In the world of chemistry, we are often taught that bonds are either ionic (transfer of electrons) or covalent (sharing of electrons).
However, the reality discovered by Polish-American physical chemist Kazimierz Fajans in 1923 is that most bonds exist on a spectrum.
In this comprehensive guide, we will explore Fajans’ Rule, the essential framework used to predict when a supposedly “ionic” bond starts behaving like a covalent one.
We will break down the concept of polarisation, examine how the size and charge of ions dictate bond character, and settle the famous debate of why AgCl behaves differently from NaCl.
By the end, you’ll be able to predict melting points, solubility, and even the colour of compounds just by looking at their ions.
Table of Contents
1. What is Polarisation? The Key to Covalency
To master Fajans’ rule, one must first understand the concept of polarisation. In a theoretical 100% ionic bond, a metal atom completely transfers its valence electrons to a non-metal, resulting in a perfectly spherical cation and anion held together by electrostatic forces.
In reality, when a cation and an anion approach each other, the cation’s positive charge exerts an attractive force on the anion’s outermost electron cloud while simultaneously repelling its positively charged nucleus. This tug-of-war results in the distortion, deformation, or polarisation of the anion.
If the degree of polarisation is small, the bond remains predominantly ionic; however, if the distortion is large, the electron density becomes concentrated between the two nuclei a state effectively equivalent to the “sharing” seen in covalent bonding.
The Polarising Power Formula
The ability of a cation to distort an anion is termed its polarising power, while the susceptibility of an anion to being distorted is called its polarisability.
Covalent Character ∝ Ionic Potential (ϕ) = (Charge on Cation) / Size (Radius) of Cation
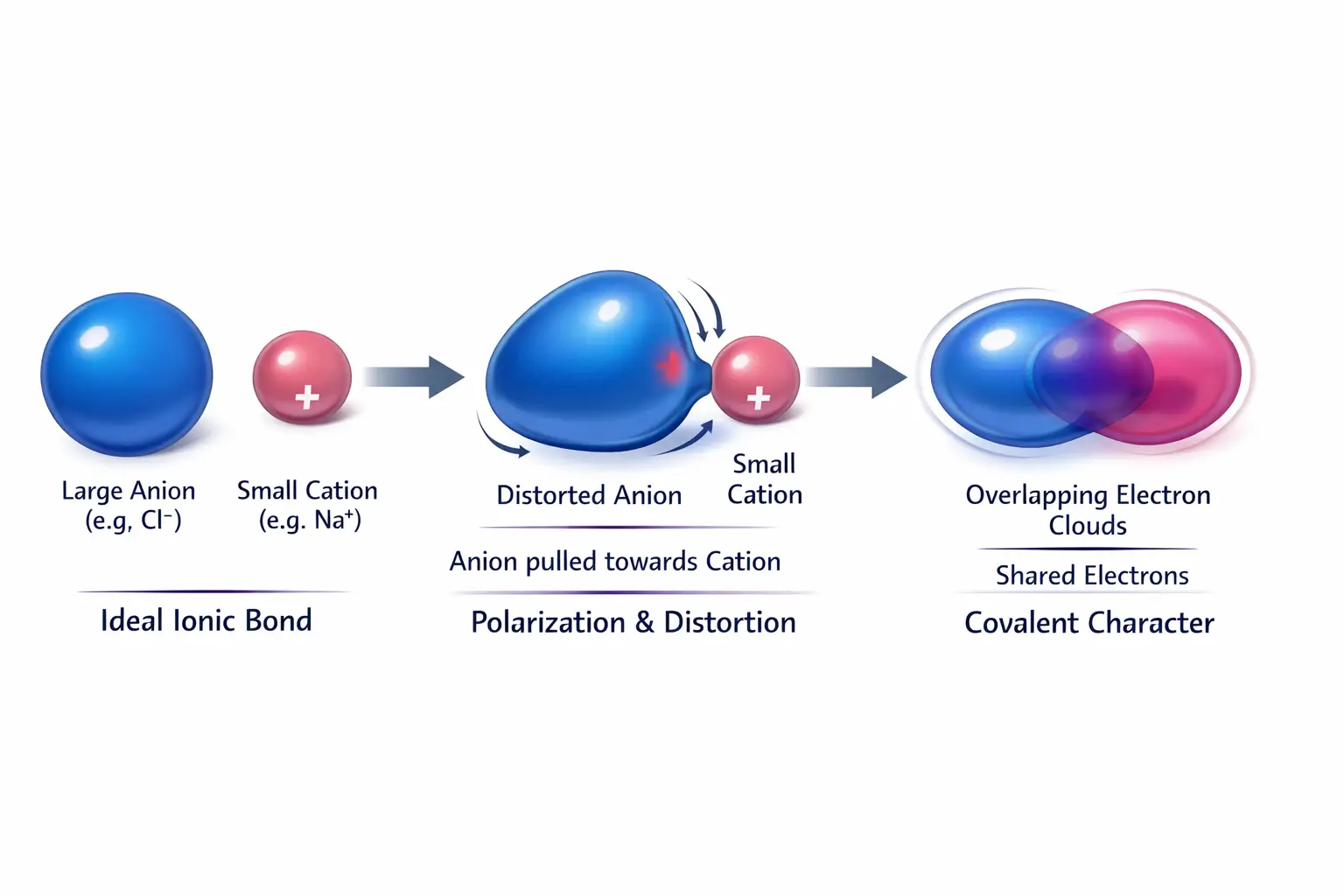
2. The Postulates of Fajans’ Rule
Fajans identified four primary conditions that increase the polarisation of an ion, thereby enhancing its covalent character.
i) Small Cation Size
As we discussed in our guide on [Atomic Radius Trends], smaller atoms have their positive charge concentrated over a much smaller surface area. This high charge density makes small cations incredibly effective at “gripping” and pulling the electron cloud of a nearby anion.
- Comparison: LiCl is significantly more covalent than KCl because the Li⁺ ion is much smaller than the K⁺ ion, giving it superior polarising power.
ii) Large Anion Size
Larger anions are far more “polarisable”. Because their outermost electrons are located further from the nucleus, they are less tightly held and experience more shielding from the inner-shell electrons. These “fluffy” electron clouds are easily stretched towards a cation.
- Comparison: Among lithium halides, LiI is the most covalent because the I⁻ ion is the largest and most easily distorted, whereas LiF is more ionic due to the small, tightly held electron cloud of F⁻.
iii) High Ionic Charge
The electrostatic attraction between ions increases directly with the magnitude of their charges. A cation with a +3 charge will pull an anion’s electrons much more violently than a +1 cation.
- Comparison: Covalency increases in the order NaCl < MgCl₂ < AlCl₃. The Al³⁺ ion’s high charge allows it to polarise the chloride ions far more effectively than Na⁺..
iv) Electronic Configuration: The “Pseudo-Noble Gas” Effect
This is a frequent trap in chemistry examinations. If two cations have nearly identical sizes and charges, the one with a pseudo-noble gas configuration (ns² np⁶ nd¹⁰, having 18 electrons in the outermost shell) will be more polarising than a cation with a standard noble gas configuration (ns² np⁶, with 8 electrons).
- The “Why”: The ten d-electrons in a pseudo-noble gas shell are notoriously poor at shielding nuclear charge. This results in a much higher Effective Nuclear Charge being felt by the anion, leading to greater distortion. For a deeper look at why these d-orbitals behave this way, refer to our article on [Ionisation Energy Exceptions].
3. Is AgCl Ionic or Covalent? (The AgCl vs NaCl Debate)
A common “People Also Asked” query involves comparing AgCl and NaCl. On paper, both appear to be standard ionic salts. They share the same halide anion, and the radii of Ag⁺ and Na⁺ are nearly identical (0.96 Å and 0.95 Å, respectively).
However, their physical properties tell a different story. NaCl is a classic ionic solid with a high melting point of 801°C, while AgCl melts at a much lower 455°C.
The Explanation: According to Fajans’ rule, the Ag⁺ ion possesses a pseudo-noble gas configuration ([Kr] 4d¹⁰), whereas Na⁺ has a noble gas configuration ([Ne]). The poor shielding of the 4d electrons makes Ag+ far more polarising, imparting significant covalent character to AgCl, which weakens the electrovalent forces in the crystal lattice and lowers the melting point.
4. Consequences and Applications of Polarisation
The shift from ionic to covalent character has profound impacts on a compound’s physical and chemical properties.
Lower Melting and Boiling Points
As covalent character increases, the strong, non-directional electrostatic lattice forces are replaced by weaker, directional orbital overlaps. This is evident in the lithium halide series:
- LiF: 870°C (Highly Ionic)
- LiCl: 613°C
- LiBr: 547°C
- LiI: 446°C (Appreciable Covalent Character).
Solubility Trends
A core rule in chemistry is “like dissolves like”. Ionic compounds typically dissolve in polar solvents like water, whereas covalent compounds prefer nonpolar solvents like benzene or carbon tetrachloride. Fajans’ rule explains why LiI is more soluble in organic solvents than LiF and why AgCl is sparingly soluble in water compared to NaCl. To understand how these bond polarities affect molecular shape, see our [VSEPR Theory Guide].
Imparting Colour
Intense polarisation can lead to the absorption of specific wavelengths of light, imparting colour to compounds that might otherwise be colourless.
- Mercury Halides: HgCl₂ is colourless, but the more highly polarised HgI₂ is a vibrant red.
- Lead Halides: PbCl₂ is colourless, while PbI₂ is golden yellow.
Chemical Stability
High polarisation can make certain ions unstable. For example, LiI turns yellow when exposed to air because the small Li+ ion so strongly polarises the large I⁻ ion that it becomes easy to oxidise the iodide into iodine. Similarly, polyatomic oxyanions like carbonates (CO₃²⁻) decompose more easily upon heating when paired with highly polarising cations like Be²⁺.
5. Summary Table: Predicting Bond Character
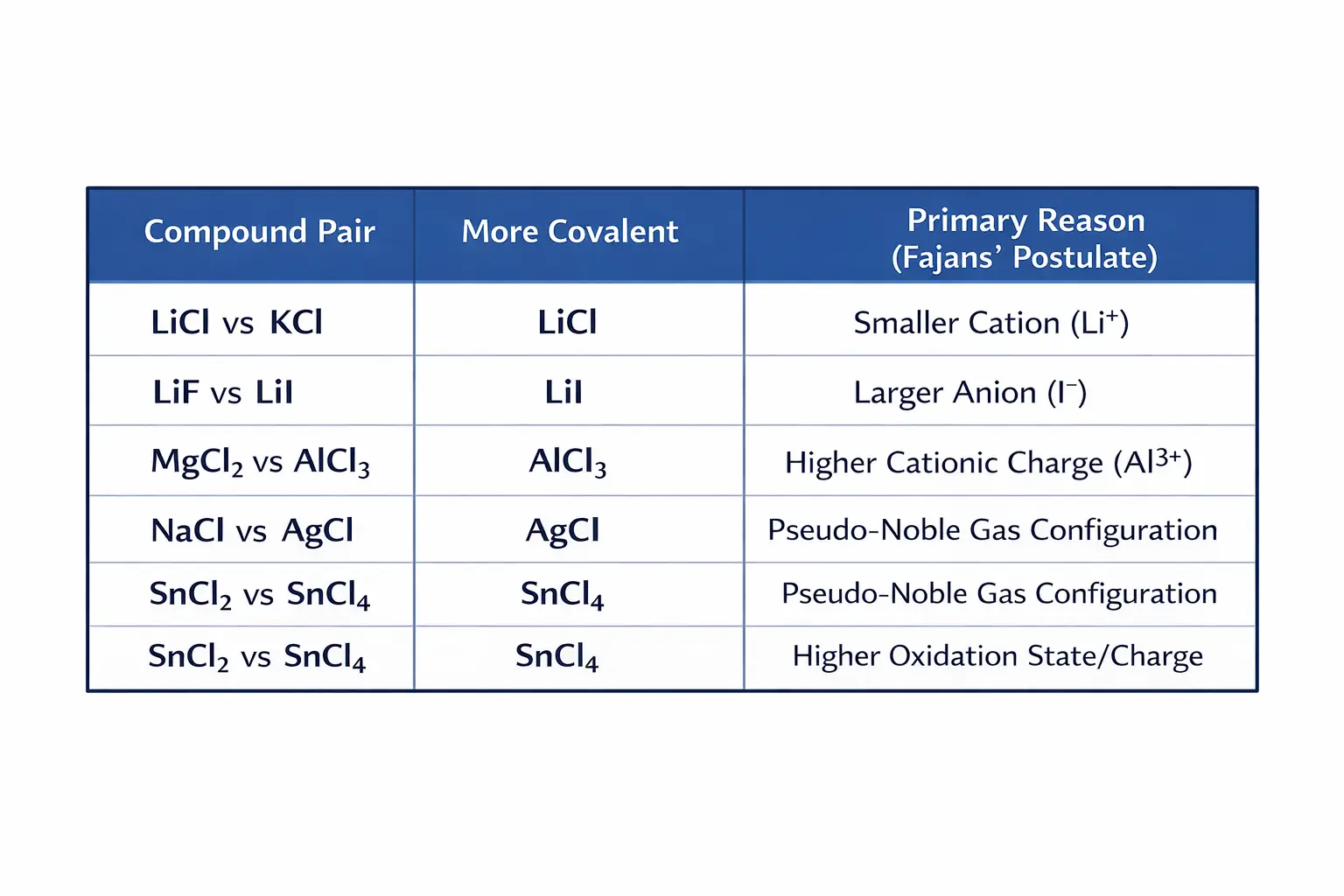
Mastering this molecular geometry guide requires understanding not just 3D shape, but the nature of the bond itself. This table provides quick-reference examples to apply Fajans’ Rule simplified across different chemical scenarios
6. Measuring the Spectrum: Percentage Ionic Character
While Fajans’ rule provides a qualitative “rule of thumb,” chemists use dipole moments for a quantitative measure. No bond is 100% ionic or 100% covalent; we simply call a compound “ionic” if its ionic character is high.
The percent ionic character can be calculated by comparing the observed dipole moment (μobs) to a theoretical dipole moment (μcalc) calculated by assuming 100% charge separation:
% Ionic Character = (μobserved / μcalculated) × 100
For example, HCl is often called a covalent gas, yet it possesses roughly 17–18% ionic character due to the electronegativity difference between Hydrogen and Chlorine. You can learn more about how these electronegativity differences are calculated in our article on the Evolution of the Periodic Table.
Conclusion
Mastering Fajans’ rule is a vital milestone for any chemistry student. It allows you to look past the simplified labels of “ionic” or “covalent” and understand the nuanced reality of electron cloud distortion.
By considering the size, charge, and electronic configuration of ions, you can accurately predict properties like melting points, solubility, and even the colour of chemical compounds.
Ready to see how these bonds affect the shapes of molecules?
- Dive into our [VSEPR Theory Guide] to learn about molecular geometry.
- Explore why certain atoms resist this polarisation in [Ionisation Energy Exceptions].
- Discover the historical context of these rules in the [Evolution of the Periodic Table].
Frequently Asked Questions
-
What is Fajans’ Rule used for in chemistry?
Ans: Fajans’ Rule is used to predict the covalent character in a chemical bond that is primarily ionic. It explains why certain “ionic” compounds show properties of covalent bonds, such as low melting points and insolubility in water, due to the polarisation of the anion by the cation.
-
Why is AgCl more covalent than NaCl?
Ans: Both Ag+ and Na+ have similar sizes and the same +1 charge. However, Ag+ has a pseudo-noble gas configuration (ns2 np6 nd10), which provides poor shielding of the nucleus. This results in Ag+ having a higher effective nuclear charge and greater polarising power, making AgCl more covalent than NaCl.
-
Which has more covalent character: LiCl or KCl?
Ans: LiCl has more covalent character. According to Fajans’ Rule, a smaller cation has a higher charge density and more polarising power. Since Li+ is significantly smaller than K+, it distorts the electron cloud of the chloride ion (Cl-) more effectively.
-
How does the size of the anion affect Fajans’ Rule?
Ans: A larger anion is more polarisable. As the size of an anion increases, its outermost electrons are further from the nucleus and more shielded, making the electron cloud “fluffy” and easier for a cation to distort. For example, LiI is more covalent than LiF because I- is much larger than F-.
-
Is AgCl ionic or covalent?
Ans: Technically, AgCl is an ionic compound with significant covalent character. While it forms a crystal lattice like other salts, the high polarisation of the Ag+ ion leads to electron sharing. This covalency is why AgCl has a lower melting point than NaCl and is insoluble in water.
-
Does a higher ionic charge increase covalency?
Ans: Yes. As the positive charge on a cation or the negative charge on an anion increases, the electrostatic attraction and subsequent polarisation increase. Therefore, AlCl3(+3) is much more covalent than MgCl2(+2) or NaCl (+1).

