Carbenes and nitrenes are among the most fascinating reactive intermediates in organic chemistry.
While carbocations and carbanions are the “bread and butter” of undergraduate studies, mastering Carbenes and nitrenes is the hallmark of a high-authority chemist.
These neutral, uncharged species defy the standard rules of valency, operating with a sextet of electrons that makes them exceptionally reactive.
As an educator, I tell my students: think of a triplet carbene as a ‘diradical’ that takes its time to flip its spin, while a singlet carbene is a ‘concerted’ specialist that acts instantly.
Understanding these intermediates is like observing the silent architects that build complex three-dimensional structures from simple precursors.
Carbenes and nitrenes are neutral, electron-deficient reactive intermediates. A carbene contains a divalent carbon with 6 valence electrons (formula: R₂C:), while a nitrene contains a monovalent nitrogen with 6 valence electrons (formula: RN:). Both exist in singlet and triplet spin states.
Table of Contents
What are Carbenes and Nitrenes? (The Sextet Species)
Defining Carbenes: Divalent Carbon Species
A carbene is a neutral, highly reactive species containing a divalent carbon atom.
Unlike standard carbon, which forms four bonds, the carbon in a carbene is linked to only two substituents and possesses two non-bonding electrons.
This leaves the carbon with only six valence electrons, creating a permanent electron deficiency that drives its high reactivity.
These divalent carbon species are typically represented by the general formula R₂C:.
Defining Nitrenes: The Nitrogen Analogues
Nitrenes are the nitrogen counterparts of carbenes.
A nitrene features a monovalent nitrogen atom surrounded by a sextet of electrons. Similar to carbenes, nitrogen possesses only one bond to a substituent and four non-bonding electrons.
Of these four electrons, two are considered the “typical” lone pair associated with nitrogen, while the other two determine the spin state.
Because they are isoelectronic with carbenes, their chemical behaviour and electronic structures are remarkably similar.
The Electron Deficiency Factor
Both intermediates are classified as electron-deficient nitrogen or carbon species. Because they lack a complete octet, they function as powerful electrophiles.
They seek electron density from multiple bonds or even single C-H bonds to achieve stability, making them indispensable in modern synthetic methods.
Understanding Spin States: Singlet vs Triplet
A fundamental concept in the study of these reaction intermediates is spin multiplicity, which dictates their geometry and reactivity.
The difference between singlet and triplet carbenes and nitrenes primarily lies in how their non-bonding electrons are distributed within their molecular orbitals.
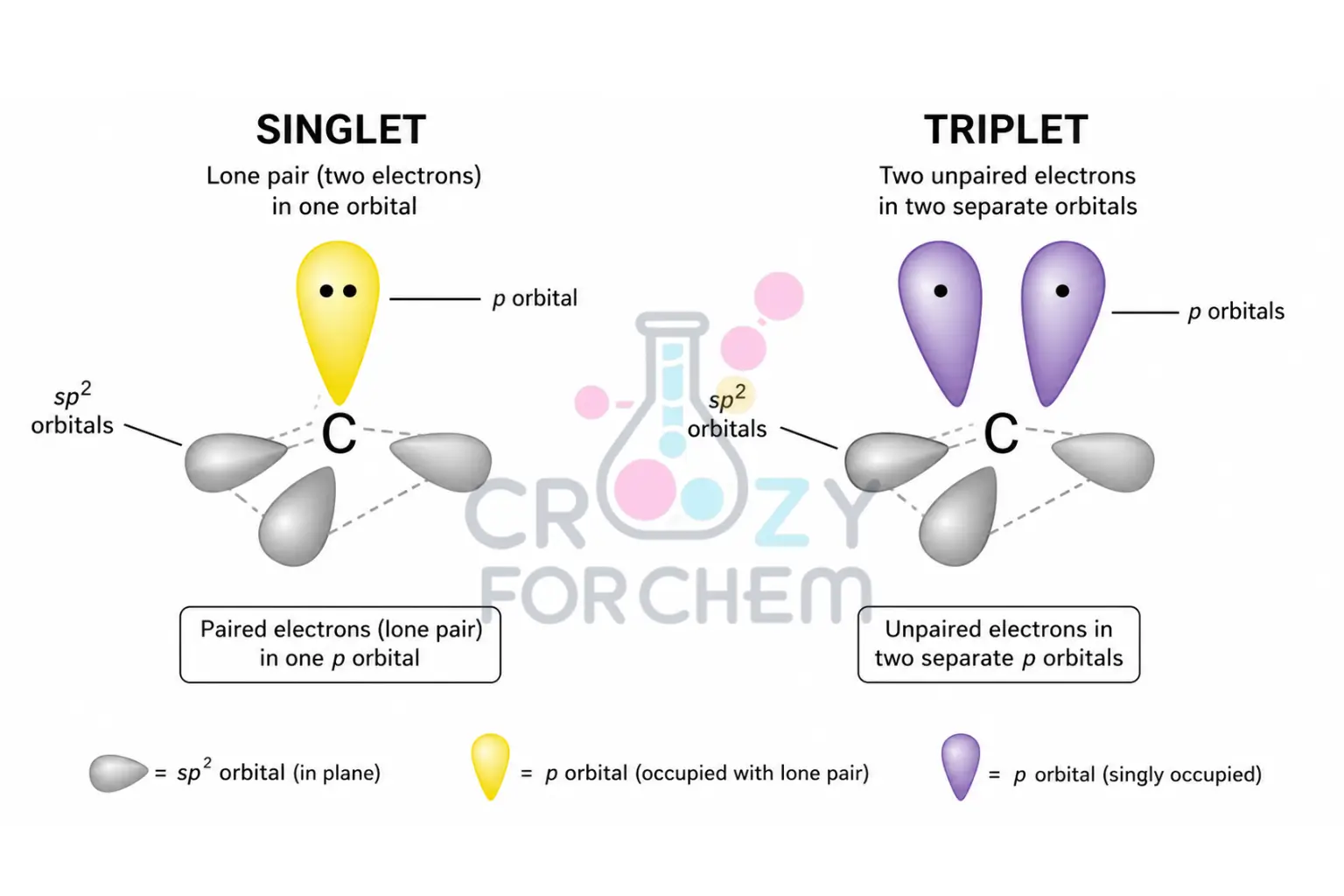
Singlet State
In the singlet state, the two non-bonding electrons have opposite spins and occupy the same sp² hybrid orbital.
This arrangement leaves a vacant p-orbital perpendicular to the plane of the molecule, giving the species a carbocation-like character.
Singlet species typically exhibit a bent trigonal planar geometry with bond angles ranging between 100° and 110°. Because the electrons are paired, singlet states are diamagnetic.
Triplet State
In the triplet state, the two electrons have parallel spins and occupy two different orbitals, one sp² orbital and one p-orbital.
This separation reduces the coulombic repulsion between the electrons. Triplet species are diradical-like in nature and generally possess a more open bond angle, typically ranging from 130° to 150°.
Because of their unpaired electrons, they are paramagnetic and can be distinguished using electron paramagnetic resonance (EPR) spectroscopy.
Spin State Stability
The spin state stability of these species is governed by Hund’s Rule, which states that for most simple Carbenes and nitrenes such as methylene (:CH₂) or the NH radical the triplet state is the lower-energy ground state.
For simple alkyl carbenes, the triplet state is typically about 8 kcal/mol more stable than the singlet.
However, this trend can be reversed if the central atom is directly bonded to heteroatoms like Oxygen, Nitrogen, or halogens.
These atoms can provide stabilisation through the mesomeric effect, donating electron density from their lone pairs into the empty p-orbital of a singlet carbene.
This explains why species like dichlorocarbene (:CCl₂) or N-heterocyclic carbenes (NHCs) possess a singlet ground state.
Methods of Generation: Bringing Intermediates to Life
Because of their extreme reactivity and short lifetimes, these intermediates are produced in situ using various thermal or photochemical methods.
Nitrene Formation
The most common pathway for nitrene formation is the decomposition of organic azides (R-N₃).
- Thermolysis/Photolysis of Azides: Heating or exposing azides to UV light causes the release of nitrogen gas (N₂), leaving behind a reactive nitrene. Following the spin conservation rule, singlet nitrenes are initially produced during thermolysis.
- Isocyanates: Nitrenes can also be generated with the emission of carbon monoxide from isocyanates.
- Sulfonamides and Iminoiodanes: These are also used as precursors; for example, iodo-compounds like iminoiodanes can release nitrenes upon photolysis.
Carbene Generation
- Diazoalkanes: Photolysis of diazo compounds (R₂CN₂) releases N₂ to form a carbene. Photolysis generally yields the triplet state through relaxation, while gentle warming can favour the singlet state.
- Alpha-Elimination: This is the standard method for generating halocarbenes. Treating chloroform (CHCl₃) with a strong base results in the alpha-elimination of a proton and a chloride ion via an ionic mechanism, yielding singlet dichlorocarbene (:CCl₂).
- Ketenes and Epoxides: Thermal or photolytic decomposition of ketenes or epoxides also serves as a viable route for carbene synthesis.
Reactivity and Mechanisms: The Stereochemistry Rule
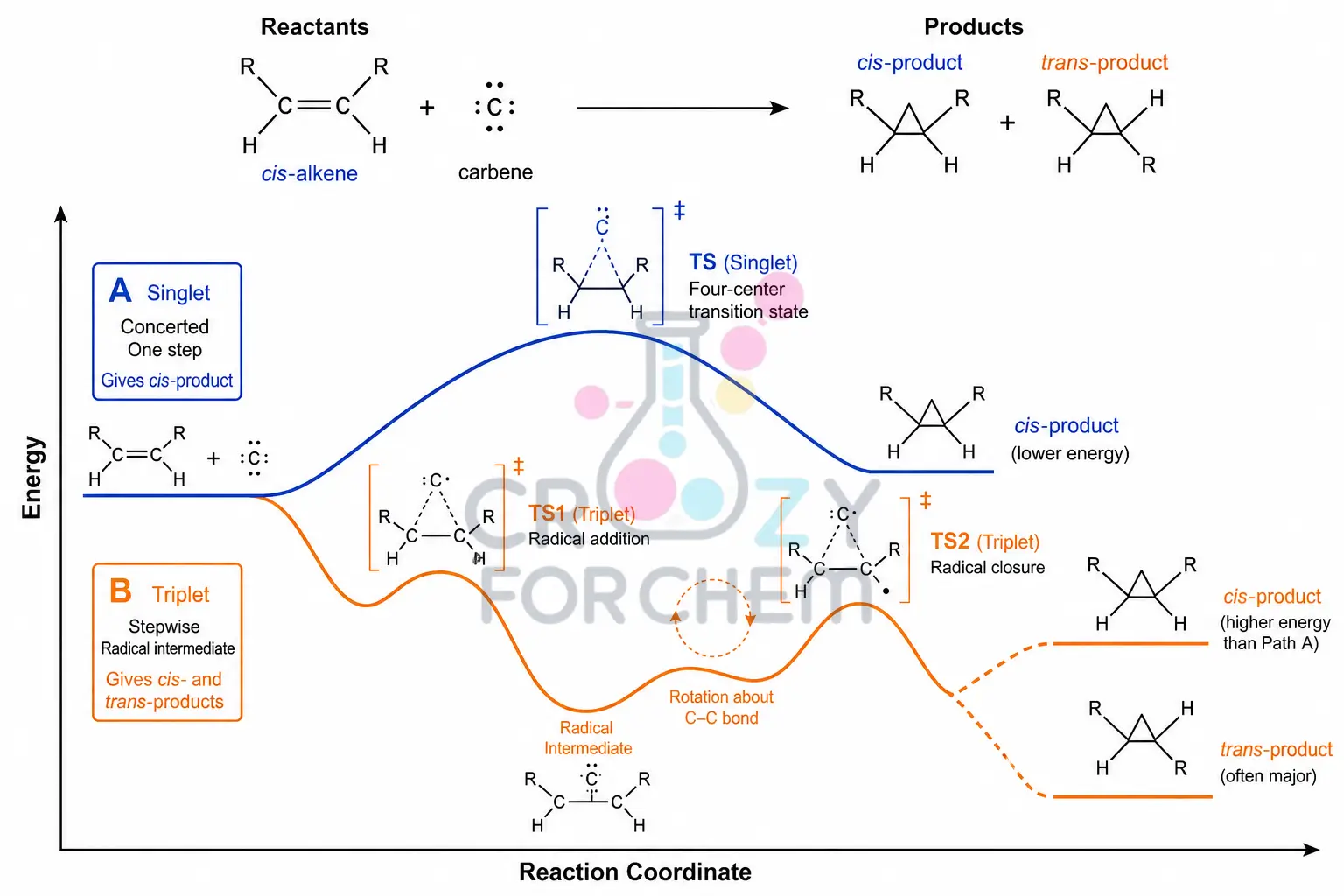
The spin multiplicity of the intermediate directly dictates the stereochemical outcome of its addition reactions.
Cyclopropanation and Aziridination
When carbenes add to alkenes, they form three-membered carbon rings (cyclopropanes), while nitrenes form three-membered nitrogen rings (aziridines).
- Singlet Reactivity: Singlet species undergo a stereospecific addition. Because their electrons are spin-paired, they form two bonds simultaneously in a single, concerted step. This means a cis-alkene will yield a purely cis-product, and a trans-alkene will yield a trans-product, with total retention of the original configuration.
- Triplet Reactivity: Triplet species react via a stepwise radical mechanism. Because their electrons have parallel spins, they cannot form two bonds at once. After the first bond forms, the intermediate radical must wait for a “spin flip” or spin inversion before the second bond can close. During this delay, free rotation around the single bond occurs, resulting in a non-stereospecific mixture of cis and trans isomers.
Expert Note: The spin multiplicity is calculated using the formula 2S + 1, where S is the total spin. For a singlet, S = 0 (paired), so the multiplicity is 1. For a triplet, S = 1 (parallel), so multiplicity is 3.
Insertion Reactions
Both species exhibit insertion reactions into C-H bonds.
- Singlet carbenes insert randomly, while singlet nitrenes do so selectively.
- The selectivity for singlet nitrene insertion follows the order: tertiary C-H > secondary C-H > primary C-H.
- Triplet carbenes insert selectively but not stereospecifically, whereas triplet nitrenes typically do not participate in C-H insertion.
Advanced Topics: Fischer, Schrock, and NHCs
Modern organometallic chemistry relies heavily on carbenoids, metal-stabilised carbenes that react similarly to carbenes but are not true divalent species.
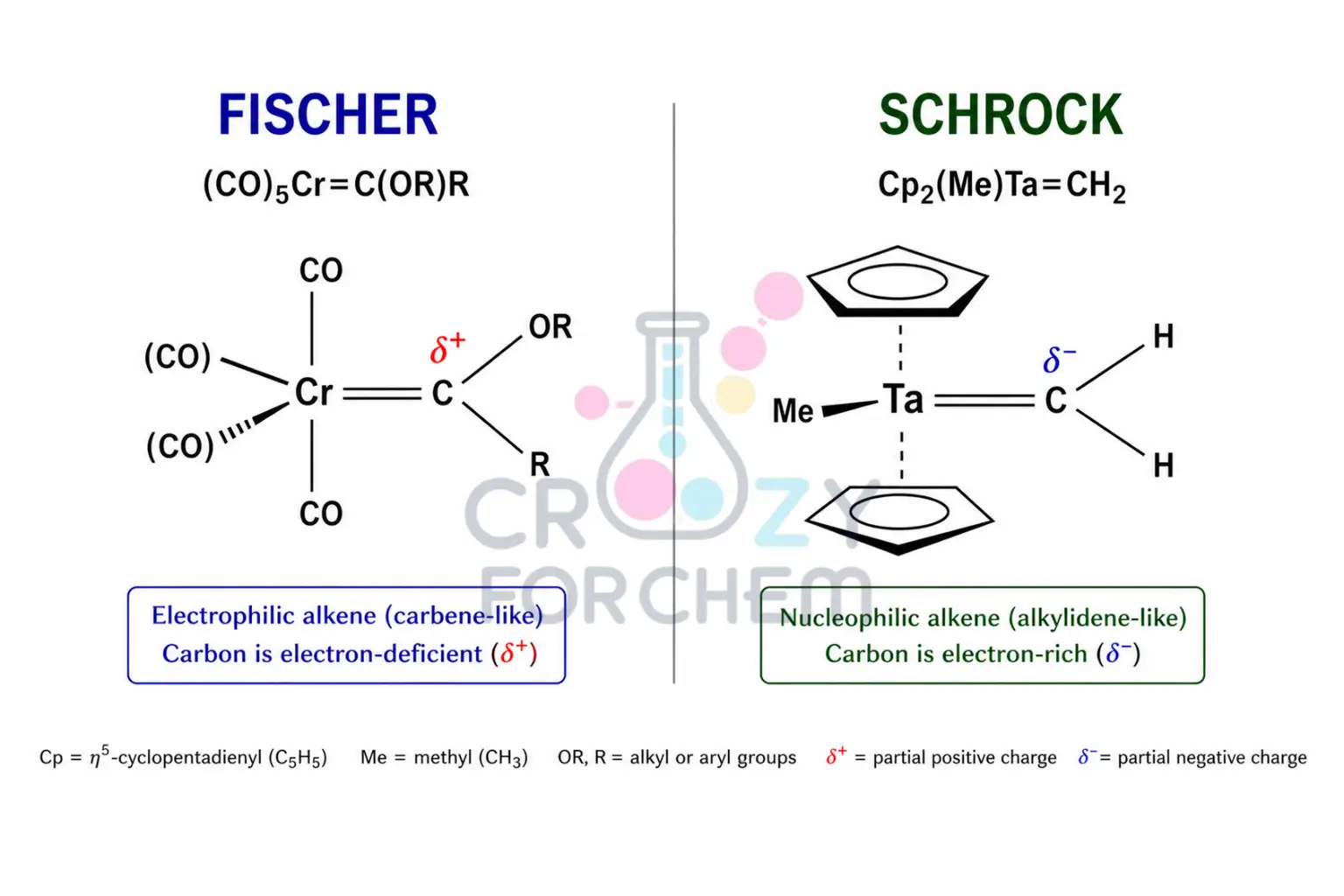
Fischer Carbenes
Named after Ernst Otto Fischer, these species feature low-oxidation-state metals (such as Fe(0) or Cr(0)) and strong π-acceptor ligands. They are characterised by an electrophilic carbene carbon.
Fischer carbenes can be likened to ketones; the alpha-hydrogens are acidic and can be deprotonated by bases to undergo Aldol-like reactions.
Schrock Carbenes
Named after Richard R. Schrock, these involve high-oxidation-state metals (such as Ti(IV) or Ta(V) and non-π-acceptor ligands. In Schrock carbenes, the bonds are polarised toward the carbon, making the centre nucleophilic, much like an organometallic reagent.
N-Heterocyclic Carbenes (NHCs)
N-heterocyclic carbenes (NHCs) are particularly stable singlet carbenes where the carbon is part of a nitrogen heterocycle.
Their stability arises from the mesomeric interaction of the nitrogen lone pairs with the empty p-orbital of the sp² hybridised carbon.
Following the milestone discovery of the first X-ray structure of a stable carbene by Arduengo in 1991, NHCs have become ubiquitous ligands for transition metals.
They are crucial components of catalysts used in olefin metathesis, such as the Grubbs’ catalyst.
Named Reactions Involving Carbenes and Nitrenes
Carbenes and nitrenes are central to several high-value synthetic rearrangements where a group migrates from a carbon to an electron-deficient centre.
Carbene-Based Reactions
- Reimer-Tiemann Reaction: This is a classic application where phenol reacts with chloroform and base to produce salicylaldehyde via a dichlorocarbene intermediate.
- Wolff Rearrangement: An α-diazoketone loses N₂ to form a carbene, which then undergoes a 1,2-shift to produce a ketene. This is the key step in the Arndt-Eistert synthesis, which allows for the conversion of α-amino acids into β-amino acids.
- Corey-Fuchs Reaction: Used to synthesise alkynes from aldehydes through a dibromocarbene intermediate.
Nitrene-Based Rearrangements
Several name reactions involve the migration of a carbon group to an electron-deficient nitrogen atom, often yielding an isocyanate intermediate:
- Hofmann Rearrangement: Converts a primary amide into an amine with one fewer carbon atom.
- Curtius Rearrangement: Pyrolysis of an acyl azide releases N₂ to form a nitrene, which rearranges into an isocyanate. Hydrolysis then yields a primary amine.
- Lossen Rearrangement: The thermal decomposition of hydroxamic acid derivatives to isocyanates.
- Beckmann Rearrangement: Converts an oxime into an amide under acidic conditions. This reaction is vital for the industrial Nylon 6 synthesis, as it produces caprolactam, the precursor to the polymer.
| Feature | Fischer Carbene | Schrock Carbene |
| Metal Oxidation State | Low (e.g. 0) | High (e.g. +4) |
| Nature of Carbon | Electrophilic | Nucleophilic |
| Typical Ligands | π-acceptors (CO) | Non-π-acceptors (CH₃) |
Conclusion: The Power of Electron-Deficient Intermediates
Mastering Carbenes and nitrenes allows chemists to move beyond simple bond-breaking to appreciate the dynamic “dance” of electrons in three-dimensional space.
Whether it is the stereospecific addition of a singlet carbene in a lab-scale synthesis or the massive industrial utility of the Beckmann rearrangement, these intermediates prove that electron deficiency is a powerful engine for chemical innovation.
By understanding the nuances of spin multiplicity and orbital stabilisation, you gain the expert insight needed to predict and control the most complex transformations in organic synthesis.
As an educator, I tell my students that once you recognise the logic behind these fleeting species, the most advanced mechanisms in organic chemistry become as clear as mixing colours on a palette.
Frequently Asked Questions
-
What is the main difference between singlet and triplet carbenes?
The primary difference lies in their spin multiplicity and electronic configuration. In a singlet carbene, the two non-bonding electrons are paired with opposite spins in a single sp² hybrid orbital, making it diamagnetic. In a triplet carbene, the electrons have parallel spins and occupy two different orbitals (sp² and p), giving it a diradical-like, paramagnetic character.
-
Why is the triplet state generally more stable for simple carbenes?
According to Hund’s Rule, the triplet state is typically the ground state for simple carbenes like methylene (:CH₂) because placing electrons in separate orbitals reduces coulombic repulsion. This state is generally about 8 kcal/mol more stable than the singlet state.
-
How do heteroatoms influence carbene stability?
Heteroatoms such as Oxygen, Nitrogen, or Halogens can reverse the stability trend through the mesomeric effect. They donate electron density from their lone pairs into the vacant p-orbital of a singlet carbene, providing significant stabilisation. This is why species like dichlorocarbene (:CCl₂) and N-heterocyclic carbenes (NHCs) have a singlet ground state.
-
What is the difference between Fischer and Schrock carbenes?
Fischer carbenes involve low-oxidation-state metals and π-acceptor ligands, making the carbene carbon electrophilic. Schrock carbenes, however, involve high-oxidation-state metals and non-π-acceptor ligands, resulting in a nucleophilic carbon centre.
-
Why is the addition of singlet carbenes to alkenes stereospecific?
Singlet carbenes react via a concerted (one-step) mechanism because their electrons are already spin-paired. This allows for the simultaneous formation of two new bonds, resulting in the total retention of the alkene’s original configuration (e.g., a cis-alkene produces a cis-cyclopropane).
-
How are nitrenes typically generated in organic synthesis?
The most common method for nitrene generation is the thermal or photochemical decomposition of organic azides (R-N₃). The process releases nitrogen gas (N₂), leaving behind a highly reactive, neutral nitrogen intermediate with a sextet of electrons.
-
What is the difference between a carbene and a nitrene?
Both are neutral, electron-deficient reactive intermediates with a sextet of electrons, but they differ in the central atom. A carbene has a divalent carbon atom bonded to two substituents (R₂C:), while a nitrene has a monovalent nitrogen atom bonded to only one substituent (RN:). Nitrenes are the nitrogen analogues of carbenes and share similar singlet and triplet spin states.
-
What is a carbene in organic chemistry?
A carbene is a neutral reactive intermediate containing a divalent carbon atom with only six valence electrons. It is represented as R₂C: and exists in two spin states, singlet and triplet, which determine whether it reacts in a concerted or stepwise manner.
-
What is the Curtius rearrangement, and how does it involve a nitrene?
The Curtius rearrangement involves the thermal decomposition of an acyl azide, which releases nitrogen gas (N₂) to generate a nitrene intermediate. This nitrene undergoes a 1,2-shift to form an isocyanate, which, upon hydrolysis, yields a primary amine with one fewer carbon atom than the starting acid.
-
Why do triplet carbenes give non-stereospecific products?
Triplet carbenes have two electrons with parallel spins, so they cannot form two bonds simultaneously. The reaction proceeds in a stepwise manner. The first bond forms a radical intermediate, which must undergo spin inversion before the second bond closes. During this delay, free rotation occurs around the single bond, producing a mixture of cis and trans products.
