The modern periodic table is a masterpiece of organisation, but it did not appear overnight. Long before Dmitri Mendeleev left his famous gaps or John Newlands looked for musical octaves, a German chemist named Johann Wolfgang Döbereiner discovered a startling hidden rhythm in the elements.
In 1817, at a time when only 53 elements were known, Döbereiner began to notice that the building blocks of our universe weren’t just random substances; they followed a distinct numerical logic.
This discovery, known as Döbereiner’s Triads, represents the very first attempt to group chemical elements based on their physical and chemical properties through mathematical patterns.
Table of Contents
1. The Arithmetic Mean: The Logic of the Law
Döbereiner’s primary breakthrough was the identification of “triads”, groups of three elements that shared strikingly similar chemical characteristics. He proposed that these elements were physically linked by their atomic masses.
If elements A, B, and C form a triad, then:
Atomic Mass of B ≈ (Atomic Mass of A + Atomic Mass of C) / 2
This mathematical relationship, known as the arithmetic mean, suggested that the middle element in a triad would have an atomic weight approximately equal to the average of the first and third elements.
Döbereiner didn’t stop at atomic mass; he observed that this “intermediate” nature extended to other quantifiable properties, such as the density and specific weight of the substances.
Master the current Periodic Table Trends to see how these mass-based patterns evolved into modern atomic number trends.
2. The “Missing Link”: Why a Triad?
You might wonder why Döbereiner specifically chose groups of three. While many educational sites simply state the facts, the “Why” is rooted in a fascinating philosophical perspective.
At the time, Döbereiner was influenced by the concept of “trinities”, the idea that nature often works in sets of three.
He believed that these elemental triplets were not coincidental but reflected a fundamental balance in the universe.
This philosophical touch is what distinguishes his work; he wasn’t just looking at numbers; he was looking for a cosmic harmony.
When he first observed that strontium (Sr) had properties perfectly intermediate between calcium (Ca) and barium (Ba), he initially doubted if strontium even existed as an independent element, wondering if it was merely a mixture of the other two.
His rigorous experiments proved its independence, solidifying Döbereiner’s Triads as a legitimate scientific law.
3. Famous Examples: Mathematics in Action
To understand the Mendeleev periodic table logic that would eventually follow, we must examine how Döbereiner’s mathematics held up.
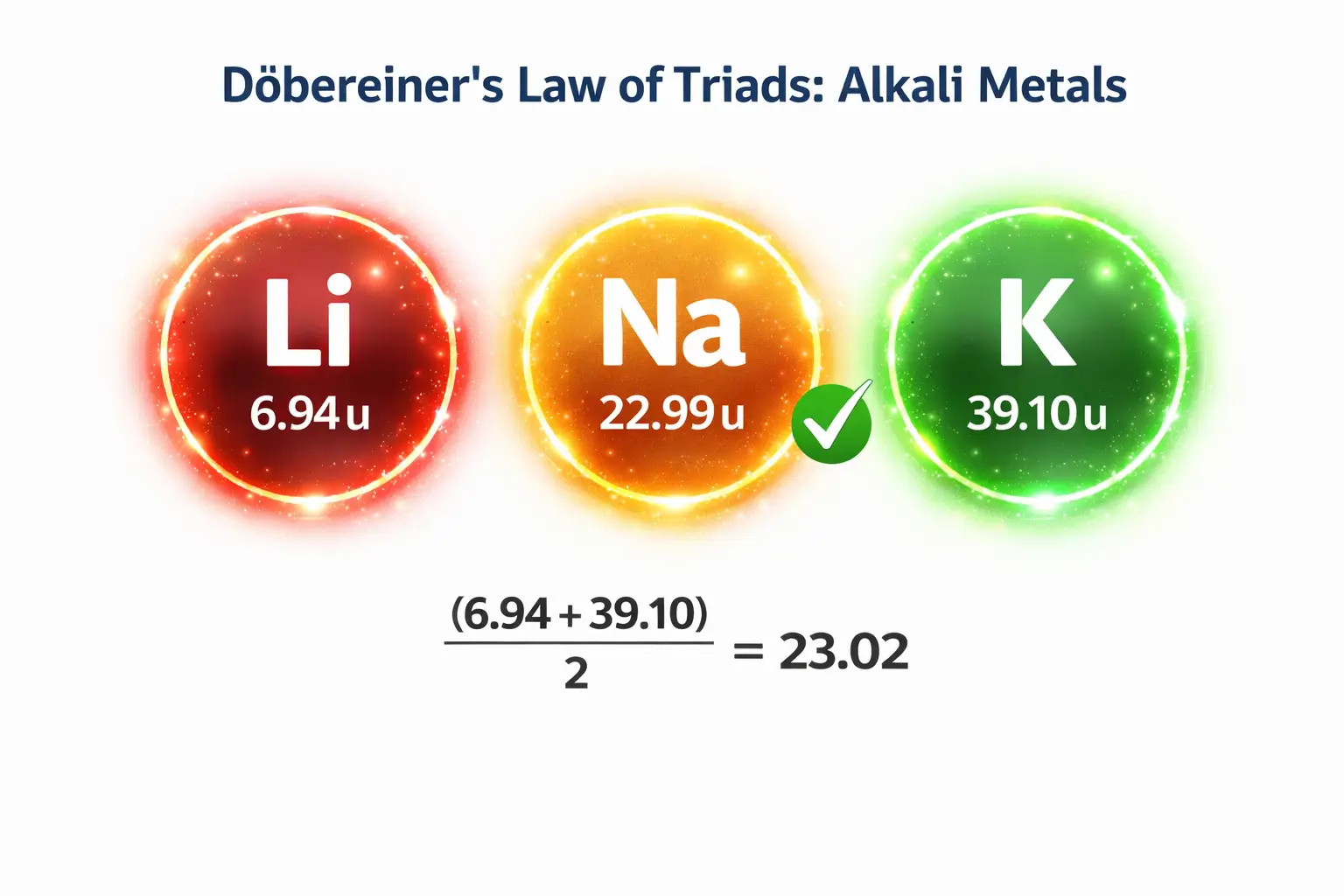
The Alkali Metal Triad (Li, Na, K)
This group remains the most textbook-perfect example of the law.
- Lithium (Li): 6.94 u
- Potassium (K): 39.1 u
- The Prediction: (6.94 + 39.1) / 2 = 23.02
- The Reality: The actual atomic mass of sodium (Na) is 22.99 u.
The elements in this triad are all highly reactive metals, possess a valency of one, and react vigorously with water to form hydrogen gas and alkalis.
The Alkaline Earth Metal Triad (Ca, Sr, Ba)
Identified in 1817, this was the first triad ever recorded.
- Calcium (Ca): 40.1 u
- Barium (Ba): 137.3 u
- The Prediction: (40.1 + 137.3) / 2 = 88.7
- The Reality: The actual mass of strontium (Sr) is 87.6 u.
These elements share similar chemical properties, and the physical properties of strontium are clearly intermediate between calcium and barium.
The Halogen Triad (Cl, Br, I)
Known as “salt-forming” elements, this group proved the law applied to non-metals too.
- Chlorine (Cl): 35.5 u
- Iodine (I): 127.0 u
- The Prediction: (35.5 + 127) / 2 = 81.25
- The Reality: The actual mass of Bromine (Br) is 80.0 u.
All three are reactive non-metals that form acids when reacted with water.
Discover the Evolution of the Periodic Table to see the 7 milestones that followed Döbereiner’s work.
4. Extended Triads and Transition Metals
By 1829, Döbereiner had identified a total of five triads. Beyond the famous ones, he noted patterns in:
- Chalcogens: Sulphur (S), Selenium (Se), and Tellurium (Te). The mean of S (32.1) and Te (127.6) is 79.85, which is remarkably close to selenium’s 79.0.
- Transition Metals: Iron (Fe), Cobalt (Co), and Nickel (Ni). While their masses are very similar (Fe: 55.8, Co: 58.9, Ni: 58.7), the mean of iron and nickel is 57.25, providing an early, if less accurate, hint at their shared properties.
5. Limitations: Why Dobereiner’s Triads Were Discarded
As chemistry moved from a philosophical pursuit to a precise science, Döbereiner’s triads encountered several insurmountable hurdles.

- Incomplete Coverage: Döbereiner could only identify a handful of triads. Most elements known at the time simply did not fit into groups of three with shared mathematical properties.
- The Halogen Exception: While chlorine, bromine, and iodine worked, adding fluorine to the mix broke the pattern. The atomic mass of chlorine is not the arithmetic mean of fluorine and bromine.
- Inaccuracy in Heavy Elements: As measurement techniques improved, the “approximate” averages began to show significant deviations, making the law feel less like a universal truth and more like a happy coincidence.
- Lack of Theoretical Basis: Döbereiner had no idea about protons, electrons, or shells. His work was purely empirical and lacked a fundamental explanation for why these patterns existed.
Read about John Newlands’ Law of Octaves to see how the next pioneer tried to fix these limitations with musical periodicity.
6. Conclusion: The Legacy of Johann Döbereiner
Although it was eventually discarded in favour of the modern periodic table, the importance of Döbereiner’s Triads cannot be overstated.
It was the first time scientists realised that relative atomic masses were crucial to understanding elemental identity. He moved the scientific world away from simple lists and towards a structured, logical order.
Every time we look at a vertical group in the periodic table today, we are seeing the modern descendant of Döbereiner’s trinities.
He laid the foundation for Mendeleev’s predictions and proved that chemistry is, at its heart, a mathematical language.
💡 CrazyForChem Fact: Döbereiner was a close friend of the famous poet Goethe. It is said that their conversations about “affinities” in chemistry influenced some of Goethe’s literary works!
Döbereiner’s Triads: NCERT Class 10 Exam Guide
For students preparing for their Class 10 Board Examinations, Döbereiner’s Triads fall under Chapter 5: Periodic Classification of Elements of the NCERT Science syllabus. The following points cover every aspect of this topic as it appears in Board examinations.
Most Frequently Asked Exam Questions:
1-Mark Questions (Definition Type) Q: State Döbereiner’s Law of Triads. A: Döbereiner’s Law of Triads states that when elements are arranged in order of increasing atomic mass in groups of three, the atomic mass of the middle element is approximately equal to the arithmetic mean of the atomic masses of the first and third elements.
2-Mark Questions (Calculation Type) Q: Using Döbereiner’s Law, calculate the atomic mass of bromine given that chlorine = 35.5 u and iodine = 127 u. A: Atomic mass of Br = (35.5 + 127) / 2 = 81.25 u (Actual: 80 u ✅)
3-Mark Questions (Limitation Type) Q: What are the limitations of Döbereiner’s Triads? A: Three key limitations are: (i) only five triads could be identified; most elements did not fit; (ii) newly discovered elements, such as fluorine, broke the arithmetic mean rule in the halogen group; (iii) the law had no theoretical basis, as atomic structure was not yet understood.
💡 Board Exam Tip: The Alkali Metal Triad (Li, Na, K) is the most commonly tested calculation in Board exams. Always remember: (6.94 + 39.1) / 2 = 23.02 ≈ 22.99 (Na). Memorise this calculation it appears almost every year.
Frequently Asked Questions
-
What is Döbereiner’s Law of Triads?
Ans: Johann Wolfgang Döbereiner proposed that certain elements with similar chemical properties could be grouped in sets of three, called triads. His law states that when these three elements are arranged by increasing atomic mass, the atomic mass of the middle element is approximately the arithmetic mean (average) of the first and third elements.
-
Give an example of a Döbereiner triad with its calculation.
Ans: The most common example is the Alkali Metal Triad:
Lithium (Li): Atomic mass 6.94
Potassium (K): Atomic mass 39.10
Calculation: (6.94 + 39.10) / 2 = 23.02. The result (23.02) is nearly identical to the actual atomic mass of Sodium (Na), which is 22.99. -
Why was the Law of Triads rejected?
Ans: Although it was the first mathematical pattern, it had several limitations:
Limited Application: Döbereiner could only identify five triads; most elements did not fit this pattern.
Inaccuracy: As measurements became more precise, the averages often showed large deviations.
Discovery of New Elements: Newly discovered elements (like Fluorine in the halogen group) broke the arithmetic mean rule. -
Who was the first scientist to classify elements based on atomic mass?
Ans: Johann Wolfgang Döbereiner was the first to provide a logical, mass-based classification in 1817. His work proved that there is a direct relationship between an element’s atomic weight and its chemical properties, paving the way for Newlands and Mendeleev.
-
Did Döbereiner’s Triads apply to non-metals?
Ans: Yes. Döbereiner identified triads for non-metals as well, such as the Halogen Triad (Chlorine, Bromine, and Iodine) and the Chalcogen Triad (Sulphur, Selenium, and Tellurium). Both groups followed the arithmetic mean logic for their atomic masses.
-
How many triads did Döbereiner identify in total?
By 1829, Döbereiner had successfully identified five triads in total. These were the Alkaline Earth Metal Triad (Ca, Sr, Ba), identified first in 1817; the Alkali Metal Triad (Li, Na, K); the Halogen Triad (Cl, Br, I); the Chalcogen Triad (S, Se, Te); and the Transition Metal Triad (Fe, Co, Ni). Beyond these five groups, the law could not be extended to the remaining known elements, which was one of its principal limitations.
-
What is the difference between Döbereiner’s Triads and Newlands’ Law of Octaves?
Döbereiner’s Triads (1817–1829) grouped elements in sets of three based on atomic mass and mathematical averages, but could only account for a handful of known elements. Newlands’ Law of Octaves (1865) was a broader attempt it arranged all known elements in order of increasing atomic mass and observed that every eighth element had properties similar to the first, analogous to musical octaves. While Döbereiner worked with small clusters, Newlands attempted a universal sequential arrangement. Both were eventually superseded by Mendeleev’s Periodic Table, which introduced the concept of leaving gaps for undiscovered elements.
