In the study of organic chemistry, a molecular formula often provides only a fraction of the story.
While a formula tells us which atoms are present, it does not dictate how they are connected.
This leads to a fascinating phenomenon: Structural Isomerism.
As a chemistry educator, I often tell my students that structural isomers are like a set of Lego bricks; you can take the exact same number and type of bricks (the molecular formula) and build a car, a house, or a tower (the structural arrangement).
Structural Isomerism at a Glance
- Definition: Compounds with the same molecular formula but different atomic connectivity (structural sequence).
- Core Difference: It is not caused by bond rotation; it requires a physical change in the structure.
- Key Types:
- Chain: Variation in carbon skeleton (e.g., n-butane vs. isobutane).
- Position: Different location of the functional group (e.g., 1-propanol vs. 2-propanol).
- Functional Group: Different families of compounds (e.g., Ethanol vs. Dimethyl ether).
- Metamerism: Unequal alkyl group distribution (e.g., Diethyl ether vs. Methyl propyl ether).
- Exam Pro-Tip: Always calculate the Degree of Unsaturation (DoU) and use IUPAC nomenclature to confirm if two structures are truly different isomers.
Table of Contents
What is Structural Isomerism? (Constitutional Isomerism)
Defining the Phenomenon
Structural Isomerism, or constitutional isomerism, occurs when two or more organic compounds possess the same molecular formula but differ in their structural formula or connectivity.
In simpler terms, while the count of every atom is identical, the sequence in which these atoms are bonded together is entirely different.
This difference in connectivity leads to distinct physical and chemical properties for each isomer.
The Difference Between Isomers and Rotations
It is a common pitfall for students to mistake a simple molecular rotation for a new isomer.
Structural Isomerism requires a fundamentally different arrangement of atoms in space, excluding any variations caused by the molecule rotating as a whole or rotating about particular single bonds.
In organic molecules, there is complete free rotation around all carbon-carbon single bonds.
Imagine a model of a molecule in your hands: if you can make a seemingly “different” shape just by twisting a bond, it is still the same molecule.
To create a true structural isomer, you would have to physically disassemble the molecule and rebuild it in a new order.
The Core Types of Structural Isomerism
Organic chemistry classifies structural isomers into several major categories based on how the atoms are rearranged.
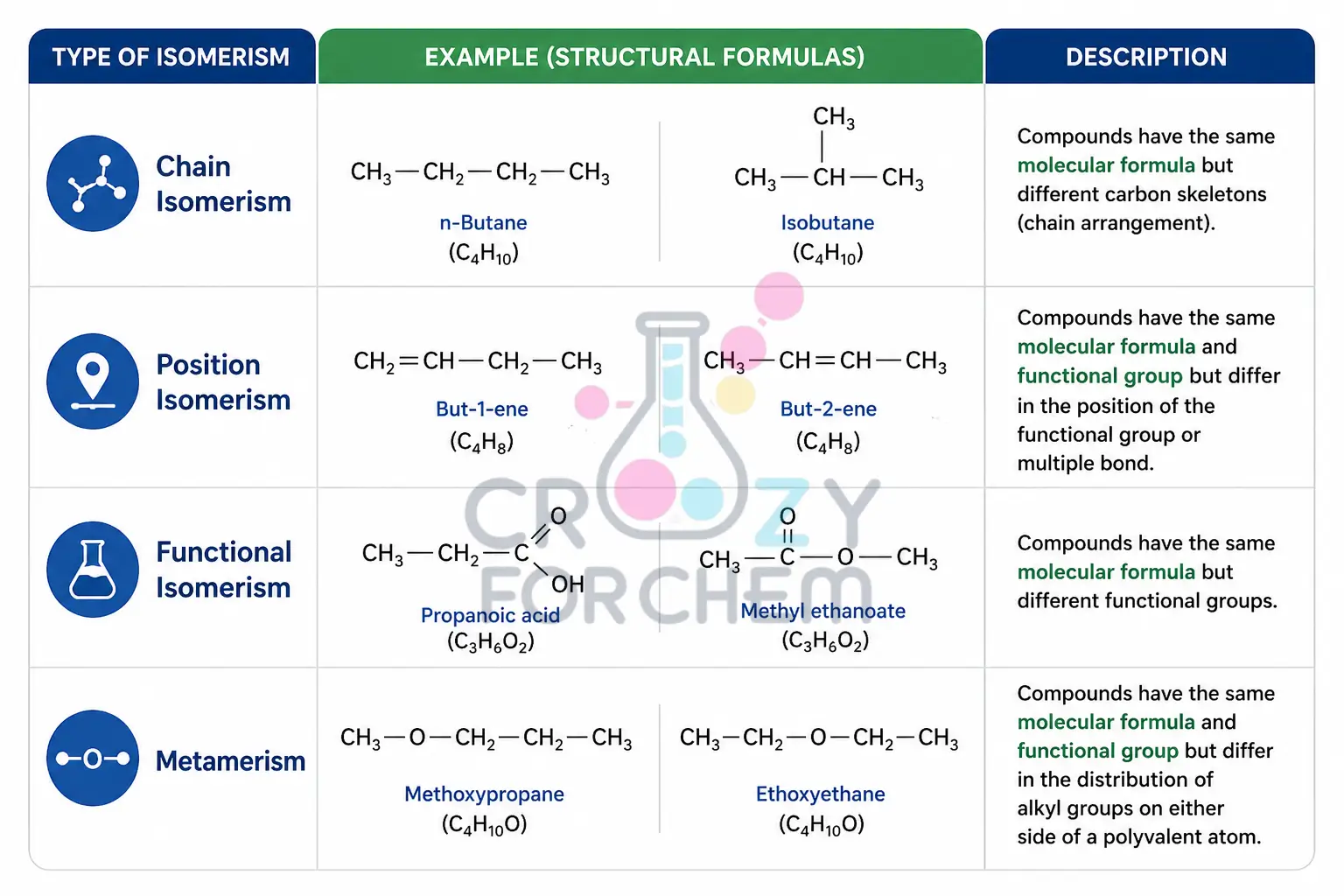
1. Chain Isomerism (Skeletal Isomerism)
Chain isomerism, also known as skeletal isomerism, arises from differences in the arrangement of the carbon skeleton. This usually involves variations between a straight “unbranched” chain and various “branched” chains.
- Butane (C₄H₁₀): This is the simplest example of chain isomerism. It can exist as n-butane (a straight chain of four carbons) or isobutane (a branched chain where one carbon is linked to the middle of a three-carbon chain).
- Pentane (C₄H₁₀): As the number of carbons increases, so does the variety of isomers. Pentane has three chain isomers: n-pentane (straight chain), isopentane (2-methylbutane), and neopentane (2,2-dimethylpropane).
2. Position Isomerism
Positional isomerism occurs when the basic carbon skeleton remains identical, but the position of a functional group or substituent atom changes. Ortho, meta, and para positions are key to understanding resonance in substituted benzenes.
- Alcohols: For the formula C₃H₈O, you can have 1-propanol (hydroxyl group on the terminal carbon) or 2-propanol (hydroxyl group on the central carbon).
- Halogens: For C₃H₇Cl, the chlorine can be at the end (1-chloropropane) or in the middle (2-chloropropane).
- Benzene Rings: Positional isomers are highly significant in aromatic chemistry. Substituents on a benzene ring can occupy three distinct positions: ortho- (adjacent carbons), meta- (separated by one carbon), or para- (opposite each other).
3. Functional Group Isomerism
Functional group isomerism arises when the same set of atoms is rearranged to form entirely different functional groups.
Because these isomers belong to different families of compounds (homologous series), their chemical properties differ drastically.
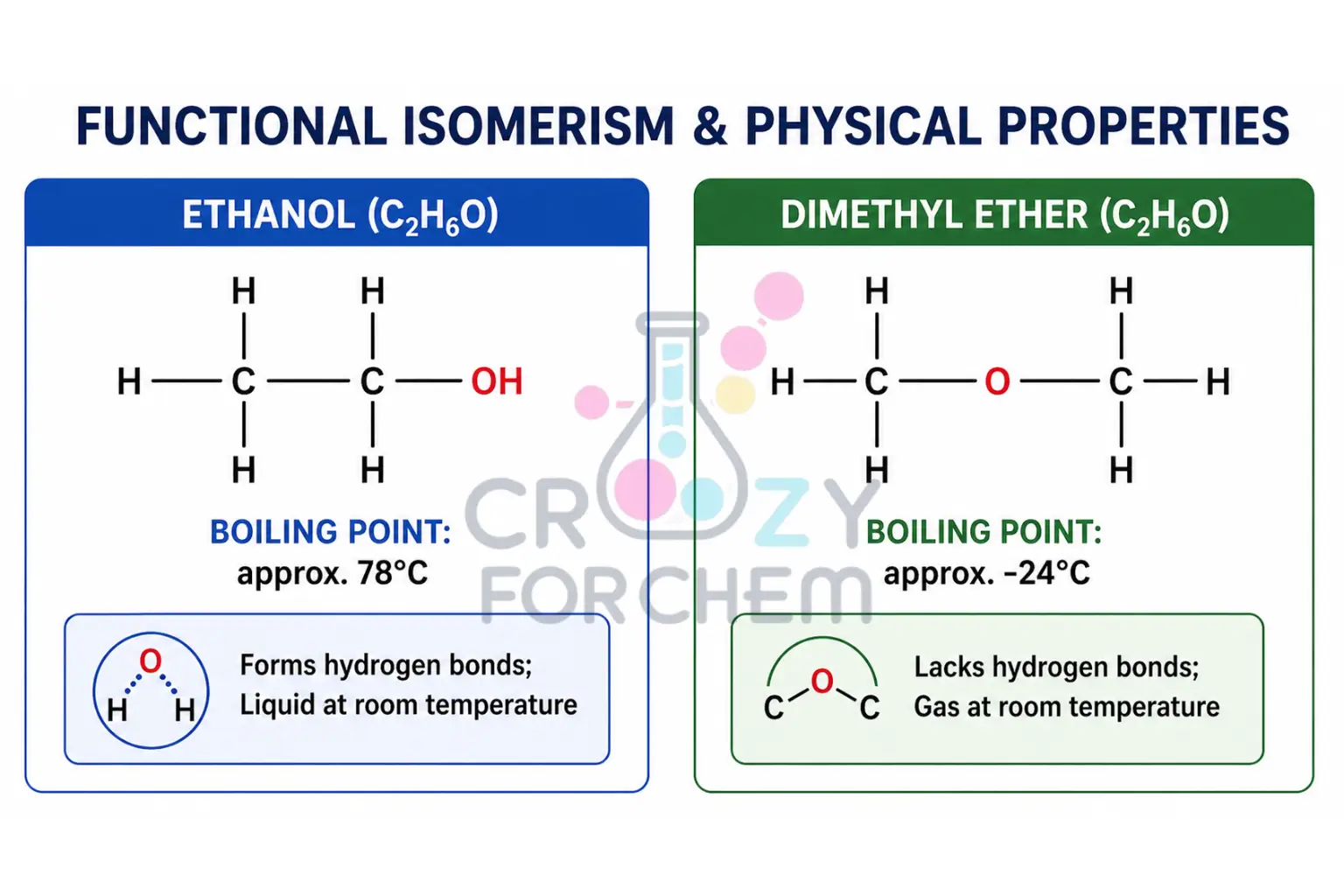
- Alcohols vs. Ethers: The molecular formula C₂H₆O represents both Ethanol (an alcohol) and Dimethyl ether (an ether). Alcohols and ethers have different properties due to their hybridisation and bonding patterns.
- Practical Application: These structural differences have real-world consequences. Ethanol, due to hydrogen bonding, has a boiling point of roughly 78°C and is a liquid. Dimethyl ether, lacking such bonds, has a boiling point of about -24°C, and exists as a gas.
- Aldehydes vs. Ketones: C₃H₆O, can be propanal (an aldehyde) or propanone (a ketone).
- Acids vs. Esters: C₃H₆O₂ can be propanoic acid or methyl ethanoate.
4. Metamerism
Metamerism is a specific type of structural isomerism caused by the unequal distribution of alkyl groups on either side of a polyvalent functional group (like an ether, ketone, or ester linkage).
- Metamerism examples: For C₄H₁₀O, you could have Diethyl ether (two carbons on each side of the oxygen) or Methyl propyl ether (one carbon on one side, three on the other).
5. Tautomerism and Ring-Chain Isomerism
- Tautomerism: A special case where isomers exist in dynamic equilibrium, often involving the migration of a hydrogen atom and a shift in a double bond (e.g., keto-enol tautomerism in acetone).
- Ring-Chain Isomerism: One isomer is an open chain while the other is a cyclic structure, such as 1-butene and cyclobutane (C₄H₈).
Difference Table: Types of Structural Isomers
| Isomer Type | Structural Difference | Primary Example |
|---|---|---|
| Chain | Arrangement of the carbon skeleton (branching). | n-Butane vs Isobutane. |
| Position | Location of the group on the same skeleton. | 1-Propanol vs 2-Propanol. |
| Functional | The type of functional group present. | Ethanol vs Dimethyl Ether. |
| Metamerism | Alkyl groups around a central functional group. | Diethyl Ether vs Methyl Propyl Ether. |
Identifying Isomers: Expert Strategies
Drawing and identifying every possible isomer can be challenging through trial and error alone. Professional chemists use specific isomerism tricks to ensure accuracy.
The IUPAC Naming Trick
The most definitive way to know if you have drawn a new structural isomer is to name it using IUPAC rules.
- If two molecules have the same formula but result in different IUPAC names, they are distinct structural isomers.
- If the names are identical, you have simply drawn the same molecule in a different orientation or after a bond rotation.
Degree of Unsaturation (Index of Hydrogen Deficiency)
The Degree of Unsaturation (DoU) or IHD is a calculation used to determine the total number of rings and/or multiple bonds in a molecule. This narrows down the possible structural arrangements.
- Formula: D.U. = (C+1) – H+X-N/2 (where C=Carbon, H=Hydrogen, X=Halogens, and N=Nitrogen).
- For example, if DoU is zero, you can rule out any structures containing rings or double bonds.
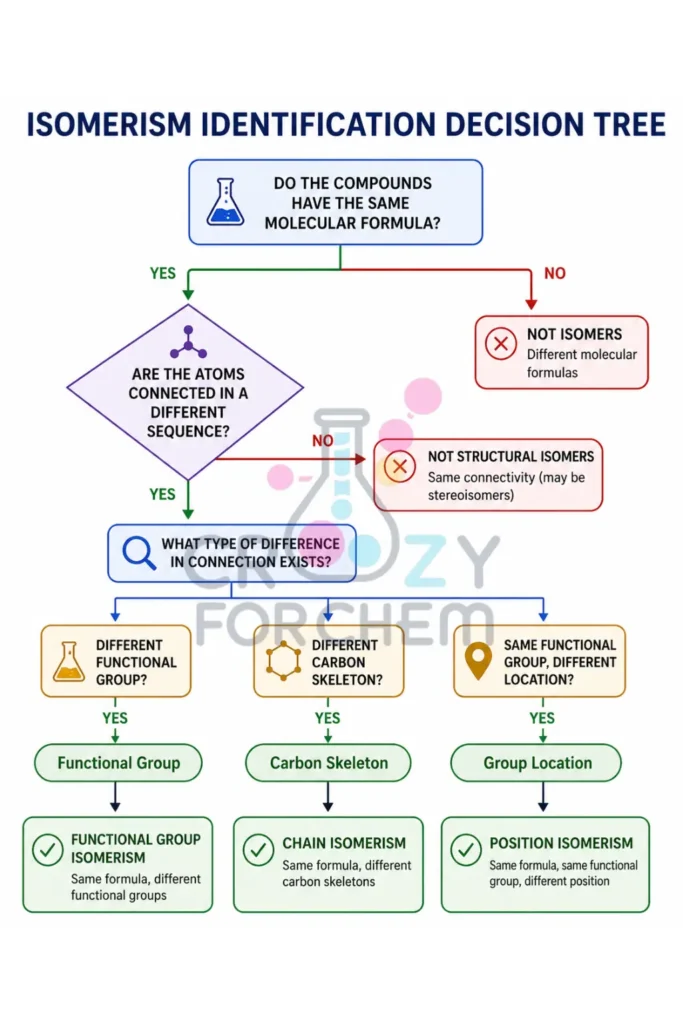
Isomerism Decision Tree
- Do the molecules have the same molecular formula? No → Not isomers. Yes → Proceed
- Are the atoms connected in a different sequence? No → Rotations or stereoisomers. Yes → Structural Isomers.
- Check Functional Groups: Different? → Functional Group Isomerism.
- Check Carbon Skeleton: Different branching? → Chain Isomerism.
- Check Group Location: Different position on the same chain? → Position Isomerism.
Educator’s Insight: “Students often ask me how to avoid drawing the same isomer twice. My secret is simple: always count your longest carbon chain first! If the longest chain stays the same and the group stays on the same carbon number, it’s not a new isomer—it’s just your drawing being turned upside down!”
Structural vs Stereoisomerism: A Brief Comparison
Isomerism is divided into two main branches: Structural and Stereoisomerism.
| Feature | Structural Isomer | Stereoisomer |
|---|---|---|
| Connectivity | Different connectivity (bonds are in a different order). | Same connectivity (bonds are in the same sequence). |
| Arrangement | Arrangement of atoms in different order. | Spatial arrangement of atoms is different. |
| Types | Chain, Position, Functional, Metamerism, etc.. | Optical and Geometric (Cis-Trans). |
Practical Applications in JEE and NEET Exams
For competitive exams, speed and accuracy in counting isomers are paramount.
- Alkanes Isomer Counting: Memorising basic counts can save time: Butane (2), Pentane (3), Hexane (5), and Heptane (9).
- Benzenoid Isomers: Identifying isomers for aromatic formulas like C₇H₈O involves considering both alcoholic and phenolic functional group isomers as well as positional variations on the ring.
Conclusion: Why Connectivity Matters
Mastering structural isomerism allows students to see past the simplicity of a chemical formula and recognise the same molecular formula in different structures that provide organic chemistry with its depth.
Whether it is predicting the vastly different boiling points of functional isomers or identifying the branching of a carbon chain, understanding connectivity is the first step in mastering the academic core of reaction mechanisms.
As an educator, I tell my students that once you can visualise how these ‘Lego bricks’ fit together, the rest of organic chemistry becomes a logical puzzle rather than a task of memorisation.
Frequently Asked Questions
-
Q1: What is the main difference between chain and position isomerism?
The main difference lies in the carbon skeleton. In chain isomerism, the arrangement of the carbon atoms themselves changes (e.g. straight vs branched chain). In position isomerism, the carbon skeleton remains identical, but the location of a functional group or substituent changes along that chain.
-
Q2: How do you identify structural isomers?
To identify structural isomers, check if the compounds have the same molecular formula but different bonding sequences. A definitive method is to use IUPAC nomenclature; if two molecules have the same formula but different IUPAC names, they are distinct structural isomers. Additionally, calculating the Degree of Unsaturation (DoU) helps narrow down the possible structures.
-
Q3: Why do structural isomers have different physical properties?
Structural isomers have different physical properties, such as boiling points and solubility, because their molecular shapes and functional groups differ. For example, ethanol (an alcohol) is a liquid at room temperature due to hydrogen bonding, while its functional isomer dimethyl ether is a gas because it lacks those intermolecular forces.
-
Q4: What is metamerism with an example?
Metamerism is a type of structural isomerism where isomers differ in the distribution of alkyl groups on either side of a polyvalent functional group (like -O-, -S-, or -CO-). A classic example is diethyl ether (CH₃CH₂-O-CH₂CH₃)) and methyl propyl ether (CH₃-O-CH₂CH₂CH₃)
-
Q5: Can two structural isomers have the same IUPAC name?
No, two distinct structural isomers cannot have the same IUPAC name. If the IUPAC names are identical, it indicates that the drawings represent the same molecule in different orientations or are simply the result of a single bond rotation, not a unique isomer.

