Understanding how atoms assemble into three-dimensional structures is a vital milestone in chemistry.
VSEPR Theory (Valence-Shell Electron-Pair Repulsion Theory) provides the essential molecular geometry guide to predicting these shapes based on the behaviour of valence electrons.
In this comprehensive guide, we will explore the fundamental postulates of VSEPR, introduced by Sidgwick and Powell and refined by Gillespie and Nyholm.
We will cover the AXE method for systematic electron counting, explain why molecules like water and ammonia have distorted angles, and provide a master table of basic molecular shapes.
By the end of this article, you will be able to visualise the 3D architecture of matter and understand how electron repulsions dictate the physical world.
Table of Contents
The “Why” Foundation: The Two Pillars of Trends
Before applying VSEPR, it is critical to remember the forces that govern atomic interactions.
To truly master this molecular geometry guide, it is essential to first understand how Atomic Radius Trends influence the size of the central atom.
These forces dictate how electron pairs will repel one another once a bond is formed. VSEPR takes these atomic fundamentals and applies them to the complex 3D arrangement of atoms within a molecule.
1. The Core Postulates of VSEPR Theory
To use this molecular geometry guide effectively, one must master the fundamental rules that govern molecular assembly:
- Repulsion is Paramount: Electron pairs, being negatively charged, repel each other and orient themselves to maximise the distance between them.
- The Repulsion Hierarchy: Not all repulsions are equal. Lone pairs (unshared electrons) are concentrated closer to the central atom because they are attracted by only one nucleus rather than two; consequently, they occupy more space and exert stronger repulsion than bonding pairs. The order of repulsive strength is: Lone Pair–Lone Pair (LP-LP) > Lone Pair–Bond Pair (LP-BP) > Bond Pair–Bond Pair (BP-BP).
- Multiple Bonds as Single Units: For predicting shape, a double or triple bond is treated as a single “electron group” or bonding region. However, multiple bonds have higher electron density and occupy more space than single bonds, which can cause minor deviations from ideal bond angles.
- The Central Atom: In polyatomic molecules, the least electronegative atom is typically identified as the central atom to which all others are linked.
2. The AXE Method: A Systematic Approach
A highly effective tool within this molecular geometry guide is the AXE method of electron counting.
- A: Represents the central atom.
- X: Represents the number of atoms bonded to the central atom.
- E: Represents the number of lone pairs on the central atom.
The sum of X and E is known as the Steric Number or Coordination Number, which determines the basic electron group arrangement.
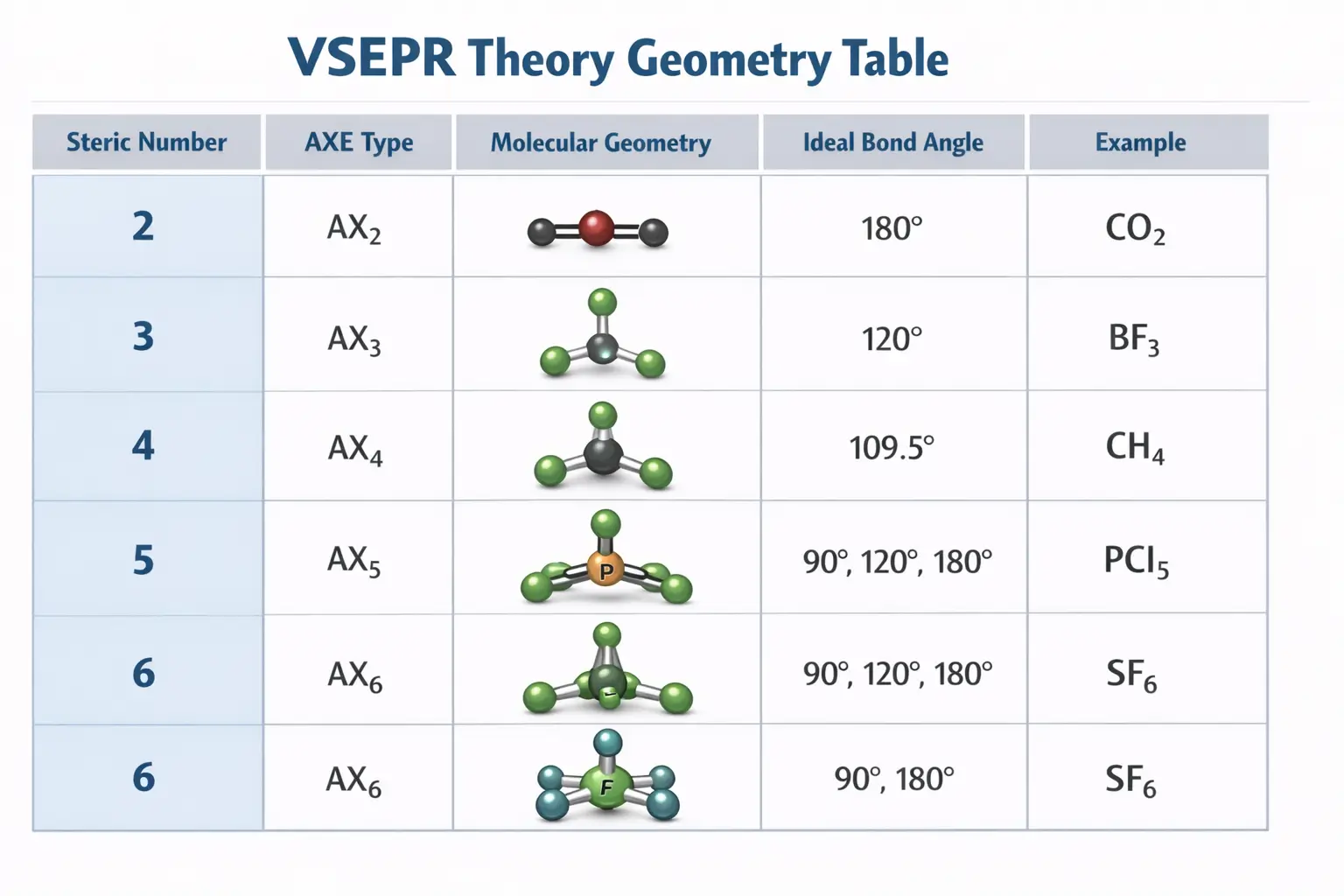
3. Basic VSEPR Shapes and the Master Table
Molecules adopt five basic “parent” shapes based on the number of electron groups around the central atom. For species with no lone pairs, the molecular geometry is identical to the electron group geometry.
4. Why is Water Bent? (Distortion Explained)
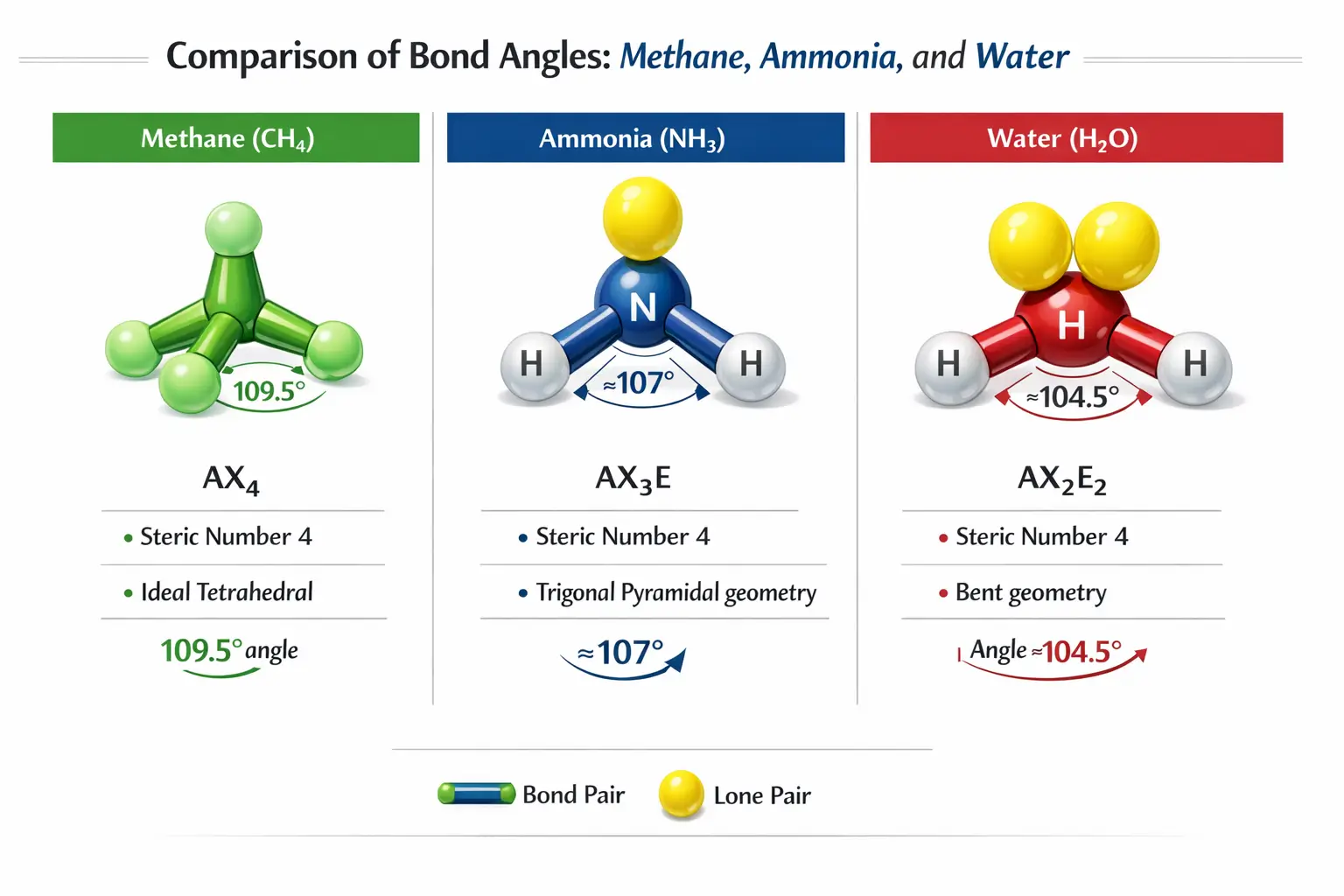
A common “Why” question in chemistry is why molecules with the same steric number have different shapes. For instance, Methane (CH₄), ammonia (NH₃), and Water (H₂O) all have a steric number of 4, yet their geometries differ.
In Water (H₂O), the central oxygen atom has two bonding pairs and two lone pairs (AX₂E₂). While the electron geometry is tetrahedral, the actual molecular shape only considers the positions of the nuclei.
Because lone pairs exert a stronger repulsive force (LP-LP > LP-BP), they “squeeze” the O–H bonding pairs closer together. This reduces the ideal tetrahedral angle of 109.5° to approximately 104.5°, resulting in a Bent or V-shaped structure.
Similarly, Ammonia (NH₃) has one lone pair and three bonding pairs (AX₃E). The LP-BP repulsion is stronger than BP-BP repulsion, pushing the hydrogen atoms down and reducing the bond angle to approximately 107°, creating a Trigonal Pyramidal shape.
While electron geometry might be tetrahedral, the actual molecular shape only considers the positions of the atoms, not the lone pairs. Let’s look at how the number of lone pairs affects the structure and bond angles of three common molecules:
5. Five and Six Electron Groups: Complex Repulsions
When a central atom has five or six electron groups, the positions around the atom are not always equivalent.
Trigonal Bipyramidal (AX₅ Derivatives)
In a trigonal bipyramid, there are two distinct positions: axial (vertical) and equatorial (horizontal). Axial bonds are longer and weaker because they experience more repulsion from equatorial bond pairs.
- Seesaw Shape (AX₄E): In molecules like SF₄, lone pairs preferentially occupy equatorial positions to minimise 90° LP-BP repulsions.
- T-Shaped (AX₃E₂ in BrF₃ or ClF₃): Two lone pairs occupy equatorial positions, leading to a T-shape with bond angles slightly less than 90°.
- Linear (AX₂E₃): In the I₃⁻ ion or XeF₂, all three lone pairs occupy equatorial positions, leaving the bonded atoms in a straight line at 180°.
Octahedral (AX6 Derivatives)
- Square Pyramidal (AX₅E ): In BrF₅): One lone pair pushes the four equatorial fluorine atoms away, reducing bond angles to 85.1°.
- Square Planar (AX₄E₂ in XeF₄): two lone pairs occupy opposite axial positions, which minimises LP-LP repulsion and results in a flat, square arrangement with 90° angles.
6. Molecules with No Single Central Atom
VSEPR can predict the structure of complex molecules by treating them as linked fragments.
- Methyl Isocyanate (CH₃–N=C=O): The carbon in CH₃ is tetrahedral (AX₄); the nitrogen is bent (AX₂E); and the isocyanate carbon is linear (AX₂).
- Propyne (H₃C–C≡CH) ): The first carbon is tetrahedral, while the two carbons sharing the triple bond are linear.
See how these structures fit into the historical Evolution of the Periodic Table.
7. Geometry and Molecular Polarity
Molecular geometry determines whether a molecule has a net dipole moment.
- Symmetrical Molecules: If individual bond dipoles cancel out due to symmetry, the molecule is non-polar (e.g., CO₂, BF₃, CH₄, PCl₅, SF₆).
- Asymmetrical Molecules: If lone pairs are present or bonded atoms are different, dipoles do not cancel, making the molecule polar (e.g., H₂O, NH₃, CHCl₃).
8. Theoretical Context: VSEPR vs. Other Models
While VSEPR accurately predicts 3D structures, it is a simplified model.
- Valence Bond Theory: Explains geometry through the hybridisation of atomic orbitals (e.g., sp, sp², sp³, sp³d, sp³d²), providing the quantum mechanical justification that VSEPR lacks.
- Molecular Orbital Theory: Describes bonding in terms of the whole molecule rather than individual atoms.
Learn why Ionisation Energy Exceptions sometimes occur despite what these geometric models predict.
9. Limitations of VSEPR Theory
Despite its power, this molecular geometry guide has significant limitations:
- It does not explain the nature or energy of chemical bonds.
- It is less accurate for transition metal compounds, where high atomic masses and inactive lone pairs influence structure.
- It fails to explain isoelectronic species, which may have different shapes despite having the same number of electrons.
- It erroneously predicts that Group 2 halides (like CaF₂) are linear when many are actually bent.
Conclusion
Mastering VSEPR theory is a fundamental milestone for any chemist. By combining the AXE method with an understanding of electron repulsion, you can visualise the invisible 3D architecture of matter.
This structural knowledge is the key to understanding how molecules react, bond, and function in the real world.
💡 CrazyForChem Fact: For a fun, hands-on way to learn these shapes, researchers have developed origami molecular models. Building a 3D paper model of Ammonia (NH₃) can make the “invisible” lone pair repulsion much easier to visualise than a flat drawing.
Frequently Asked Questions
-
What is the main purpose of VSEPR Theory?
Ans: The primary purpose of VSEPR Theory (Valence Shell Electron Pair Repulsion) is to predict the three-dimensional shape and molecular geometry of a molecule. It is based on the idea that electron pairs around a central atom repel each other and stay as far apart as possible to achieve maximum stability.
-
Why does a lone pair cause more repulsion than a bond pair?
Ans: A lone pair is attracted by only one nucleus (the central atom), which allows its electron cloud to spread out more widely. In contrast, a bond pair is pulled by two nuclei, keeping it more “contained.” Because lone pairs occupy more space, they exert a stronger repulsive force, following the hierarchy: LP-LP > LP-BP > BP-BP.
-
What is the difference between Electron Geometry and Molecular Geometry?
Ans: Electron Geometry: Considers the arrangement of all electron groups (both lone pairs and bonding pairs).
Molecular Geometry: Only describes the actual positions of the atoms (nuclei) in space. For example, in Water (H₂O), the electron geometry is Tetrahedral, but because of the two lone pairs, the molecular geometry is Bent. -
How do you use the AXE method in this molecular geometry guide?
Ans: The AXE method is a formula used to identify molecular structure:
A = Central Atom
X = Number of atoms bonded to the central atom
E = Number of lone pairs on the central atom. By finding the sum of X + E (the Steric Number), you can quickly determine the molecule’s parent shape from the VSEPR Master Table. -
Why is the bond angle in Ammonia (NH₃) 107° instead of 109.5°? °?
Ans: Although Ammonia has a tetrahedral electron arrangement, it contains one lone pair. According to VSEPR Theory, the repulsion between the lone pair and the bond pairs (LP-BP) is stronger than the repulsion between the bond pairs themselves. This extra “push” from the lone pair squeezes the H-N-H bonds closer together, reducing the angle from the ideal 109.5° to approximately 107°.

