Chemistry is often perceived as a series of complex formulas confined to a laboratory, but it is actually the “central science” that explains how the world around us works.
It is the scientific study of the properties, composition, and behaviour of matter and the changes it undergoes.
From the moment we wake up and use toothpaste to the fuel that powers our commute, chemistry in everyday life is an invisible force driving innovation, health, and sustainability.
By understanding the fundamental framework of chemistry, we can better comprehend diverse fields such as botany, geology, ecology, and pharmacology.
This expert guide explores how chemical principles permeate every aspect of our existence, ensuring our quality of life, safety, and comfort.
Table of Contents
1. The Domestic Laboratory: Chemistry in the Kitchen
The kitchen is perhaps the most accessible chemistry laboratory in any home. Every time you prepare a meal, you are performing chemical reactions that transform the taste, texture, and appearance of raw ingredients.
The Magic of Cooking
When we apply heat to food, we trigger complex chemical transformations. For instance, when onions turn golden or dough rises, specific reactions are at play.
One of the most significant is the Maillard reaction, which occurs when proteins and sugars are heated together. This reaction is responsible for the delicious brown crust and rich flavour found in roasted meats and toasted bread.
Furthermore, acids and bases play a critical role in balancing flavours and textures. Adding vinegar to milk causes proteins to coagulate into curd, while baking soda releases carbon dioxide gas, helping cakes and bread to rise.
Even the act of refrigeration is a chemical decision, as it slows down bacterial reactions to extend the freshness of food.
Food Additives and Preservation
The food we consume is made entirely of chemical compounds, including essential nutrients like carbohydrates, proteins, fats, vitamins, and minerals.
Beyond these, manufacturers use various chemical additives to enhance food quality and safety:
- Artificial Sweeteners: These provide sweetness without the high-calorie count of sucrose. Aspartame is a popular choice for cool drinks and ice cream, being roughly 200 times sweeter than sugar. Other potent examples include alitame, which is 2000 times sweeter than sucrose.
- Preservatives: These prevent spoilage by inhibiting the growth of microorganisms. Common examples include sodium benzoate, salts, and sodium metabisulfite.
- Antioxidants: Chemicals like Butylated Hydroxytoluene (BHT) and Butylated HydroxyAnisole (BHA) prevent food from spoiling by stopping oxidation.
Table 1: Common Chemicals in Food Materials
| Category | Examples | Primary Function |
|---|---|---|
| Artificial Sweeteners | Aspartame, Sucralose, Alitame | Provide sweetness without calories |
| Preservatives | Sodium Benzoate, Sodium Meta-bisulphite | Prevent microbial growth and spoilage |
| Antioxidants | BHT, BHA | Prevent spoilage due to oxidation |
| Nutritional Supplements | Vitamins, Minerals | Improve nutritional value |
2. The Science of Beauty: Chemistry in Cosmetics
Cosmetics are not merely superficial products; they are sophisticated chemical formulations designed to protect the skin and enhance appearance.
Understanding chemistry in everyday life allows consumers to make informed choices about the products they apply to their bodies.
Key Cosmetic Ingredients
Most cosmetic products, from lipsticks to moisturisers, rely on several categories of chemicals to function effectively:
- Emulsifiers: These chemicals, such as potassium cetyl sulfate, increase the stability of emulsions, preventing oil and water from separating.
- Thickeners: Ingredients like cetyl alcohol and stearic acid give products an appealing and consistent texture.
- Emollients: These soften the skin by preventing water loss; common examples include glycerine and zinc oxide.
- Preservatives: Because many cosmetics contain water, they are prone to microbial growth. Preservatives like parabens (methylparaben, ethylparaben) and salicylic acid are essential to increase shelf life and ensure safety.
Sun Protection Mechanisms
One of the most vital applications of chemistry in personal care is sun protection. Sunscreens are designed to shield the skin from harmful ultraviolet (UV) radiation.
There are two main types of rays: UV-A, which penetrates deeply to cause wrinkles and age spots, and UV-B, which causes surface sunburn.
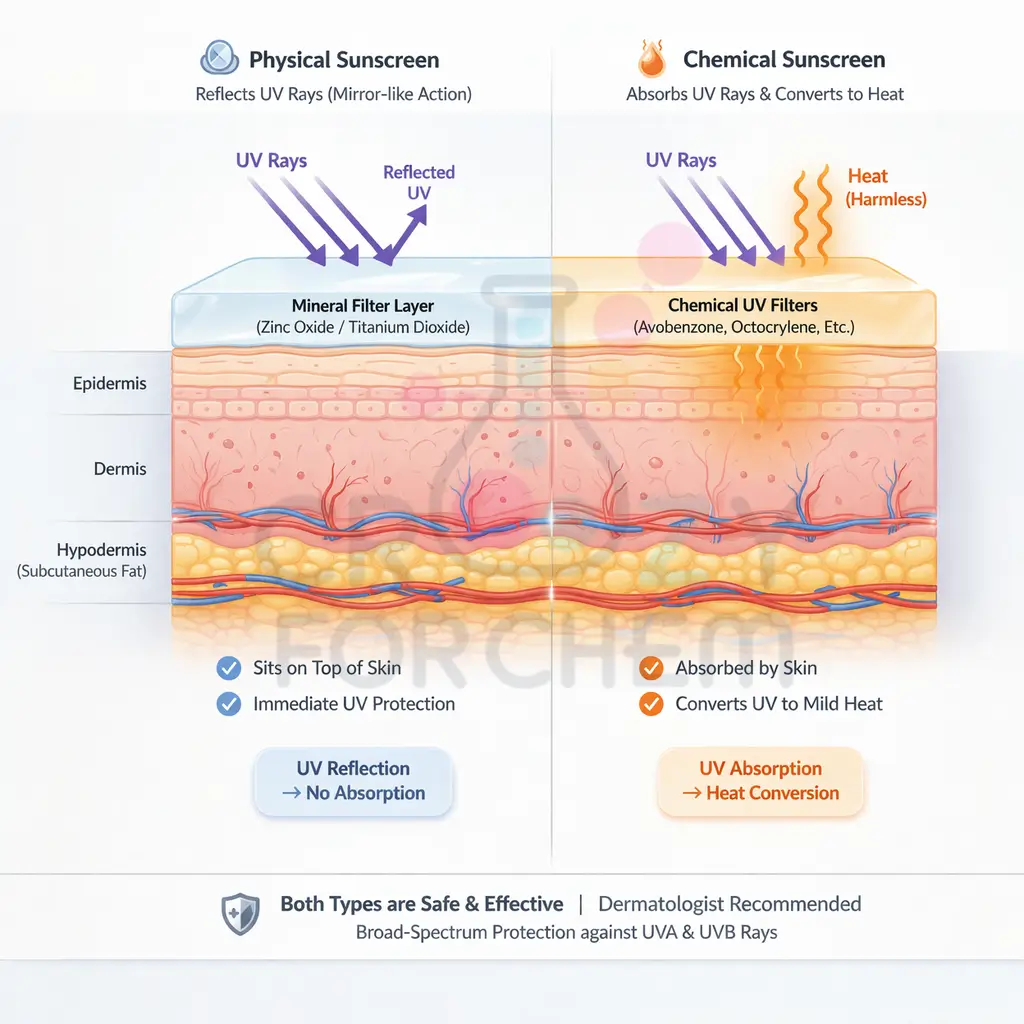
Table 2: Physical Sunblock vs. Chemical Sunscreen
| Feature | Physical Sunblock | Chemical Sunscreen |
|---|---|---|
| Mechanism | Reflective nature; sits on top of the skin | Absorbs UV rays and converts them to heat |
| UV Protection | Blocks both UV-A and UV-B | Primarily protects against UV-B (unless broad spectrum) |
| Common Ingredients | Zinc Oxide, Titanium Dioxide | Organic compounds like oxybenzone |
3. Cleansing Agents: Soaps and Detergents
Cleaning is a chemical process aimed at removing dirt, grease, and microbes from surfaces. Both soaps and detergents are surfactants (surface-active agents) that reduce the surface tension of water, allowing it to spread and wet surfaces more effectively.
The Chemistry of Soap
Soaps are traditional cleansing agents produced through saponification, a reaction between fats (glyceryl esters of fatty acids) and an alkali like sodium hydroxide. A soap molecule has two distinct ends:
- Hydrophilic Head: This end is “water-loving” and clings to water molecules.
- Hydrophobic Tail: This end is “water-hating” and attaches to oil and dirt.
When washing, these molecules form micelles, spherical clusters where the hydrophobic tails trap dirt in the centre, allowing it to be rinsed away by water.
Synthetic Detergents
Detergents are petroleum-based synthetic cleansers that offer an advantage over soap because they remain effective in hard water. They are classified into three types:
- Anionic Detergents: These have a negatively charged head (e.g., sodium lauryl sulphate) and are widely used in laundry detergents and toothpastes.
- Cationic Detergents: These have a positively charged head (e.g., cetyltrimethylammonium bromide) and possess germicidal properties; they are commonly found in hair conditioners.
- Non-ionic Detergents: These lack ions and are typically used in liquid dishwashing detergents.
Table 3: Soap vs. Hand Sanitiser
| Feature | Soap and Water | Hand Sanitizer |
|---|---|---|
| Primary Goal | Physical removal of dirt and microbes | Killing or neutralizing microbes on contact |
| Key Mechanism | Micelle formation traps and lifts contaminants | Alcohol (ethanol) disrupts microbial cell walls |
| Best Used For | Visibly greasy or soiled hands | Quick disinfection when water is unavailable |
🚨 PRO-TIP: Dangerous Chemical Mixes
A command of chemistry can help keep you safe by teaching you which household chemicals are dangerous to mix. Never mix bleach with ammonia or acids (like vinegar). Such combinations can trigger chemical reactions that release toxic gases, which are extremely hazardous if inhaled. Always read labels and ensure proper ventilation when using cleaning agents.
4. Chemistry in Health and Medicine
The impact of chemistry in everyday life is perhaps most profound in the field of medicine. Medicines are chemical substances with low molecular masses (100–500 u) that interact with biological targets such as proteins, nucleic acids, and lipids to produce a therapeutic response.
Therapeutic Actions of Drugs
Chemistry has provided a vast array of life-saving and pain-relieving substances:
- Analgesics: Used to reduce various types of pain (e.g., aspirin, paracetamol).
- Antacids: Chemicals like ranitidine or cimetidine are used to treat hyperacidity by blocking histamine receptors in the stomach, thereby reducing acid production more effectively than simple neutralisers like sodium hydrogen carbonate.
- Antibiotics: Discovered by Alexander Fleming, antibiotics like penicillin are produced by microorganisms to kill or inhibit the growth of infectious bacteria.
- Antiseptics: Applied to living tissues to prevent the growth of microbes on wounds or cuts (e.g., Dettol, iodine).
- Tranquillizers: Used to treat stress and mental diseases by affecting the transmission of messages between nerves and receptors.
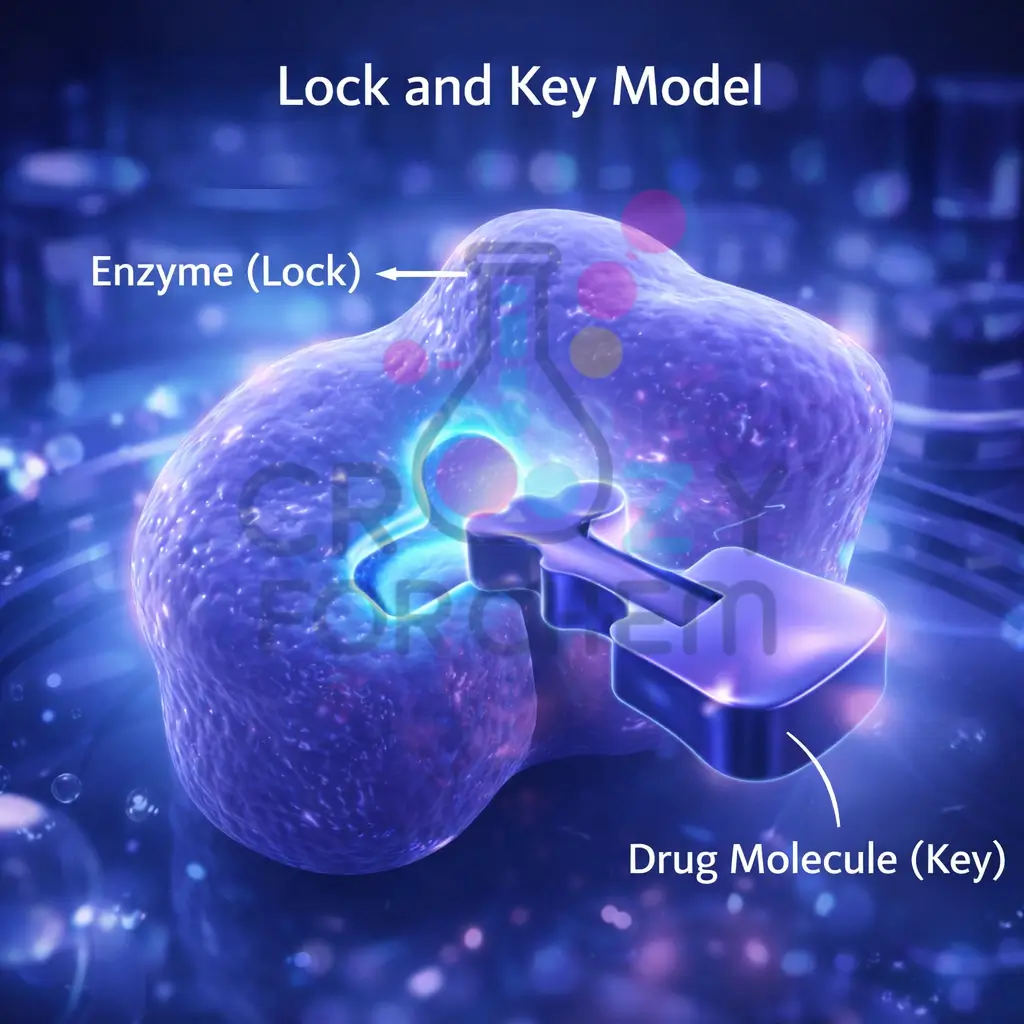
Drug-Target Interaction
Medicinal chemists study how drugs interact with enzymes and receptors. Enzymes act as biological catalysts; drugs can inhibit them by blocking their active sites, preventing the natural substrate from binding.
Similarly, drugs can bind to receptors to either mimic natural chemical messengers (agonists) or block them (antagonists).
5. Chemistry Beyond the Home: Industry and Environment
The influence of chemistry extends to the materials we use and the environment we inhabit.
Textiles and Construction
The textile industry relies on chemistry for both natural and synthetic fibres. While cotton and wool are natural, they undergo chemical treatments for smoothing and dyeing. Synthetic fibres like nylon and polyester are entirely the products of chemical polymers.
In construction, chemical products such as cement, concrete, and steel are fundamental. Modern buildings also use heat-resistant polymers for piping, electrical switches, and floor tiles, ensuring strength and safety.
Fuel and Energy
Our ability to travel by land, sea, and air depends on the chemistry of fuels. Refining crude oil produces petrol, diesel, and LPG. Furthermore, electrochemistry governs the operation of the batteries in our mobile phones, laptops, and electric vehicles, where chemical energy is converted into electrical energy.
Green Chemistry and Sustainability
Green Chemistry is an essential field focused on reducing the environmental impact of human activity. It involves monitoring pollutants in the air, water, and soil and developing sustainable technologies.
A key success of this field was the replacement of ozone-depleting Chlorofluorocarbons (CFCs) in refrigerators with safer alternatives. Today, chemists are also working on developing biodegradable plastics and renewable energy sources like solar cells and biofuels.
6. Conclusion
Chemistry is far more than just equations; it is the “invisible architect” shaping our physical world.
Recognising that everything from the food we eat to the emotions governed by neurotransmitters in our brains is chemical in nature transforms our perspective of science.
By appreciating chemistry in everyday life, we gain the analytical skills to make safer, more sustainable, and more informed decisions for ourselves and our society.
Whether it is the Maillard reaction in your frying pan or the surfactants in your shampoo, chemistry is truly life in motion.
Frequently Asked Questions
-
What are 5 examples of chemistry in everyday life?
Chemistry is all around us. The five most common examples are:
Cooking: The Maillard reaction, which transforms the taste and colour of food.
Cleaning: The formation of micelles by soaps and detergents to lift away dirt.
Digestion: The use of enzymes and hydrochloric acid (HCl) in the stomach to break down food.
Medicine: How painkillers interact with specific receptors in the body.
Photosynthesis: The process by which plants convert sunlight and CO2 into glucose. -
Why is chemistry called the “Central Science”?
Chemistry is known as the “Central Science” because it bridges other natural sciences, such as physics, biology, geology, and environmental science. Without understanding chemical reactions, we cannot fully comprehend human biology or develop new technologies like advanced mobile batteries.
-
How is chemistry used in the kitchen?
The kitchen is essentially a mini-laboratory. Whether you are fermenting milk into yoghurt, using baking soda to make a cake rise (CO2 release), or adding salt to water to increase its boiling point, you are applying chemistry. Even the reason “why onions make you cry” is rooted in complex sulphur-based chemical reactions.
-
Are all chemicals in our daily life harmful?
No, that is a common myth. Everything in the world, from water (H2O) to the oxygen we breathe (O2), is a chemical. Nothing is truly “chemical-free.” The difference lies in whether a substance is Natural vs Synthetic and, more importantly, the Dosage. Some chemicals save lives (Medicines), while others can be dangerous if mixed incorrectly (like Bleach + Ammonia).
-
How does chemistry help in improving our health?
Medicinal chemistry is the reason we have life-saving drugs today. Antibiotics fight infections, antacids relieve acidity, and sunscreens protect our skin from UV-induced cancers. Chemistry allows us to understand exactly how a compound will affect our cell receptors.
-
What is the importance of chemistry in cosmetics?
In cosmetics, chemistry ensures both product stability and safety. Emulsifiers keep oil and water mixed, preservatives prevent microbial growth, and pH balancers ensure the product does not irritate the skin.

