“As a chemist, I don’t just see a lather; I see a structural assault on viral membranes.”
Hand hygiene has always been a cornerstone of public health, but the recent global pandemic underscored just how vital these simple tools are in disrupting the chain of infection.
While both soap and alcohol-based sanitisers are designed to keep us safe, understanding the Soap vs hand sanitiser science reveals that they operate via fundamentally different chemical pathways to achieve the same goal: neutralising pathogens that survive on our skin for hours or even days.
To truly appreciate this domestic laboratory at our fingertips, we must look beyond the bubbles and the sting of alcohol to the molecular level, where surfactants and denaturants engage in a high-stakes battle against microscopic invaders.
Table of Contents
1. The Anatomy of a Virus: Understanding the Target
To understand Soap vs hand sanitiser science, we must first identify what we are attacking. Viruses like SARS-CoV-2 (the cause of COVID-19) are essentially self-assembled nanoparticles.
The Virus Lipid Envelope
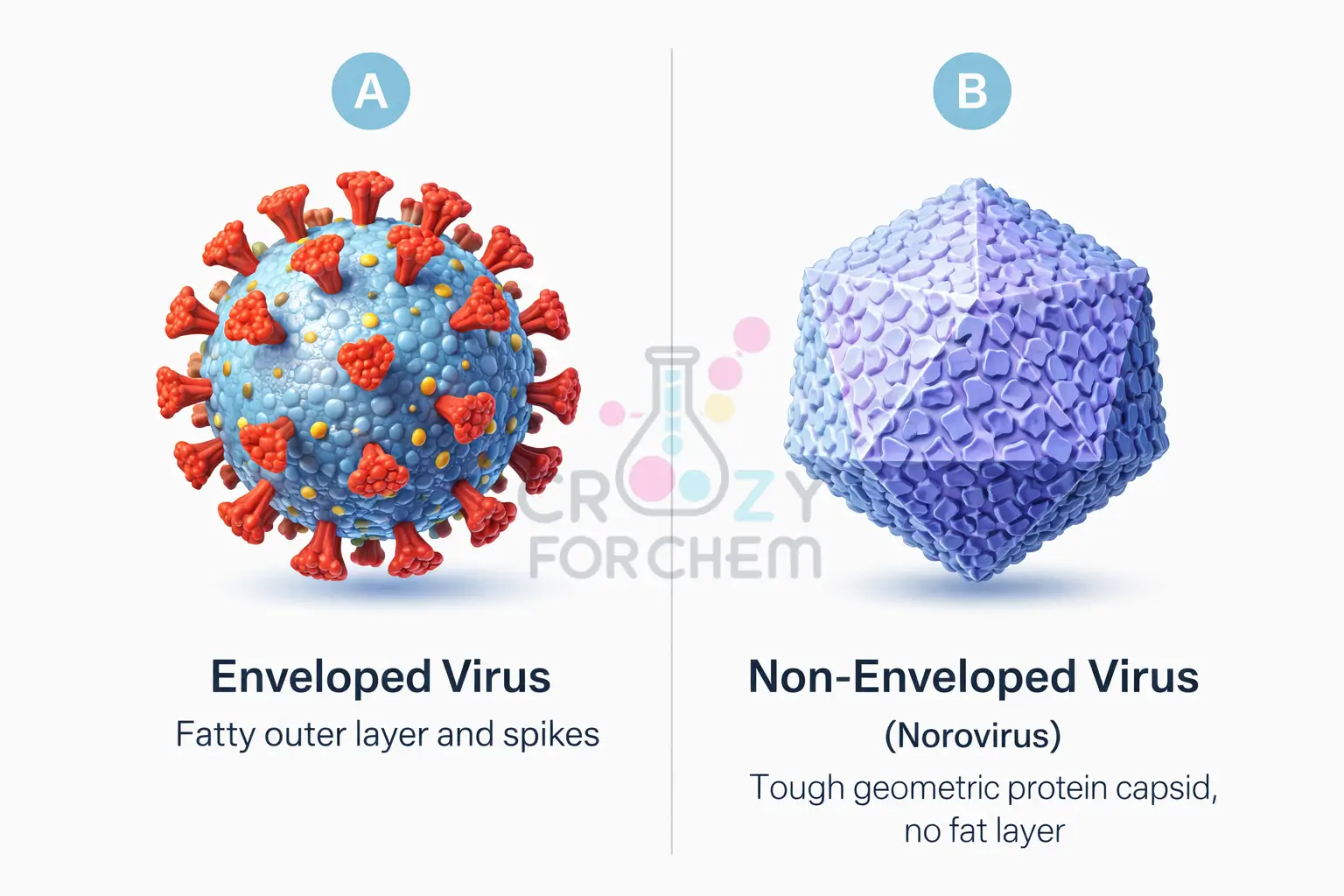
Many of the most infectious viruses are “enveloped”. This means their genetic material (RNA or DNA) is wrapped in a protective shell of fats known as a virus lipid envelope.
This envelope is not just a shield; it is studded with spike proteins that act like keys to unlock and infect human cells.
Non-Covalent Bonds and the Hydrophobic Effect
Critically, there are no strong “covalent” bonds holding these viral units together. Instead, they rely on weak “non-covalent” interactions that function like chemical Velcro.
The Hydrophobic Effect is the “invisible glue” that gives these bonds their strength. Non-polar parts of the virus’s proteins and lipids push together to avoid water, thereby maintaining the virus’s shape.
If we can disrupt this effect, the entire structure falls apart like a house of cards.
2. How Soap Works: The “Molecular Crowbar”
In the realm of Soap vs hand sanitiser science, soap is often considered the superior intervention because it performs a dual action: it removes and inactivates.
Amphiphilic Molecules
All soaps are surfactants made of amphiphilic molecules. These pin-shaped molecules have a hydrophilic (water-loving) head that binds to water and a hydrophobic (water-hating) tail that repels water and seeks out oils and fats.
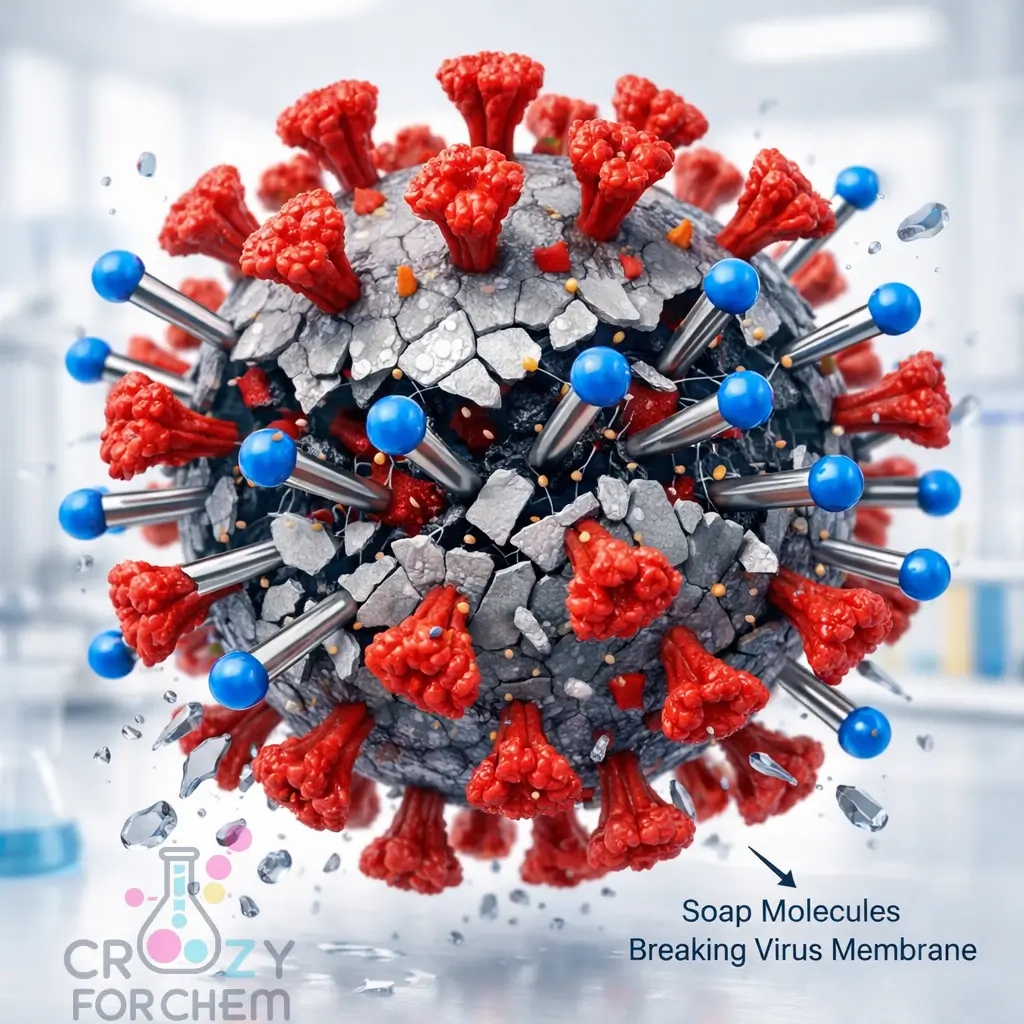
Prying the Virus Apart
When you apply soap to your hands, these molecules surround the microorganisms on your skin.
The hydrophobic tails wedge themselves into the virus lipid envelope in a desperate attempt to escape the surrounding water.
Prof. Pall Thordarson describes this as a “molecular crowbar” effect. The soap molecules pry the envelope apart, causing the virus to disintegrate and releasing its internal contents (like RNA and proteins) into the soapy water.
Micelle Formation and Emulsification
Once the virus is broken, the soap molecules assemble into “micelles”, tiny bubbles where the tails point inward, trapping viral fragments and dirt inside.
In this way, soap acts as an emulsifier. Just as emulsifiers in the food industry allow oil and water to mix, soap bridges the gap between the water from the tap and the greasy viral particles on your skin.
These “cages” of destroyed virus are then easily rinsed away down the drain.
3. How Sanitiser Works: The “Earthquake” Mechanism
While soap prys things apart, learning how hand sanitiser works involves looking at a more chaotic chemical process. Alcohol-based hand sanitisers (ABHS) typically contain ethanol, isopropanol, or n-propanol as their active components.
Protein Denaturation
Alcohols attack pathogens by denaturing proteins and disrupting the lipid membranes. If soap is a crowbar, sanitiser is like an “earthquake”.
It removes the “nails and mortar” (the hydrophobic effect) that hold the viral structure together. This causes the cell’s membrane to collapse and the vital spike proteins to crumble, rendering the virus unable to attach to your cells.
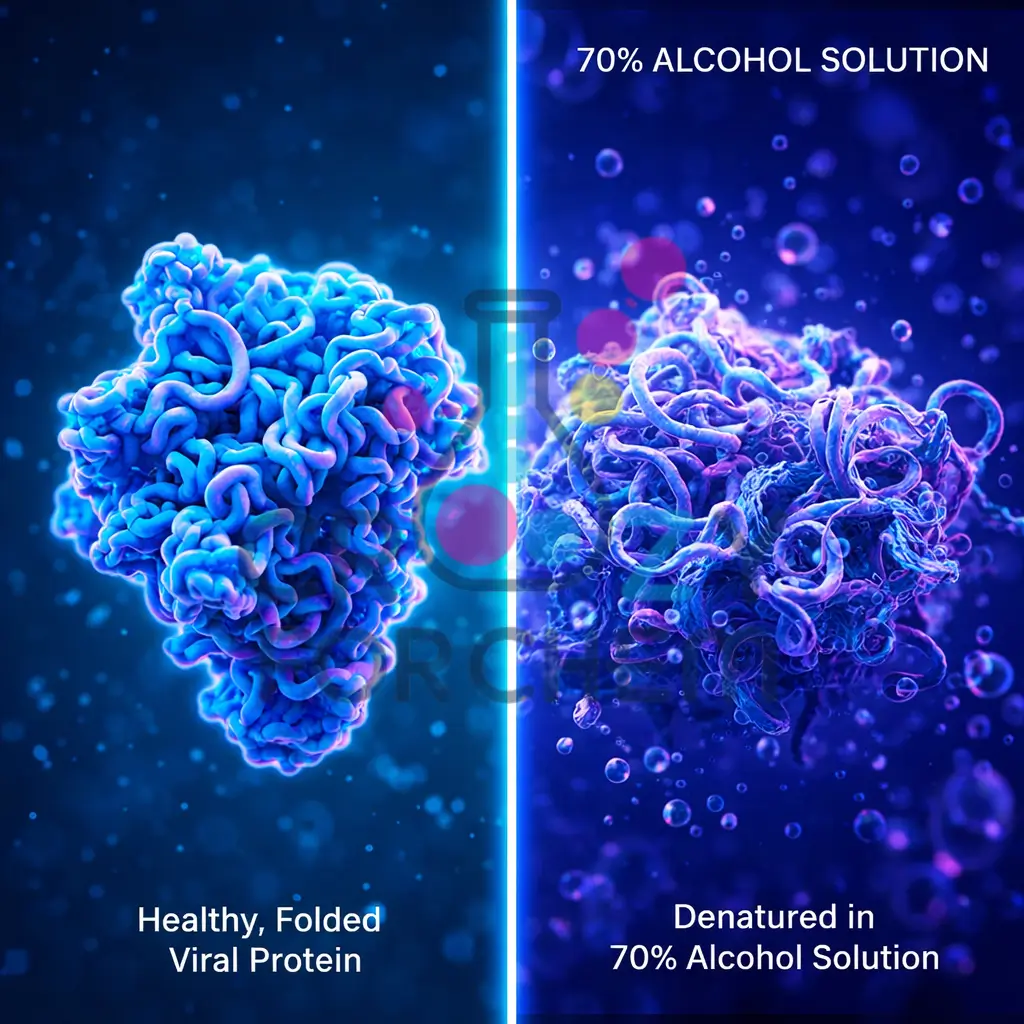
Technical Note: The 60–70% Rule
One of the most frequent questions in Soap vs hand sanitiser science is why 70% alcohol is more effective than absolute (100%) alcohol. The reasons are two-fold:
- The Necessity of Water: Water is a crucial catalyst in the protein denaturation process. Without enough water, the alcohol cannot penetrate the virus effectively, destroying its internal proteins.
- Evaporation Control: Water slows down the evaporation of alcohol. Absolute alcohol evaporates almost instantly, whereas a 70% solution stays on your hands longer, providing the necessary contact time (at least 20 seconds) required to kill the germs.
The “Sterile Dirt” Problem
A key limitation of sanitiser is that it does not remove anything from your hands. While it inactivates the virus, it leaves behind “sterile dirt”, dead viral pieces and grime. This is why sanitiser is a backup rather than a replacement for soap and water.
4. The “Skin Chemistry” Factor
A vital aspect of Soap vs hand sanitiser science is how these chemicals interact with our own biology. Frequent washing and sanitising can take a toll on the skin’s health.
Saponification and pH
The pH of human skin is typically between 4 and 6, forming a natural “acid mantle” that supports a healthy community of beneficial microbes.
Many soaps, particularly traditional bars created through saponification, have a high (alkaline) pH. This can cause the detachment of our beneficial skin flora and irritate the stratum corneum.
Humectants in Sanitiser
Because alcohol is a powerful drying agent that strips natural skin lipids, high-quality sanitisers are formulated with humectants like glycerol (glycerin) or aloe vera.
These molecules help moisten the skin and neutralise the dehydrating effects of the alcohol, though they must be used in specific concentrations to avoid making the product feel sticky.
5. Why Soap is Generally Superior
In the debate of Soap vs hand sanitiser science, health agencies like the CDC and WHO recommend soap and water whenever possible.
- The Difficulty of “Dirty” Hands: Sanitisers work best in clinical settings. In community settings where hands are greasy from food, soiled from equipment, or covered in sweat, dirt acts as a physical shield for the virus. Alcohol cannot penetrate these layers effectively.
- Chemical Removal: Soap and water are far more effective at removing harmful chemicals, such as pesticides or heavy metals, which sanitisers cannot inactivate.
- Resilient Viruses: Not all viruses have a lipid envelope. Non-enveloped viruses, such as norovirus (the winter vomiting bug) or Rhinovirus (common cold), protect their genetic core with a sturdy protein shell called a capsid. Soap and vigorous scrubbing are far more reliable at mechanically removing these tougher microbes than alcohol, which often fails to destabilise their structures.
6. The 20-Second Rule: Why Friction Matters
Chemistry takes time. Adherence to the 20-second rule, roughly the time it takes to sing “Happy Birthday” twice, is critical for two scientific reasons:
- Solvation Time: It takes time for soap molecules to penetrate the lipid layer of a virus and for the lather to solvate fats and oils.
- The Landscape of the Hand: Your hands are not smooth; they are a complex terrain of “peaks and valleys,” rifts, and folds. It takes mechanical friction and thorough coverage to ensure that the soap or sanitiser reaches every nook and cranny where a virus might be hiding.
7. Safety, Toxicology, and Contaminants
As we rely more on hand hygiene, we must be aware of the safety of the products we choose.
- Alcohol Poisoning in Kids: ABHS products are safe when used as directed, but they can cause alcohol poisoning if swallowed. Children may be attracted to products with pleasant fragrances or bright colours, so these should always be stored out of reach.
- Contaminants in Technical-Grade Ethanol: During periods of high demand, “technical-grade” ethanol was sometimes used in sanitisers. This can occasionally contain harmful contaminants like methanol (which is toxic via skin absorption), benzene (a known carcinogen), or acetaldehyde. Always check for a reputable manufacturer and an expiry date, as alcohol content can decrease over time through evaporation.
8. Conclusion: A Molecular Strategy for Safety
Understanding Soap vs hand sanitiser science proves that a bit of soap in water, an ancient and fundamentally unaltered recipe, remains one of our most valuable medical interventions.
While sanitiser is an indispensable tool for convenience “on the go,” nothing beats the combined chemical and mechanical efficiency of soap and water for destroying the virus lipid envelope and ensuring a safe environment.
Soap is more than a personal protectant; it is a communal safety net. At the molecular level, it works by breaking things apart, but at the level of society, it is the chemistry that helps hold us together.
Chemist’s Safety Alert:- “Always check the batch number. If your sanitiser has a strong ‘chemical’ or ‘tequila-like’ smell, it may contain impurities like acetaldehyde. As a chemist, I recommend products with USP-grade ethanol for maximum skin safety.”
Frequently Asked Questions
-
How does soap chemically kill a virus?
Soap molecules are amphiphilic, meaning they have a water-loving (hydrophilic) head and a fat-loving (hydrophobic) tail. These tails act like molecular crowbars, wedging themselves into the virus lipid envelope that protects many pathogens. This prys the virus apart, and the fragments are then trapped in tiny bubbles called micelles to be rinsed away safely by water.
-
Why is hand sanitiser less effective than soap on dirty hands?
Hand sanitiser works through protein denaturation, but alcohol cannot easily penetrate physical layers of dirt, grease, or organic matter. Soap is a surfactant that actively dissolves these oils and lifts grime from the skin’s surface, ensuring the chemical action reaches the virus hidden underneath.
-
Does water temperature matter for killing viruses during handwashing?
No, water temperature does not significantly affect the killing of viruses. Water hot enough to achieve thermal inactivation of a virus would cause second-degree burns to human skin. The effectiveness of handwashing comes from the chemical action of the soap and the mechanical friction of scrubbing for at least 20 seconds, regardless of whether the water is warm or cold.
-
Can I use alcoholic beverages as a hand sanitiser?
No, most alcoholic beverages are ineffective as disinfectants. To successfully denature viral proteins, a solution must contain an alcohol concentration between 60% and 95%. Most spirits (like vodka or gin) only contain approximately 40% ethanol, which is insufficient to disrupt the virus lipid envelope effectively.
-
Is “antibacterial” soap more effective against viruses?
Not necessarily. All soaps work via the same amphiphilic mechanism to break down the fatty outer layer of enveloped viruses. Specific antibacterial agents, such as triclosan, are designed to target bacterial cell walls and often have no additional impact on a virus’s structure. Furthermore, overusing these agents may contribute to antimicrobial resistance.
-
Why is 70% alcohol better than 100% alcohol for sanitising?
In the “earthquake” mechanism of sanitiser, water acts as a catalyst. Absolute (100%) alcohol evaporates too quickly and often coagulates the protein’s outer layer so fast that it creates a protective shield, preventing the alcohol from reaching the interior. A 70% solution stays in contact with the virus longer and uses water to help penetrate and unfold the viral proteins more efficiently.
-
Should I moisturise after washing my hands?
Yes, moisturising is chemically necessary to protect the skin’s barrier. Frequent washing with high-pH soaps can strip the skin’s natural lipids and disrupt the acid mantle. Applying a moisturiser or humectant while the skin is slightly damp helps lock in hydration and prevents the “cracked” skin that can actually provide a hiding place for pathogens.
