As a chemist, I see dahi-making as a controlled protein denaturation experiment.
While many view it as a routine kitchen task, the Chemistry of curd formation is a complex, irreversible chemical change where liquid milk is transformed into a semi-solid gel matrix.
This process is governed by a delicate interplay of microbiology, molecular physics, and thermodynamics, marking a permanent transition from one chemical state to another.
By examining the transformation of milk into curd, we can better understand how chemistry shapes our daily lives, specifically through the lens of fermentation and protein functionality.
Table of Contents
1. The Biological Catalyst: The Role of Lactobacillus
The science of dahi making begins with the “starter” or inoculum, a small quantity of existing curd added to warm milk. This starter acts as a delivery system for billions of Lactic Acid Bacteria (LAB), specifically species such as Lactobacillus bulgaricus and Streptococcus thermophilus.
The Role of Lactobacillus in Curd
These beneficial bacteria function as microscopic biological factories. Their primary objective is to produce energy (ATP) to multiply. They achieve this by consuming lactose, the natural disaccharide sugar found in milk.
Energy Conversion and the Fermentation Equation
During milk to curd fermentation, the bacteria utilise enzymes to break down lactose into simpler sugars, eventually producing lactic acid as a metabolic by-product. The balanced chemical equation for this conversion is:
C₁₂H₂₂O₁₁ (Lactose) + H₂O → 4 C₃H₆O₃ (Lactic Acid)
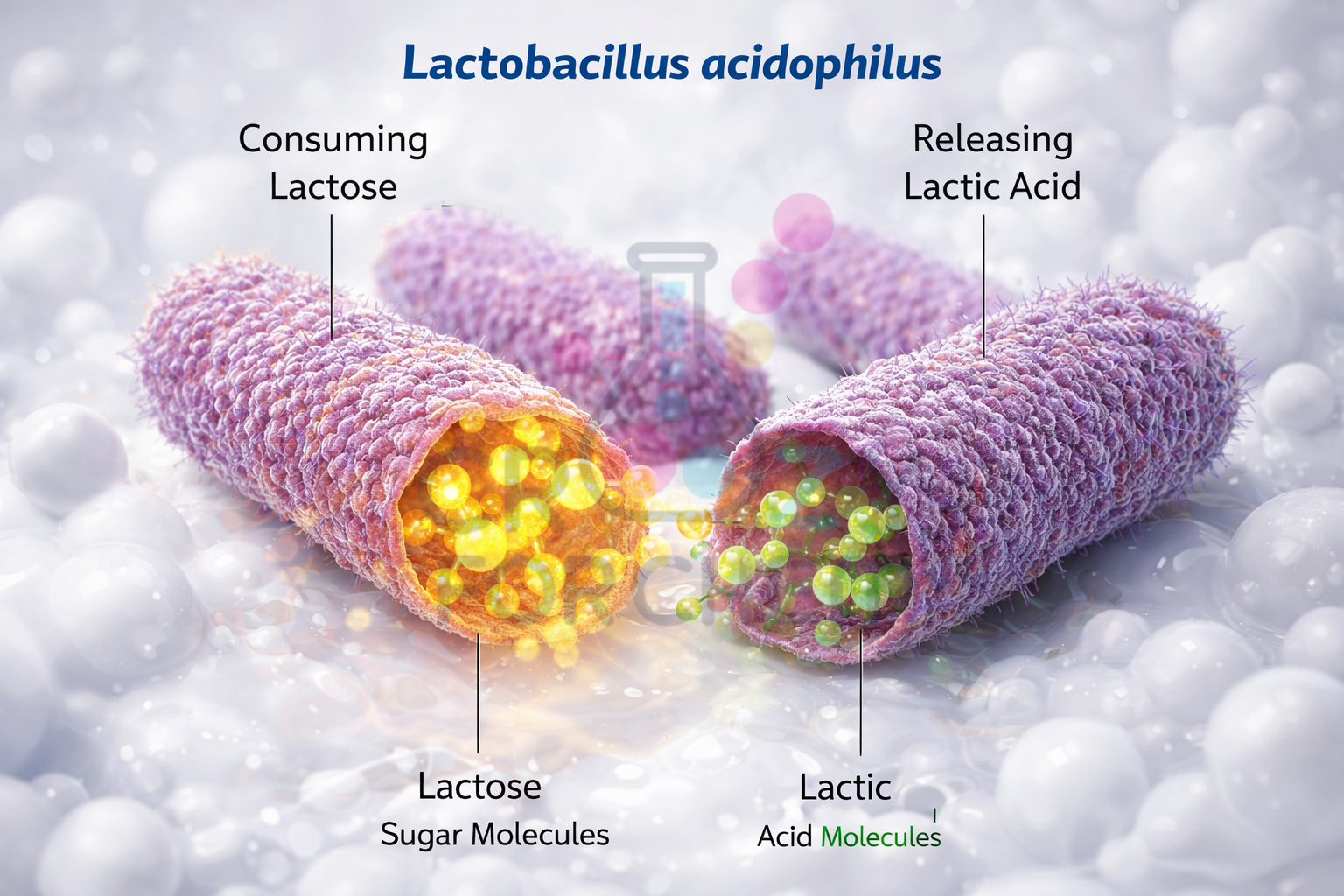
The accumulation of lactic acid is the primary driver of the Chemistry of curd formation, as it gradually increases the acidity and lowers the pH of the milk from a near-neutral 6.7–6.8 to an acidic range of 4.5–5.5.
2. The Molecular Phase Change: Casein Coagulation
To understand why milk remains liquid until this reaction occurs, we must look at its protein structure. Roughly 80% of milk protein is casein, which exists in the form of four major variants: αₛ₁, αₛ₂, β, and κ-casein
Casein Micelles and Surface Charge
Under normal conditions (pH 6.7), casein molecules are aggregated into spherical clusters called micelles. These micelles are stabilised by a negative electrical charge and steric repulsion. This negative charge causes the micelles to repel one another, keeping them suspended in a liquid state rather than clumping together.
Reaching the Isoelectric Point (pH 4.6)
As Lactobacillus produces lactic acid, the concentration of positively charged hydronium ions (H₃O⁺) increases. These ions neutralise the negative charges on the surface of the casein micelles. When the milk reaches the isoelectric point (pH 4.6), the repulsive forces vanish.
At this precise pH, the casein proteins lose their solubility and coagulate, forming a three-dimensional gel network that traps milk fats and water, resulting in the thick, white texture of curd.

3. Comparing the Medium: Cow Milk vs Buffalo Milk Curd
The Chemistry of curd formation yields different results based on the source of the milk, primarily due to variations in fat, protein, and mineral density.
Table 1: Comparative Chemistry of Cow and Buffalo Milk Curd
| Feature | Cow Milk Curd | Buffalo Milk Curd |
|---|---|---|
| Consistency | Lighter and softer | Thicker, creamier, and firmer |
| Fermentation Speed | Slower acidification | Rapid fermentation |
| Protein Content | Approx. 3.5% | Significantly higher |
| Calcium Content | ~120 mg per 100 ml | ~180 mg per 100 ml |
| Nutrient Density | High protein quality | Richer in Vitamin A and E |
Buffalo milk’s higher solid content means it acidifies more quickly, but this rapid acid production requires careful management to prevent the curd from becoming overly sour too fast.
4. Genetic Variables: The A1 vs A2 Casein Difference
In modern chemistry, we now distinguish between milk based on the genetic variants of β-casein, most notably A1 and A2 milk. The difference between these two is a single amino acid at position 67: A1 milk has histidine, while A2 milk has proline.
Coagulation Properties of A2 Milk
Scientific studies indicate that A2 milk actually produces a higher acid gel density and shows increased curd firmness compared to standard blended milk.
During rennet or acid-induced coagulation, A2 milk exhibits a higher aggregation rate.
From a nutritional perspective, some research suggests that the A1 variant can release a peptide called beta-casomorphin-7 (BCM-7) during digestion, which may cause gastrointestinal discomfort, whereas A2 milk is often better tolerated.
5. Thermodynamics: The 30°C–40°C “Sweet Spot”
The science of dahi making is highly temperature-dependent, as temperature dictates the kinetic energy and growth rate of the Lactobacillus bacteria.
- Kinetic Energy and Bacterial Growth: Lactobacillus is most active between 30°C and 40°C. In this range, the inoculum multiplies rapidly, leading to fast acid development.
- The Danger of Excessive Heat: If the milk is too hot (above 45°C), the bacteria responsible for fermentation become inactive or die. This disrupts the chemical transformation, preventing the milk from coagulating or resulting in a grainy, watery texture.
- Seasonal Chemistry: In the warmer climates of Indian summers, curd may set in just 4 to 6 hours. Conversely, during winters, the growth of the bacteria slows down, often requiring 10 to 12 hours or the use of insulated environments to maintain the “sweet spot” temperature.
6. Curd vs Yogurt vs Paneer: The Scientific Distinction
Though often used interchangeably, these three dairy products are the result of fundamentally different chemical and biological processes.
- Curd (Dahi): A traditional product of natural fermentation. It uses a diverse, often uncontrolled population of natural lactic acid bacteria from a previous batch.
- Yoghurt: An industrial product created under controlled laboratory conditions. It uses specific, standardised strains of Lactobacillus bulgaricus and Streptococcus thermophilus to ensure a uniform texture and predictable probiotic density.
- Paneer: Unlike curd, paneer is a product of direct acid precipitation. No bacterial fermentation occurs; instead, a food acid (like lemon juice or vinegar) is added to boiling milk to instantly coagulate the proteins.
7. Ginger Milk Curd: A Protease Alternative
A unique variation of curd is Ginger milk curd, a Chinese dessert that sets instantly without bacterial fermentation.
- Scientific Principle: The coagulation is triggered by a protease enzyme in ginger called Zingibain.
- The Mechanism: Unlike Lactobacillus, which produces acid, Zingibain directly acts on the milk proteins to cause clotting. It has a maximum activity at 70°C, but its effectiveness drops significantly if the milk exceeds 75°C, illustrating the precise thermal limits of enzymatic chemistry.
8. The “Chilli Stalk” Experiment: A Chemist’s Warning
A popular household hack involves adding red chilli stalks to warm milk to set curd when a starter is unavailable. The stalks contain enzymes and bacteria that can indeed trigger coagulation.
Technical Warning: “A pH of 1.15 is nearly as acidic as stomach acid (HCl). While traditional methods are fascinating, as a chemist, I strongly advise monitoring the fermentation time to keep the pH within the gut-friendly range of 4.5–5.5.”
🚨 PRO-TIP: The pH Danger Zone
Chemist-led experiments have revealed a significant risk: curd made with chilli stalks can reach an extremely high acidity level, with the pH dropping as low as 1.15. This is far more acidic than the healthy range of 4.5–5.5. Such low pH levels can be harmful to the digestive system and are not advisable for regular consumption.
9. Authority Boosters: Advanced Insights into Dahi

- Whey Syneresis (Dahi ka Pani): If a layer of water appears on top of your curd, it is known as syneresis. This occurs when the protein network shrinks, squeezing out the liquid whey. To prevent this, it is essential to boil the milk first. Boiling denatures whey proteins (specifically β-lactoglobulin), allowing them to bind to the casein and create a firmer, more stable gel that holds water better.
- Lactose Intolerance: Curd is a “pre-digested” food. Because Lactobacillus has already converted much of the lactose into lactic acid, it is significantly easier for lactose-intolerant individuals to digest than fresh milk.
- The Role of Calcium: Lactic acid reacts with the calcium phosphate that acts as the “molecular glue” (preservative) for casein micelles. As the acidity rises, this calcium phosphate dissolves and is released into the whey. This is why buffalo milk, which acidifies rapidly, can lose more calcium if whey is drained during processing.
- Vitamin Enhancement: Interestingly, the fermentation process can increase levels of B-vitamins such as riboflavin, folic acid, and B12, depending on the bacterial strains used.
10. Conclusion: The Mastery of Domestic Chemistry
Understanding the Chemistry of curd formation transforms dahi-making from a game of chance into a mastered science.
By managing the role of Lactobacillus in curd and controlling for variables like temperature and milk type, you are effectively conducting a high-level experiment in protein denaturation and bio-transformation.
Whether you prefer the creamy density of buffalo milk curd or the lighter consistency of cow milk dahi, you are participating in a chemical tradition that has preserved milk and supported human gut health for millennia.
Frequently Asked Questions
-
Is the conversion of milk to curd a physical or chemical change?
The conversion of milk to curd is an irreversible chemical change. In this process, the lactose sugar in milk is chemically transformed into lactic acid by bacteria. Furthermore, the molecular structure of the milk protein (casein) undergoes denaturation, creating a new substance with different properties that cannot be reverted to its original liquid state.
-
Why does milk turn into curd (Dahi)?
Milk turns into curd due to lactic acid fermentation triggered by Lactobacillus bacteria. These bacteria consume the natural milk sugar (lactose) and release lactic acid as a byproduct. This acid increases the acidity of the milk, causing the proteins to coagulate and form a thick gel matrix.
-
What is the role of Lactobacillus in curd formation?
The primary role of Lactobacillus is to act as a biological catalyst. It fermentatively converts lactose into lactic acid, which lowers the pH of the milk. Once the milk reaches its isoelectric point (pH 4.6), the Lactobacillus-driven acidity neutralises the negative charges on casein micelles, allowing them to clump together and set the curd.
-
Why is buffalo milk curd thicker than cow milk curd?
Buffalo milk produces a thicker curd because it contains a higher concentration of milk solids and fats (approx. 7-8% fat vs 3-4% in cow milk). This higher density of casein proteins creates a more tightly packed 3D molecular network, resulting in a firmer, creamier, and more stable curd structure.
-
What is the “sweet spot” temperature for setting curd?
The optimal temperature for setting curd is between 30°C and 40°C. This range provides the ideal kinetic energy for Lactobacillus enzymes to remain highly active. If the temperature exceeds 45°C, the bacteria may denature and die; if it falls below 20°C, their metabolic rate slows significantly, preventing the curd from setting.
-
Why does water separate from curd (Whey Syneresis)?
Water separation, or syneresis, occurs when the protein network in the curd shrinks and squeezes out the liquid whey. This is often caused by over-acidification or mechanical disturbance. Boiling milk before setting dahi can help prevent this by denaturing whey proteins, allowing them to bind more effectively with casein.
-
Can you use chilli stalks to set curd safely?
While red chilli stalks contain enzymes and bacteria that can coagulate milk, it is not always recommended for regular consumption. Chemistry-led experiments show that chilli stalks can cause the pH to drop as low as 1.15, making the curd excessively acidic and potentially irritating to the digestive system compared to the healthy range of 4.5–5.5.
