In the intricate landscape of organic chemistry, the distribution of electron density is rarely static or restricted to a single location.
Instead, many molecules exhibit a phenomenon where electrons are spread across several atoms, a process known as the Resonance Effect, the Mesomeric Effect, and Electron Delocalisation.
This fundamental concept is one of several electronic factors alongside the inductive effect, hyperconjugation, and the temporary electromeric effect that dictate the chemical reactivity and physical characteristics of organic compounds.
By understanding how electrons move through a system, chemists can predict the stability of molecules, the outcome of reactions, and the specific behaviour of various functional groups.
Table of Contents
Defining the Phenomenon: Resonance vs Mesomerism
The terms resonance and mesomerism are often used interchangeably in modern literature, yet they possess distinct historical and technical roots.
Historical Context: Linus Pauling vs Christopher Ingold
The conceptualisation of delocalised bonding emerged through two different schools of thought in the early 20th century.
Linus Pauling, an American chemist, established the resonance effect in 1928, drawing on a mathematical analogy between the behaviour of wave functions in quantum mechanics and mechanical resonance.
Simultaneously, in Britain, Christopher Kelk Ingold developed the “mesomeric effect” or “mesomerism” in the 1930s as a way to account for electron displacement in organic reactions.
Ingold preferred his terminology because he believed Pauling’s “resonance” suggested a physical oscillation between structures that did not actually occur.
Are they the same? IUPAC and Technical Nuances
According to the IUPAC Gold Book, the terms are essentially synonymous.
However, some technical distinctions are still taught: resonance is frequently viewed as a broader “umbrella” term for electron delocalisation, whereas the mesomeric effect specifically refers to the electron-donating or withdrawing nature of a substituent group within a conjugated system.
While resonance describes the interaction between lone pairs and bond pairs to determine structure, the mesomeric effect is particularly associated with the stabilisation provided by functional groups.
Defining Delocalisation
The core of both effects is electron delocalisation. This refers to a state where electrons, typically π electrons or those in unhybridised p-orbitals, are not limited to one atom or one specific covalent bond.
These electrons are “free” to move across several atoms, which lowers the overall potential energy of the molecule and increases its stability.
What is Resonance in Organic Chemistry?
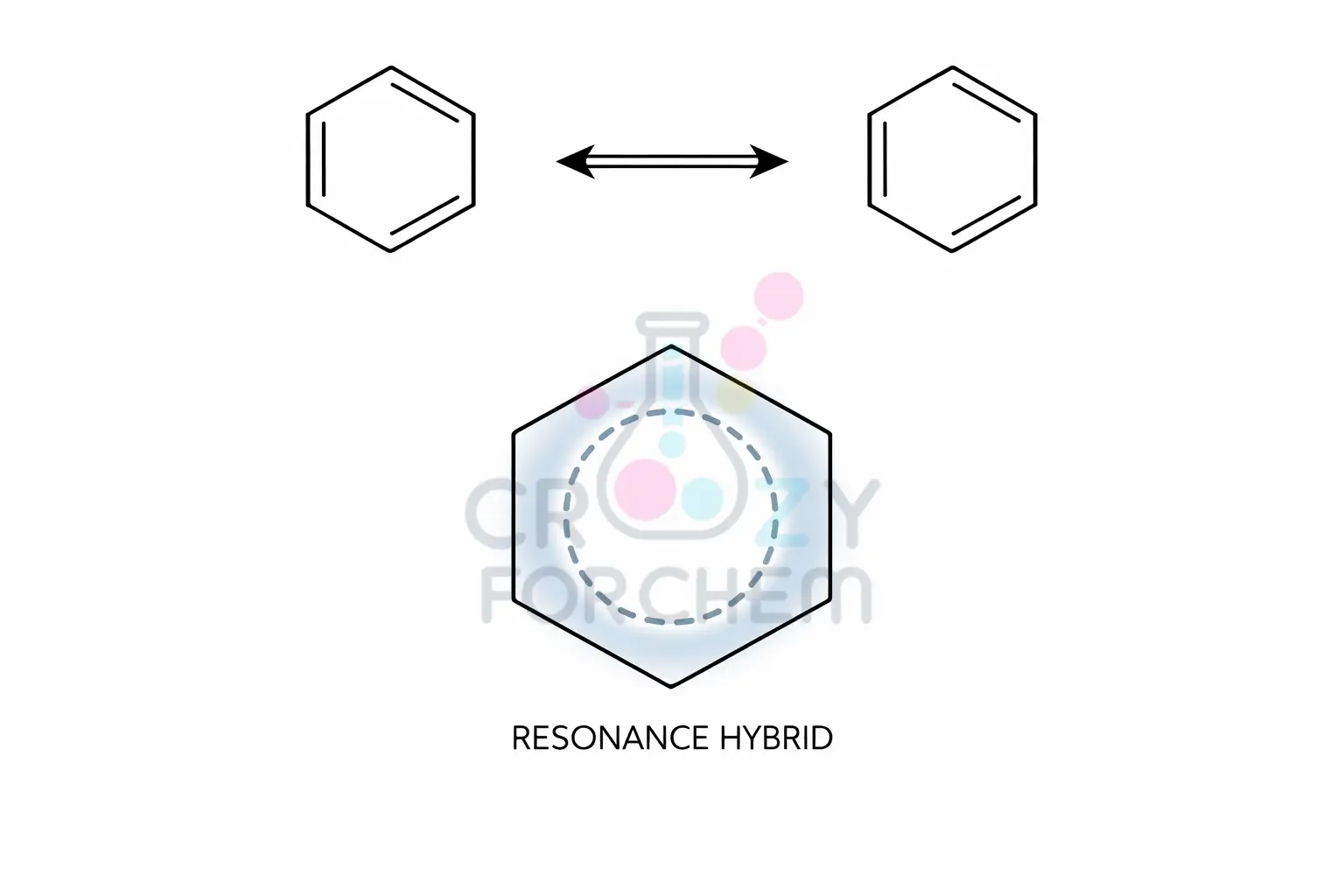
Resonance is the representation of a molecule with delocalised electrons as a weighted average of two or more hypothetical Lewis structures.
The Concept of Resonance Hybrids
A molecule that cannot be represented by a single Lewis structure is assigned multiple canonical structures (or contributing structures). The actual, real-world molecule is a resonance hybrid of all these valid structures.
For example, benzene is not a mixture of two alternating cyclohexatriene isomers; it is a single hybrid where the pi electrons are distributed evenly around the hexagonal ring.
This hybrid is always more stable and has lower energy than any of its individual canonical forms.
Necessary Conditions for Resonance
For Resonance and Mesomeric Effect, Electron delocalisation to occur, specific structural requirements must be met:
- Coplanarity: All atoms involved in the delocalisation must lie in the same plane to allow for the parallel overlap of p-orbitals.
- Conjugation: There must be a system of connected p-orbitals. Delocalisation occurs when:
- A π bond is conjugated with another π bond.
- A π bond is adjacent to a lone pair or a negative charge.
- A π bond is conjugated with a positive charge (vacancy) or a free radical.
The Mechanics of Electron Delocalisation
Conjugated Systems
A conjugated system consists of alternating single and multiple bonds or a sequence of p-orbitals that allow electrons to be delocalised across the entire framework. This overlap of orbitals lowers the energy of the molecule.
The Role of Pi (π) Electrons
The resonance effect primarily involves the movement of π electrons or unshared lone pairs.
Unlike the inductive effect, which operates through sigma (σ) bonds, resonance strictly involves the π framework; sigma bonds are never broken or rearranged in resonance structures.
Resonance Hybrids vs Canonical Structures
It is a common misconception that a molecule shifts rapidly between its canonical structures. In reality, every molecule has the same hybrid structure.
The canonical forms are merely an “esoteric way of human expression” used to depict a sub-atomic reality that we cannot easily see or represent with a single static diagram.
Types of Resonance and Mesomeric Effects
Substituents are classified by their ability to either donate or withdraw electron density within a conjugated system.
Positive Mesomeric Effect (+M or +R)
In a +M effect, electrons are transferred or donated away from a substituent group and into the conjugated system.
This increases the electron density at certain positions in the molecule. +M and -M effect examples of electron-donating groups include:
- –OH (Hydroxyl) and –OR (Alkoxy).
- –NH₂ (Amino) and substituted amines like –NHR or –NR₂.
- –X (Halogens).
- –O⁻ and –S⁻.
Negative Mesomeric Effect (-M or -R)
In a -M effect, a substituent withdraws electron density towards itself and away from the rest of the molecule.
This decreases the electron density on the rest of the molecular entity. +M and -M effect examples of electron-withdrawing groups include:
- –NO₂ (Nitro).
- –CHO and –COR (Carbonyl groups).
- –CN (Cyano).
- –COOH and –COOR.
Essential Rules for Drawing Valid Resonance Structures
To maintain accuracy in rules for resonance structures, several principles must be followed:
- Atomic Positions: The arrangement of atoms and nuclei must remain identical across all structures. Only electrons move.
- The Octet Rule and Charge Minimisation: Valid canonical forms must be proper Lewis structures; for example, carbon cannot have five bonds. Structures that obey the octet rule and have the least number of formal charges are more significant.
- Planarity: The atoms involved must be coplanar to ensure the parallel p-orbitals can overlap effectively.
- Electron Count: All resonance contributors must have the same number of paired and unpaired electrons.
Real-World Applications and Chemical Significance
Stability of Aromatic Systems
Resonance is the primary reason for the exceptional stability of aromatic compounds like benzene.
The delocalisation of pi electrons around the ring provides “resonance stabilisation,” which prevents benzene from undergoing standard addition reactions typical of alkenes.
Resonance Energy: The Measure of Stability. The difference in energy between the actual resonance hybrid and the most stable canonical structure is called Resonance Energy.
For instance, Benzene is roughly 36 kcal/mol (150.6 kJ/mol) more stable than expected. This “extra” stability is why aromatic compounds are so resistant to breaking their pi-system.
Impact on Acid-Base Behaviour
Resonance significantly influences acidity and basicity. The high acidity of carboxylic acids is due to the resonance stabilisation of the resulting carboxylate ion, where the negative charge is dispersed over two oxygen atoms.
In contrast, phenol is less acidic than carboxylic acids but more acidic than ethanol because the phenoxide ion’s negative charge is delocalised into the benzene ring.
Directing Effects in Electrophilic Substitution
In aromatic chemistry, substituents determine where an attacking electrophile will land.
Groups showing a +M effect (like –OH in phenol or –NH₂ in aniline) increase electron density at the ortho and para positions, making them ortho/para directing.
Conversely, groups with a M effect (like –NO₂) decrease density at these positions and are meta-directing.
Resonance vs Inductive Effect: The Battle of Electronic Factors
The inductive effect and resonance effect often operate simultaneously in a molecule, sometimes in the same direction and sometimes in opposition.
Which Predominates?
In most instances, the Resonance Effect, Mesomeric Effect, and Electron Delocalisation are stronger and outweigh the inductive effect.
For example, the –OH and –NH₂ groups are electron-withdrawing by the inductive effect (-I) but electron-donating by resonance (+R).
Because the +R effect is more profound, these groups act as net electron donors to an aromatic ring.
The critical exception occurs with halogens. Halogens are electronegative and exert a strong -I effect, but they also have lone pairs that allow for a +R effect.
In their case, the inductive effect is stronger regarding reactivity (making them deactivating), while the resonance effect controls the orientation (making them ortho/para directing).
How to Determine the Stability of Resonating Structures
Identifying major and minor contributors is key to understanding the true nature of a resonance hybrid. Use these stability of resonating structures rules:
- Rule 1: Non-polar vs Polar structures: The structure with the lowest overall formal charge is the most significant. Non-polar forms are generally more stable than those with charge separation.
- Rule 2: Maximum number of covalent bonds: The contributor with the greatest number of covalent bonds is usually more stable.
- Rule 3: Negative charge on more electronegative atoms: Stability is higher when negative charges are on the most electronegative atoms (like O or N), and positive charges are on the most electropositive atoms.
- Rule 4: Separation of opposite charges: If charges are present, unlike charges should be as close as possible to increase stability.
Resonance vs Mesomeric Effect: What is the Difference?
While IUPAC considers them essentially synonymous, the following comparison table highlights the distinctions often used in educational settings.
| Feature | Resonance Effect | Mesomeric Effect |
|---|---|---|
| Meaning | Interaction between lone pairs and bond pairs determining overall structure. | Stabilisation of a molecule through a functional group or substituent. |
| Causative Agent | Lone electron pair adjacent to a double bond. | Presence of a functional group or substituent in a conjugated system. |
| Nature of Effect | Can describe simple delocalisation within a molecule. | Often describes the polarity developed specifically due to a substituent. |
| Permanence | Always a permanent electronic effect. | A permanent effect shown by resonance structures. |
| Scope | Broader term for electron delocalisation. | Specifically for substituents in conjugated systems. |
| Origin | Linus Pauling (Mathematical analogy). | Christopher Ingold (Electronic displacement). |
| IUPAC Status | Preferred and widely used synonym. | The interaction between lone pairs and bond pairs determines the overall structure. |
Practical Applications of Resonance
Bond Length Equalisation
Resonance explains “abnormal” bond lengths. In benzene, instead of having alternating single (154 pm) and double bonds (134 pm), all C–C bonds are an identical 139 pm.
This intermediate value confirms the delocalised nature of the pi electrons.
Stability of Benzyl and Allyl Carbocations
Resonance delocalisation makes species like allyl and benzyl carbocations much more stable than standard primary carbocations.
The positive charge is not fixed on one carbon but is dispersed over several atoms through the conjugated system.
Acidity of Phenol vs Alcohols
Phenol is a stronger acid than alcohols like ethanol because the resulting phenoxide ion is resonance-stabilised, with the negative charge delocalised into the ring.
The ethoxide ion from ethanol enjoys no such delocalisation and is further destabilised by the positive inductive effect (+I) of the ethyl group.
Conclusion: The Power of Electron Delocalisation
The Resonance Effect, Mesomeric Effect, and Electron Delocalisation represent a cornerstone of modern chemical theory.
By moving beyond rigid, single Lewis structures and embracing the concept of the resonance hybrid, chemists can explain the “impossible” bond lengths of benzene, the high acidity of carboxylic acids, and the specific reactivity of conjugated systems.
Whether preparing for JEE Organic Chemistry or NEET Organic Chemistry, mastering the rules of electron flow is essential for navigating the complex transformations of the molecular world.
Frequently Asked Questions
-
What is the main difference between resonance and the mesomeric effect?
While often used as synonyms, the Resonance Effect is a broad term describing electron delocalisation within any molecule or ion. In contrast, the Mesomeric Effect specifically refers to the permanent polarity or electron shift induced by a functional group (substituent) within a conjugated system, categorised as the +M or -M effect.
-
Why is the resonance hybrid more stable than its canonical structures?
The resonance hybrid is more stable because electron delocalisation spreads the electron density over a larger molecular volume. This dispersal reduces inter-electronic repulsions and lowers the overall potential energy of the molecule. The energy difference between the hybrid and the most stable contributing structure is known as Resonance Stabilisation Energy.
-
Is planarity a requirement for the resonance effect to occur?
Yes, planarity is a strict structural requirement for resonance. For effective electron delocalisation, the p-orbitals of the involved atoms must remain parallel to allow for maximum overlap. If a molecule becomes non-planar (distorted), the orbital alignment is broken, and resonance is effectively prevented.
-
Which is stronger: The Inductive Effect or the Resonance Effect?
In most organic systems, the Resonance Effect is significantly stronger than the Inductive Effect. This is because resonance involves the delocalisation of π-electrons, whereas induction only involves a slight shift in σ-electrons. However, a notable exception occurs in halogens (like Chlorine), where the electron-withdrawing -I effect outweighs the +R effect in terms of overall reactivity.
-
Can resonance happen in saturated hydrocarbons like Alkanes?
No, resonance cannot occur in saturated hydrocarbons like alkanes. Resonance requires a conjugated system with π-bonds, lone pairs, or empty p-orbitals. Alkanes only contain σ-bonds, which are localised between two nuclei and cannot undergo delocalisation in the same manner.

