“To a chemist, sunscreen is much more than a holiday essential; it is a sophisticated photo-protective topical product designed to shield our DNA from invisible attackers.”
While many consumers take the white cream for granted, the Chemistry of sunscreen involve a complex series of molecular interactions that prevent our skin from turning “lobster red” under the summer sun.
These formulations are essentially your skin’s personal bodyguard against invisible attackers known as ultraviolet (UV) radiation.
Understanding the physical chemistry behind these mechanisms is the first step in making smarter, scientifically backed choices for your skin’s long-term health.
How sunscreen works depends on its filters. Chemical sunscreens (organic) act like sponges, absorbing UV rays and converting them into harmless heat. Physical sunscreens (mineral) act like shields, primarily absorbing and scattering UV radiation. SPF measures protection against UVB rays, with SPF 30 filtering 97% of radiation.
Table of Contents
1. Understanding the Enemy: UVA, UVB, and UVC Radiation
The sun produces three distinct forms of ultraviolet radiation, categorised by their wavelengths and the energy they carry.
While we often discuss “sunlight” as a single entity, the Chemistry of sunscreen must account for the varied ways these rays interact with human biology.
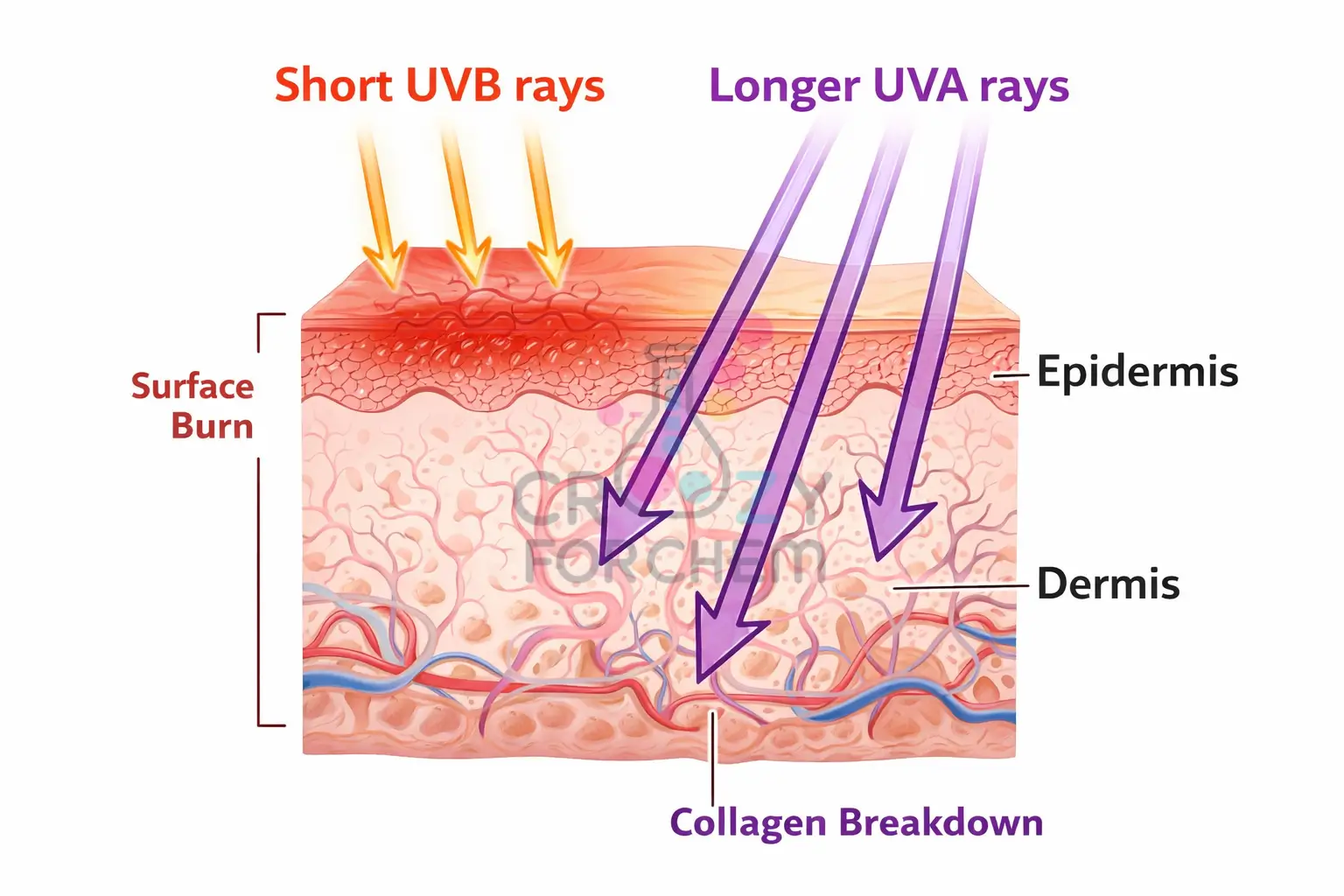
- UVC (100–280 nm): This is the highest energy and most harmful form of UV radiation. Fortunately, it is entirely absorbed by the Earth’s ozone layer and does not reach the surface.
- UVB (280–320 nm): Responsible for approximately 5% of the UV radiation reaching Earth, these rays are the primary cause of sunburn. UVB rays possess shorter wavelengths that hit the surface of the skin, causing direct DNA damage and triggering the production of melanin, which results in a tan. They are also essential for the synthesis of Vitamin D.
- UVA (320–400 nm): Accounting for roughly 95% of the UV radiation that reaches us, UVA has longer wavelengths that penetrate much deeper into the connective tissue. These rays are “sneakier” as they do not cause the immediate pain of a burn, but they generate reactive species that cause indirect DNA damage, leading to premature ageing, wrinkles, and liver spots. Unlike UVB, UVA rays can pass through window glass.
Just as we identify hazardous adulteration in food to prevent long-term toxicity, protecting our DNA from UV rays is a critical safety step.
2. Chemical Absorbers: The “Molecular Sponge”
Chemical vs physical sunscreen represents two fundamentally different scientific approaches to photo-protection. Chemical sunscreens, often referred to as “organic” because they are carbon-based, act as molecular factories that transform dangerous energy into harmless forms.
The Mechanism of Action
Chemical filters such as avobenzone, oxybenzone, octisalate, and octinoxate work through a process called absorption. At a molecular level, these chemicals have electrons that sit in a stable, low-energy state.
When UV radiation of the correct wavelength hits these molecules, the electrons absorb the energy and jump to a higher, “excited” state.
The way filters absorb photons is a physical-chemical process, similar to how the Chemistry of painkillers involves molecular binding to receptors.
Energy Conversion and Relaxation
Because molecules are unstable in this excited state, they must release the energy to return to their “ground state”.
This process, known as relaxation, involves converting the absorbed UV radiation into harmless heat, which is then dissipated from the skin’s surface.
This micro-reaction happens in microseconds and can repeat thousands of times throughout the day.
While some might worry about this heat, the amount generated is minimal; converting all the UV hitting your skin into heat would raise its temperature by less than one degree.
The “Wait Time” Requirement
A key disadvantage of chemical formulas is that they must be absorbed into the skin’s surface to create their protective shield from within.
Consequently, dermatologists and manufacturers recommend applying these products 15–30 minutes before sun exposure to allow the filters to become fully active.
Unlike the immediate reaction of pH levels we see in kitchen experiments, molecular absorption takes time to settle on the skin.

3. Physical Blockers: The “Mineral Shield”
Physical sunscreens, also known as mineral sunscreens or sunblocks, utilise inorganic minerals—primarily zinc oxide and titanium dioxide.
Reflection vs. Absorption: The Great Myth
Historically, the cosmetics industry and many consumers believed that physical blockers worked solely by sitting on top of the skin and reflecting or scattering UV rays away like a mirror.
However, modern physical chemistry has debunked this as a partial truth. While these minerals do scatter and reflect some light, recent research confirms they protect the skin primarily through absorption up to 95%, just like chemical filters.
The “White Cast” Chemistry
Traditional mineral sunscreens are notorious for leaving a white, chalky residue on the skin. This occurs because the mineral particles were historically large enough to scatter and reflect not just UV rays, but also visible light.
Nanoparticles: The Modern Solution
To improve “cosmetic elegance,” manufacturers now use nanoparticles or micronised formulations, where the minerals are ground into particles less than 200 nm in diameter.
These particles are so small that they do not scatter visible light, making them appear “invisible” or transparent on the skin while remaining highly effective at absorbing UV radiation.
Zinc oxide is particularly valued as a “gold standard” because it provides comprehensive protection across both the UVA and UVB spectrums.
Comparison Table
| Feature | Chemical Sunscreen (Organic) | Physical Sunscreen (Mineral) |
| Mechanism | Absorbs UV & converts to heat | Reflects & absorbs UV rays |
| Ingredients | Avobenzone, Oxybenzone | Zinc Oxide, Titanium Dioxide |
| Wait Time | 15–20 minutes | Immediate protection |
| Texture | Usually clear/invisible | May leave a white cast |
| Skin Type | Better for water resistance | Better for sensitive skin/kids |
4. Decoding SPF: What the Number Actually Measures
The SPF meaning (Sun Protection Factor) is frequently misunderstood as a linear timer for sun exposure.
In reality, it is a measure of how much solar energy is required to cause a sunburn on protected skin compared to unprotected skin.
- UVB Specificity: SPF exclusively measures a product’s ability to protect against UVB rays—the rays responsible for burning. It does not quantify protection against UVA radiation.
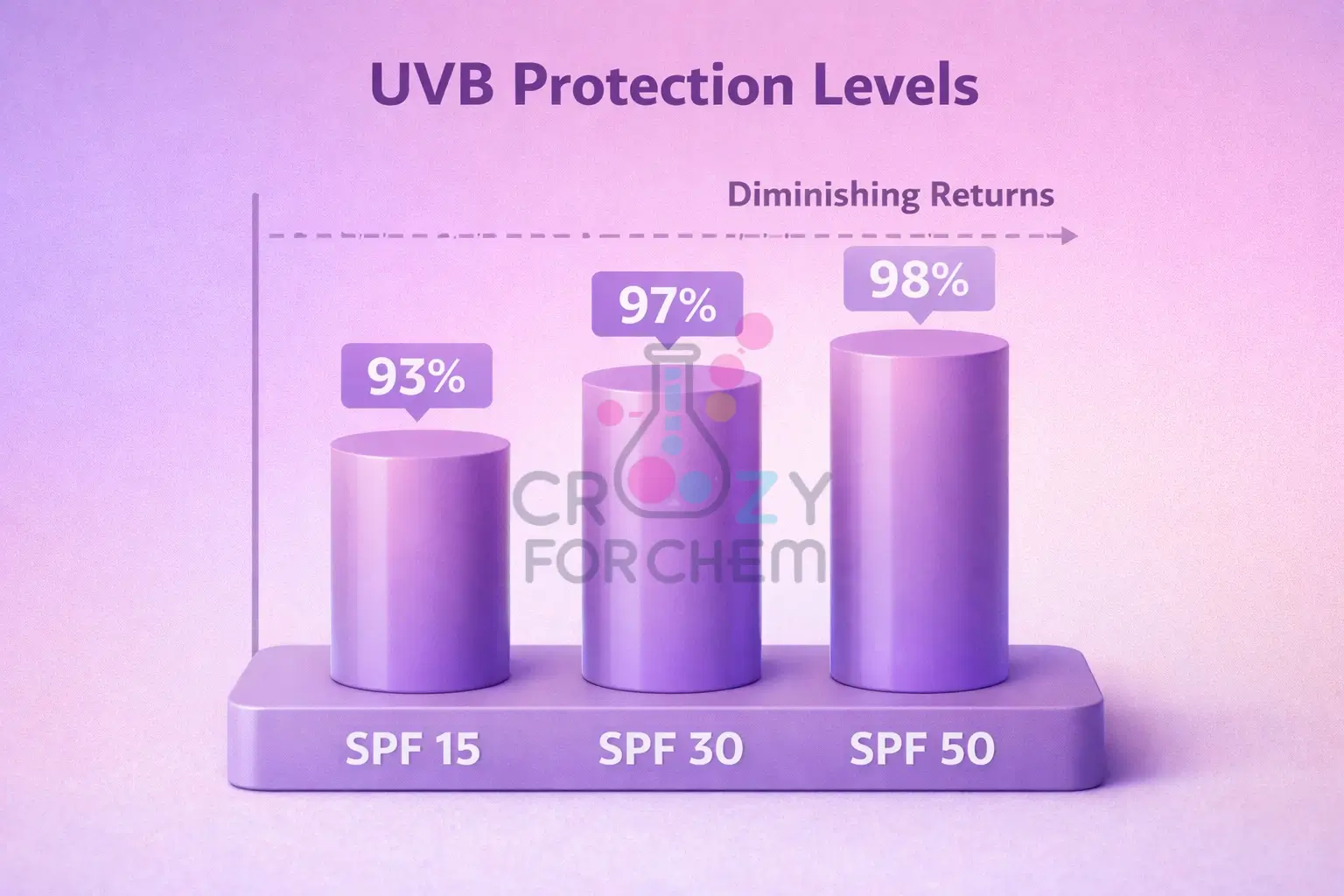
- The Percentage Breakdown: The protection offered by different SPF numbers is not a simple double-up.
- SPF 15 filters out approximately 93% of UVB rays.
- SPF 30 filters out approximately 97%.
- SPF 50 filters out 98%.
- The “Broad Spectrum” Necessity: Because high SPF only guarantees UVB protection, it is vital to select a product labelled “Broad Spectrum”. This ensures the formula also includes ingredients that screen for UVA, such as avobenzone or zinc oxide.
5. Formulation Science: Stability and Substantivity
The Chemistry of sunscreen also involves ensuring the product remains effective under harsh environmental conditions.
Photounstability
Some organic filters are photounstable, meaning they can break down irreversibly when exposed to sunlight.
Avobenzone, the primary UVA filter in many chemical sunscreens, is a prime example; its chemical bonds can break as it absorbs UV light, causing it to lose effectiveness.
To counter this, chemists add photostabilisers like octocrylene to help prevent these ingredients from decomposing.
Substantivity and Water Resistance
Substantivity refers to a sunscreen’s ability to remain on the skin despite swimming, sweating, or physical rubbing. No sunscreen is truly “waterproof”; instead, the FDA regulates the term “water-resistant”.
To be labelled as such, a product must retain its SPF rating after either 40 or 80 minutes of water immersion.
6. Safety and Environmental Considerations
While health experts agree that the benefits of sunscreen usage in preventing skin cancer far outweigh the risks, there are specific chemical and environmental factors to consider.
- Inhalation Risks: Sprays and aerosol sunscreens introduce the risk of inhalation. For this reason, dermatologists often recommend spraying the product into your hands before applying it to your face to avoid breathing in the chemical particles.
- Environmental Impact: Certain chemical filters, specifically oxybenzone and octinoxate, have been linked to coral reef deterioration and bleaching. This has led to bans on these ingredients in eco-sensitive regions like Hawaii and Key West. Mineral-based sunscreens are generally considered a safer, “reef-friendly” alternative. Chemical safety isn’t just about our skin… this is why understanding the risks of mixing bleach and ammonia and other reactive chemistries is so vital.
- Sensitive Skin and Kids: Because chemical filters rely on a reaction within the skin, they are more likely to cause irritation or allergic responses. Mineral sunscreens (physical blockers) are often recommended for young children and those with sensitive skin because they sit primarily on the surface.
7. Application Guide: The 2-mg Rule
The most significant failure in how sunscreen works is not the chemistry, but the application. Most people apply only 25–50% of the recommended amount, which can turn an SPF 30 product into an effective SPF 15.
- The “Shot Glass” Rule: For full-body coverage, you need approximately one ounce (about 30 ml or a full shot glass).
- The “Three-Finger” Rule: For the face and neck alone, you should use about a quarter of a teaspoon, which is roughly the amount that fits on three fingers lined up.
- Reapplication: Regardless of the SPF or the “water-resistant” label, all sunscreens should be reapplied every two hours when outdoors, as sweat and movement gradually wear away the protective molecular film.

Conclusion: Mastering the Science of Protection
Understanding how sunscreen works transforms a messy chore into a vital scientific tool for health.
By mastering the Chemistry of sunscreen, you can navigate the marketing hype and choose the right combination of “molecular sponges” and “mineral shields” to protect your skin’s DNA.
Whether you opt for the cosmetic elegance of nanoparticle-based mineral blockers or the high-stability hybrid formulas, the best sunscreen is ultimately the one you will apply liberally and consistently every day.
Frequently Asked Questions (FAQs)
-
What is the primary chemical difference between physical and chemical sunscreens?
Physical blockers (mineral), such as Zinc Oxide, act primarily as a mineral shield to reflect and scatter UV rays, providing immediate protection. In contrast, chemical blockers (organic), like Avobenzone, act as molecular sponges that absorb UV radiation and convert it into low-level heat through a process called “internal conversion”.
-
Does a higher SPF rating mean I can stay in the sun all day?
No, a higher SPF does not grant all-day protection. SPF specifically measures protection against UVB rays based on solar intensity, not time. Factors such as sweat, water immersion, and the natural “photodegradation” of chemical filters mean that even SPF 50 must be reapplied every two hours to maintain its protective molecular film.
-
Does “waterproof” sunscreen actually exist?
Legally and chemically, no. The term “waterproof” is considered a marketing misnomer and is prohibited by most regulatory bodies. Products are now strictly labelled as “water-resistant” for either 40 or 80 minutes. This indicates the duration the SPF remains effective while the wearer is swimming or sweating.
-
Why is UV exposure significantly higher in the Southern Hemisphere?
Peak UV levels in the Southern Hemisphere can be up to 40% higher than at similar northern latitudes. This is due to three chemical and orbital factors: the Earth is 7% closer to the sun during the southern summer, the ozone layer is naturally thinner in these regions, and there is significantly less atmospheric pollution to scatter incoming UV radiation.
-
Can I safely use sunscreen past its expiry date?
It is not recommended. Over 2–3 years, the active organic filters and emulsifiers in sunscreen can separate or undergo chemical degradation. This breakdown significantly reduces the actual SPF protection, potentially leaving your skin vulnerable to DNA damage despite the product appearing normal in texture.
-
Should I apply sunscreen or insect repellent first?
Always apply sunscreen first and insect repellent second. For the sunscreen’s molecular absorption to work effectively, it must be in direct contact with the skin. Be aware that DEET, a common ingredient in repellents, can reduce a sunscreen’s efficacy by up to 30%, making frequent reapplication essential.
-
Is the “white cast” from mineral sunscreen dangerous?
No, the “white cast” is simply the result of large Titanium Dioxide particles scattering visible light. Modern nanoparticles have solved this by reducing particle size so they remain invisible to the eye while still absorbing UV rays. This is a purely aesthetic chemical property and does not affect safety.

