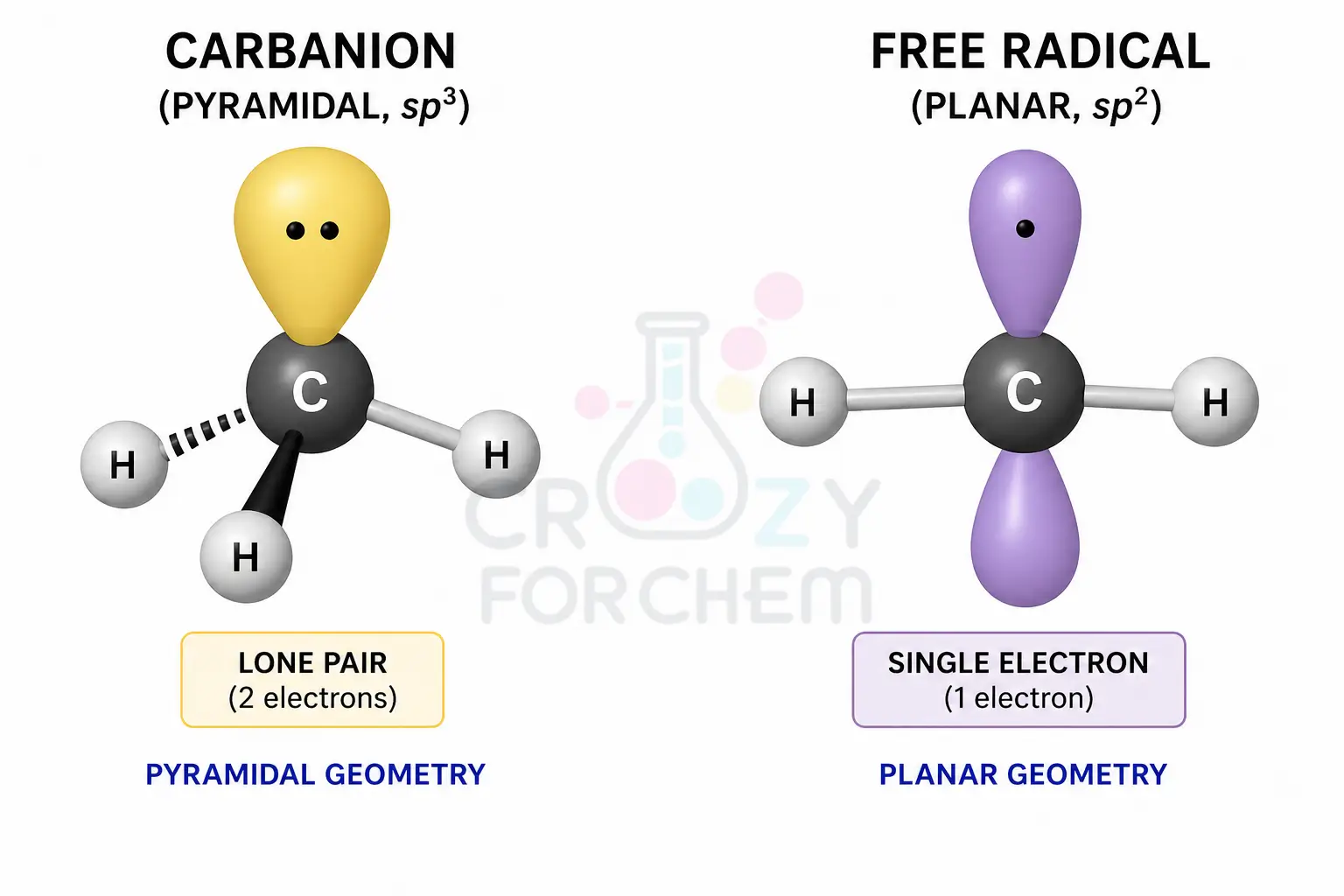
Carbanions are electron-rich, negatively charged species (sp³) formed via heterolytic fission, whereas free radicals are neutral species (sp²) with an unpaired electron generated through homolytic fission. Their stability is determined by their electronic environment and geometry.
Intermediate Stability Order Primary Stabilisation Carbanions Methyl > 1° > 2° > 3° s-character, -I groups Free Radicals 3° > 2° > 1° > Methyl Hyperconjugation, +I groups Key Distinction: The carbanion stability order is the exact inverse of the free radical and carbocation trends because electron-donating alkyl groups destabilise an existing negative charge via the inductive effect (+I).
Table of Contents
Introduction to Reactive Intermediates
Beyond Carbocations
Organic reactions often involve the temporary existence of species that are neither the starting material nor the final product.
These reaction intermediates are highly unstable and typically exist for only a few seconds or less before transforming into more stable molecules.
They represent a “local minimum” in a reaction’s potential energy diagram, distinguishing them from transition states, which are maximum energy peaks.
To predict whether a reaction will proceed or which product will dominate, chemists must understand the electronic environment, hybridisation, and geometry of these intermediates.
The Role of Bond Fission
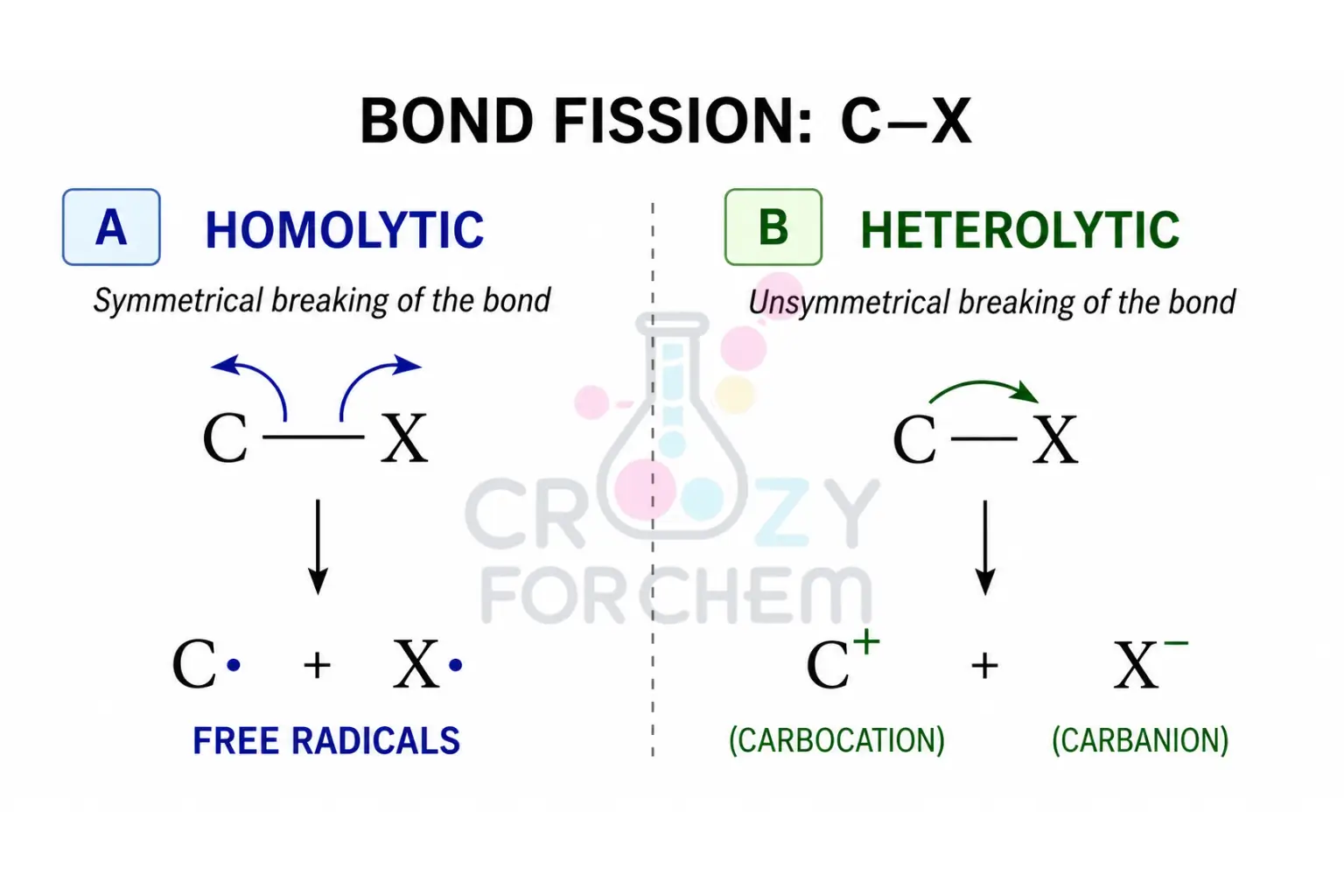
The formation of Carbanions and free radicals is determined by how a covalent bond breaks, a process known as bond fission or cleavage.
- Heterolytic Fission (Heterolytic Cleavage): This occurs when a covalent bond breaks unsymmetrically, and one of the atoms takes both electrons from the shared pair. This process creates ions: a positively charged carbocation (electron-deficient) and a negatively charged carbanion (electron-rich).
- Homolytic Fission: This happens when a bond breaks symmetrically, and each atom retains one electron from the shared pair. This results in the formation of neutral species with unpaired electrons, known as free radicals.
Understanding Carbanions (The Electron-Rich Species)
Definition and Generation
A carbanion is a chemical species where a carbon atom bears a formal negative charge and possesses eight valence electrons in its outer shell.
Because they have a complete octet and an unshared pair of electrons, they are electron-rich and act as powerful nucleophiles or bases in chemical reactions.
They are primarily generated through two pathways:
- Deprotonation: A strong base removes a proton (H⁺) from a carbon atom, leaving the bonding pair of electrons behind on the carbon. This is common in acetylide ion reactions, where terminal alkynes are treated with strong bases like sodamide.
- Nucleophilic Addition: A nucleophile adds to an unsaturated carbon (like a double bond), breaking a π bond and creating a new carbanionic centre.
Structure and sp³ Hybridisation
The carbanion geometry is typically characterised by sp3 hybridisation at the central carbon.
The three bonding pairs and the one lone pair of electrons arrange themselves to minimise repulsion, resulting in a pyramidal geometry similar to that of ammonia (NH₃).
This structure is not static; carbanions can rapidly interconvert between two pyramidal forms.
However, if the negative charge is adjacent to a multiple bond or a π-system, the carbanion may adopt a planar structure to facilitate resonance delocalisation.
Stability Trends of Carbanions
The Inverse Alkyl Trend
The stability order of carbanions and free radicals, with examples, reveals a striking difference between these species. For carbanions, the stability decreases as alkyl substitution increases.
Stability Order: Methyl > Primary (1°) > Secondary (2°) > Tertiary (3°)
As an educator, I often compare a negative charge to a heavy backpack.
If you are already struggling with the weight (the extra electron pair), and your neighbours (alkyl groups) keep pushing more “weight” (electron density) into your bag via the inductive effect (+I), the burden becomes unbearable, and you become less stable.
Consequently, the tertiary carbanion, surrounded by three electron-donating alkyl groups, is the most unstable alkyl carbanion.
The Role of s-Character
Hybridisation significantly influences carbanion stability. The more s-character a hybrid orbital has, the closer the electrons are held to the nucleus, which better neutralises and stabilises a negative charge.
- sp-hybridised (50% s): Most stable (e.g., acetylide ions).
- sp2-hybridised (33% s): Intermediate stability.
- sp3-hybridised (25% s): Least stable.
The Influence of Solvents on Carbanion Stability
Beyond internal structural factors, the environment plays a critical role in stabilising these species.
Carbanions are significantly more reactive and stable in polar aprotic solvents such as Dimethyl Sulfoxide (DMSO) or Hexamethylphosphoramide (HMPA).
Unlike protic solvents, these do not hydrogen-bond with the anion. Instead, they effectively solvate and ‘trap’ the counter-cation, leaving the carbanion ‘naked’ and highly accessible for synthetic transformations.
Resonance and Aromaticity
Resonance delocalises the negative charge over multiple atoms, greatly enhancing stability. For example, allylic and benzylic carbanions are more stable than simple alkyl ones because the charge is “smeared” over a π-system.
A fascinating “expert nuance” is the Cyclopentadienyl Anion. Despite being a carbanion, it is exceptionally stable because its 6π electrons satisfy Hückel’s Rule (4n+2), making the species aromatic.
In contrast, the acyclic version of the same formula is significantly less stable, proving the power of aromatic delocalisation.
Understanding Free Radicals (The Neutral Species)
Definition and Characteristics
Free radicals are species that contain one or more unpaired electrons.
While they are electrically neutral, they have only seven valence electrons, making them electron-deficient and highly reactive as they seek to complete their octet.
Because of their unpaired electron, they possess a net magnetic moment and are paramagnetic, allowing them to be detected using electron spin resonance (ESR).
Generation via Homolytic Fission
Free radicals are the typical by-product of homolytic fission. This process is usually triggered by:
- Thermal Cleavage: High temperatures provide the energy needed to break a bond symmetrically.
- Photochemical Fission: Exposure to ultraviolet (UV) light triggers bond breaking, as seen in the halogenation of alkanes.
Structure and sp² Hybridisation
Simple alkyl radicals generally possess a trigonal planar structure. The central carbon is sp2 hybridised, with the three sigma bonds at 120° angles and the odd unpaired electron residing in a p-orbital perpendicular to the plane.
The planar geometry of the radical intermediate is particularly significant in stereochemistry.
Since the reagent can attack the vacant p-orbital with equal probability from either the top or bottom face, reactions occurring at a chiral centre inevitably lead to racemisation.
This loss of optical activity is a hallmark of radical-mediated mechanisms.
Stability Trends of Free Radicals
Alkyl Substitution and Hyperconjugation
The radical stability order follows the same trend as carbocations, which is the exact opposite of carbanions. Stability Order: Tertiary (3°) > Secondary (2°) > Primary (1°) > Methyl.
The inductive effect and hyperconjugation in radicals primarily provide this stability.
In a tertiary radical, nine α-hydrogens are available to share electron density with the half-filled p-orbital through sigma-bond delocalisation, providing significant stabilisation.
As the number of methyl groups increases, the intensity of the “lack of electron” at the central carbon is reduced through redistribution over the molecule.
Resonance in Radicals
Like other intermediates, free radicals are heavily stabilised by resonance. Allylic and benzylic radicals are far more stable than simple alkyl radicals because the unpaired electron is delocalised over the adjacent π-cloud.
A classic reaction involving these species is the chlorination of methane, which proceeds via a methyl radical intermediate.

Advanced Intermediates: Carbenes and Nitrenes
For a truly comprehensive understanding, one must look at advanced reactive species like carbenes and nitrenes, which further demonstrate the diversity of electron-deficient carbon and nitrogen.
Carbenes
Carbenes are neutral species carrying two non-bonding electrons on a carbon atom, giving it a total of only six valence electrons. They exist in two states:
- Singlet Carbene: The two electrons are paired in a single sp² hybrid orbital with opposite spins.
- Triplet Carbene: The two electrons are unpaired and reside in different orbitals, making this state paramagnetic and generally more stable for simple hydrocarbons.
Nitrenes
Nitrenes are the nitrogen analogues of carbenes. They are highly reactive and neutral, possessing only six electrons in the valence shell of nitrogen.
Like carbenes, they can exist as singlet or triplet species and are generated through the thermal or photochemical decomposition of azides.
Comparative Analysis: Carbanions vs Free Radicals
Understanding the core differences between Carbanions and free radicals is vital for predicting reaction pathways.
| Feature | Carbanion | Free Radical |
|---|---|---|
| Bond Fission | Heterolytic cleavage | Homolytic fission |
| Electronic Nature | Charged (Negative) | Neutral |
| Valence Electrons | Eight (Octet complete) | Seven (Incomplete octet) |
| Geometry | Pyramidal geometry (sp³) | Planar (sp²) |
| Stability Order | Methyl > 1° > 2° > 3° | 3° > 2° > 1° > Methyl |
| Primary Stabilisation | s-character, -I groups | Hyperconjugation, +I groups |
| Character | Nucleophilic / Basic | Paramagnetic / Radical |
Conclusion: Predicting Reaction Pathways
Mastering the formation and properties of Carbanions and free radicals allows chemists to look under the “bonnet” of a chemical reaction.
By identifying the type of bond fission involved, whether it be homolytic fission producing a neutral radical or heterolytic fission producing an electron-rich carbanion, one can accurately predict the stability of the transition state and the final product.
Whether you are analysing the radical stability order in a chain reaction like the halogenation of alkanes or the carbanion geometry in a base-catalysed synthesis, remember the fundamental electronic “safety nets”.
Delocalisation, hybridisation, and substitution are the invisible forces that determine the life and death of these fascinating reactive intermediates.
Frequently Asked Questions
-
Q1: What is the difference between homolytic and heterolytic fission?
Homolytic fission occurs when a covalent bond breaks symmetrically, and each atom retains one electron, resulting in two neutral free radicals. In contrast, heterolytic fission is the unsymmetrical breaking of a bond where one atom takes both bonding electrons, forming a positive carbocation and a negative carbanion.
-
Q2: Why is the carbanion stability order the reverse of carbocations?
Carbanion stability follows the order: Methyl > Primary (1°) > Secondary (2°) > Tertiary (3°). This is because alkyl groups are electron-donating via the inductive effect (+I); adding electron density to an already negatively charged carbon increases repulsion and destabilises the species.
-
Q3: What is the geometry and hybridisation of a free radical?
A simple alkyl free radical typically adopts a trigonal planar geometry with sp² hybridisation. The central carbon forms three sigma bonds at 120° angles, while the single unpaired electron resides in a p-orbital perpendicular to the molecular plane.
-
Q4: How does s-character affect the stability of carbanions?
Higher s-character leads to greater carbanion stability because s-orbitals are closer to the nucleus, allowing the positive nuclear charge to better neutralise the negative charge. Consequently, a sp-hybridised acetylide ion (50% s-character) is significantly more stable than sp² or sp³ hybridised carbanions.
-
Q5: Why do free radical reactions often result in racemisation?
Because free radicals have a planar sp² structure, a reacting reagent can attack the unpaired electron’s p-orbital from either the top or bottom face with equal probability. This lack of facial selectivity at a chiral centre leads to the formation of a 50:50 mixture of enantiomers, known as racemisation.
-
Q6: Are free radicals charged or neutral species?
Free radicals are electrically neutral species because they possess the same number of protons and electrons. However, they are highly reactive because they have an incomplete octet (seven valence electrons) and are paramagnetic due to the presence of an unpaired electron.

