The periodic table is not merely a chart of elements; it is a sophisticated map that reveals the fundamental patterns of nature.
These patterns, known as periodic table trends, allow chemists to predict the physical and chemical behaviour of an element based on its position.
First conceptualised by Dmitri Mendeleev in the mid-1800s, the table’s power lies in its predictive capability. Mendeleev famously used these gaps to forecast the existence of elements like gallium and germanium before they were ever discovered.
In this comprehensive guide, we will explore the core trends of the periodic table, diving deep into the “why” behind the anomalies that frequently appear in advanced chemistry examinations.
Quick Study Tool: Short on time? Scroll down to the bottom of this post for our Periodic Table Trends Cheat Sheet, perfect for last-minute revision before your chemistry practical or mid-terms!
Table of Contents
The “Why” Foundation: The Two Pillars of Trends
Before we can understand specific Periodic Table Trends, we must examine the two underlying forces that govern almost every atomic property: Effective Nuclear Charge (Zeff) and the Shielding Effect.
1. Effective Nuclear Charge (Zeff)
Zeff is the net positive charge experienced by a valence electron in a multi-electron atom. While the nucleus contains a specific number of protons (atomic number, Z), the outer electrons do not feel the full pull of this positive charge.
This is because the inner-shell electrons act as a buffer. Mathematically, Zeff is often approximated as Z minus the shielding constant (S).
To calculate the exact values, you can refer to the standard Effective Nuclear Charge formulas used in advanced research.
2. The Shielding (Screening) Effect
The shielding effect occurs because electrons in the inner shells repel the outermost valence electrons, effectively “shielding” them from the nucleus’s attractive force.
As you move down a group, the number of inner shells increases, which significantly enhances the shielding effect.
Across a period, however, electrons are added to the same principal energy level, meaning shielding remains relatively constant while the nuclear charge increases.
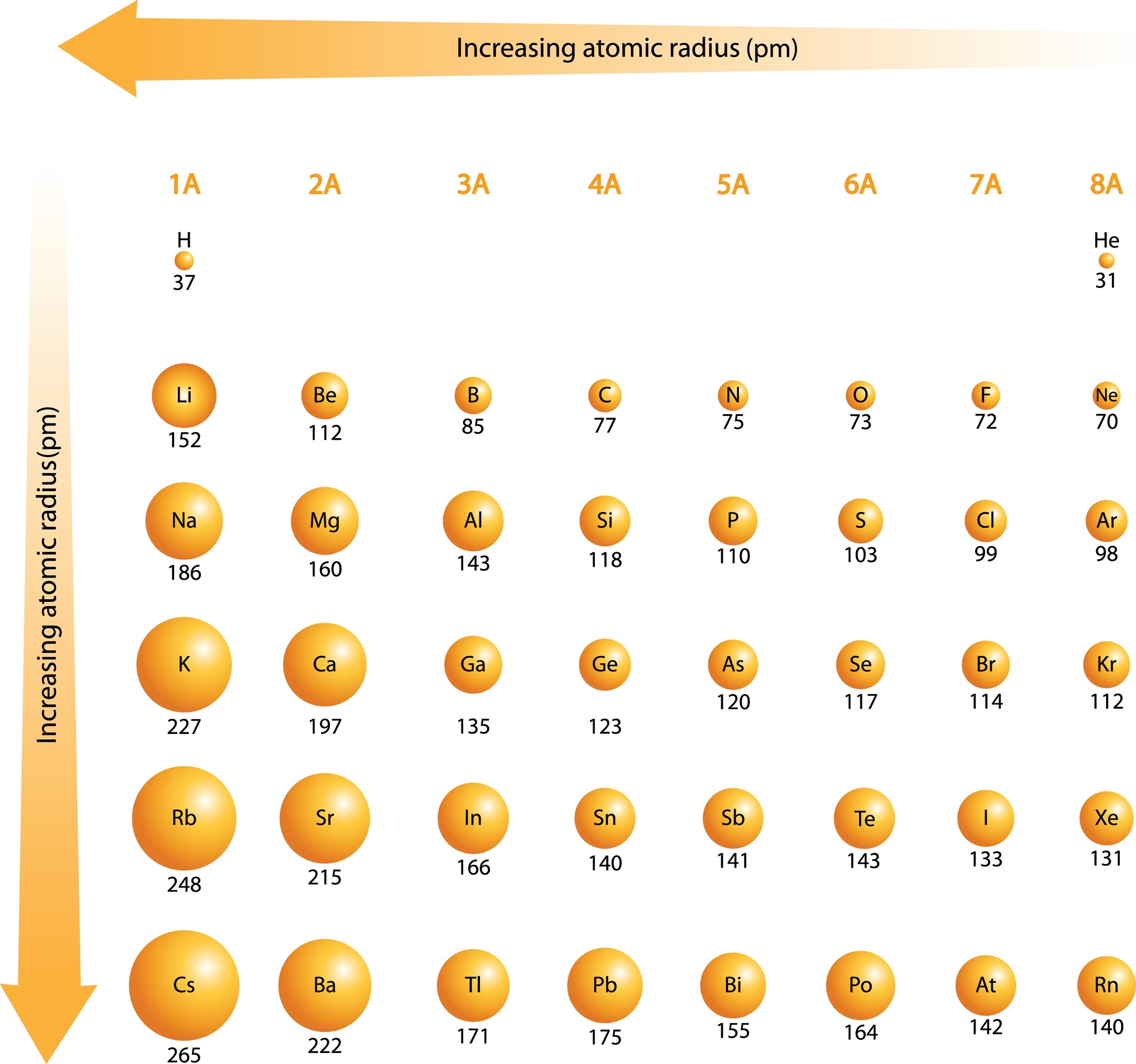
1. Atomic Radius: Defining Atomic Size
The atomic radius is generally defined as the distance from the centre of the nucleus to the outermost electron orbital. Because the boundaries of an atom are “fuzzy,” scientists often measure it as half the distance between the nuclei of two bonded atoms.
The General Trend
- Across a Period (Left to Right): Decreases. As we proceed to the right, each element has one more proton in the nucleus. This increases the Zeff, pulling the electrons in the same shell closer to the nucleus and causing the radius to shrink.
- Down a Group (Top to Bottom): Increases. Moving down a group adds entirely new electron shells or “layers”. Each new shell is further from the nucleus, and the increased shielding from inner electrons outweighs the increase in nuclear charge, leading to a larger atom.
The “Why” Behind the Exceptions
- Gallium vs. Aluminium: According to group trends, gallium should be larger than aluminium. However, Gallium (135 pm) is smaller than aluminium (143 pm). Why? This is due to the d-block contraction. Gallium follows the first row of transition metals; the 3d electrons provide poor shielding, allowing the increased nuclear charge to pull the outer electrons in more tightly.
⚠️ Exam Alert: The Gallium Trap! This is a favourite for examiners in JEE, NEET, and A-Level Chemistry. While you’d expect Gallium to be larger than Aluminium because it’s further down the group, the reality is the opposite. The Secret: It’s all down to d-block contraction. The 3d electrons are terrible at shielding, letting the nucleus “grab” the outer electrons much harder than expected. Always remember: Size of Ga < Size of Al. - The Transition Metal Plateau: Transition metals across a period show less variation in size than representative elements. This is because electrons are added to an inner d-subshell, which shields the outer s-electrons quite effectively, counteracting the increasing nuclear pull.
2. Ionic Radius: When Atoms Gain or Lose Electrons
Ionic radius is the distance from the nucleus to the outermost electron in an ion. The size of an ion differs dramatically from that of its parent neutral atom.
- Cations (Positive Ions): These are smaller than their parent atoms. When an atom loses an electron, it often loses its entire outermost shell. Furthermore, with fewer electrons remaining, the Zeff per electron increases, pulling them closer to the nucleus.
- Anions (Negative Ions): These are larger than their parent atoms. Adding electrons increases electron-electron repulsion within the same shell, forcing the electron cloud to spread out.
Isoelectronic Series Anomaly
Ions like N³⁻, O²⁻, and F⁻ all have 10 electrons (isoelectronic), yet their sizes vary.
- The “Why”: Nitrogen has only 7 protons, whereas Fluorine has 9. Fluorine’s higher proton count exerts a much stronger pull on those 10 electrons, making the F⁻ ion the smallest in the series.
3. Ionisation Energy (IE): The Cost of Removing Electrons
Ionisation energy is the minimum energy required to remove an electron from a neutral gaseous atom.
The General Trend
- Across a Period: Increases. As the atomic radius decreases and Zeff increases, the nucleus holds the valence electrons more tightly, making them harder to remove.
- Down a Group: Decreases. The valence electrons are further from the nucleus and more shielded by inner shells, making the “pull” of the nucleus much weaker.
Mastering these JEE and NEET Important Exceptions is what separates a topper from an average student. Let’s break down the anomalies that defy the standard periodic trends.
While mastering these trends, it is also helpful to understand how they influence Coordination Compounds and their bonding.
Crucial Exceptions (The “Why” Factor)
- Nitrogen vs. Oxygen: Nitrogen has a higher 1st IE than Oxygen, despite being to the left. Why? Nitrogen has a precisely half-filled 2p subshell (2p³), which provides extra stability. Oxygen (2p⁴) has one pair of electrons in a single p-orbital; the repulsion between these two paired electrons makes it easier to remove one.
- Beryllium vs. Boron: Beryllium (2s²) has a higher IE than Boron (2s²2p¹). Why? Beryllium’s 2s orbital is fully filled and “penetrates” closer to the nucleus. Boron’s 2p electron is at a higher energy level and is shielded by the 2s electrons, making it easier to strip away.
- The Lead (Pb) vs. Tin (Sn) Flip: In Group 14, Lead has a higher IE than Tin. Why? This is due to Lanthanoid contraction. The poor shielding of 4f electrons in Lead increases the Zeff on the outer electrons, making them harder to remove than those in Tin.
4. Electron Affinity (EA): Gaining Electrons
Electron affinity is the energy change, usually released when an atom in the gas phase gains an electron to form a negative ion.
The General Trend
- Trend: EA generally increases (becomes more negative) across a period and decreases within a group. Atoms on the right (like Halogens) are very close to a full octet and want to gain electrons desperately to achieve stability.
The Chlorine vs. Fluorine Mystery
While Fluorine is more electronegative, Chlorine has the highest electron affinity in the periodic table.
- The “Why”: Fluorine is a tiny atom. When you try to add an electron to its small 2p orbital, it experiences massive inter-electronic repulsion from the electrons already crowded there. Chlorine, being larger, can accommodate the new electron more comfortably.
5. Electronegativity: The Tug-of-War
Electronegativity is a qualitative measure of an atom’s ability to attract shared electrons in a chemical bond. The Pauling scale is the most common way to quantify this.
The General Trend
- Trend: It increases towards the top-right (Fluorine) and decreases towards the bottom-left (Caesium). Smaller atoms with high Zeff are better at “tugging” shared electrons towards themselves.
The Noble Gas Exception
Noble gases (He, Ne, Ar) are generally assigned an electronegativity of zero.
- The “Why”: Because they possess full valence shells, they are exceptionally stable and have no desire to attract or share additional electrons.
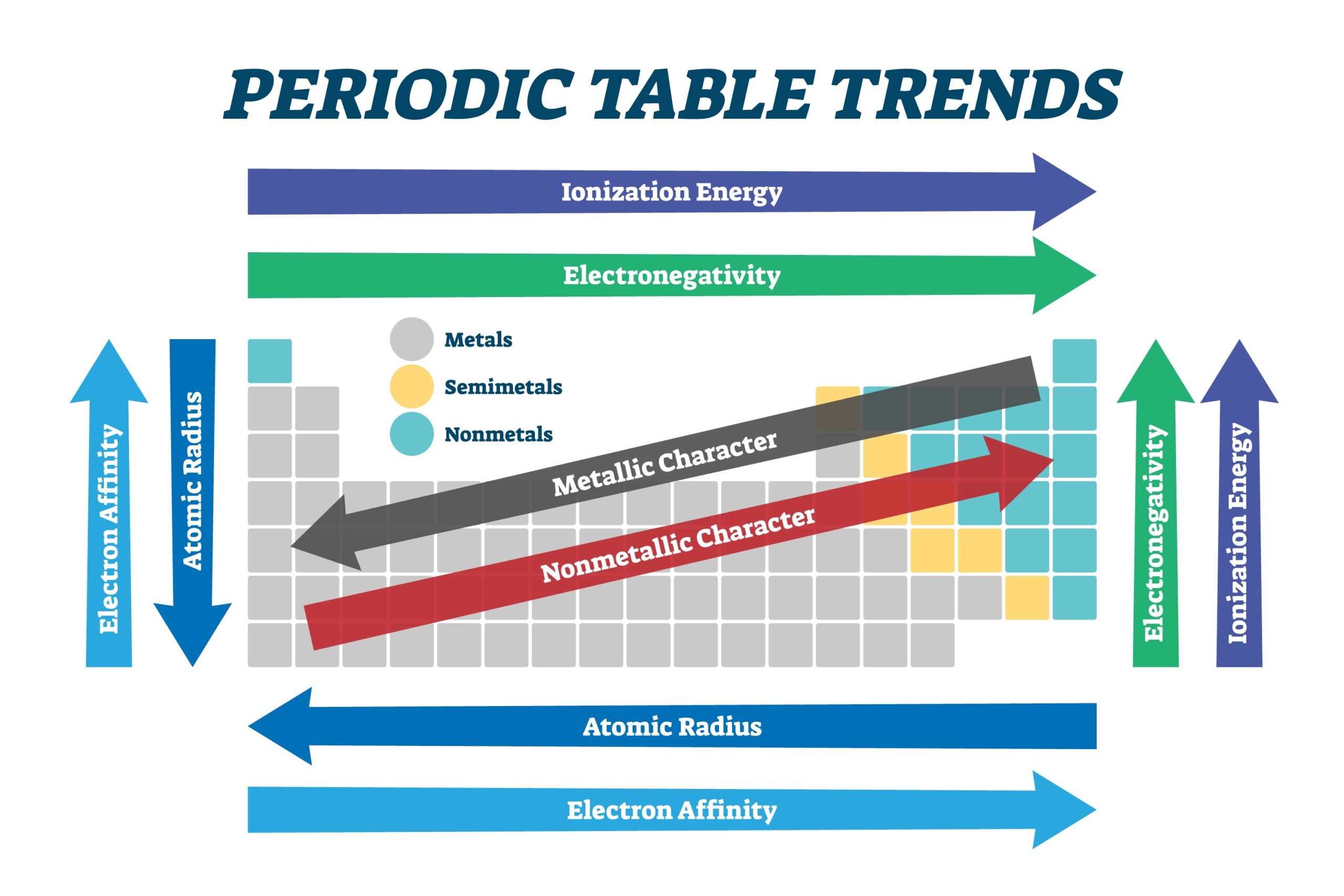
6. Metallic Character and Reactivity
Metallic character is defined by how readily an atom can lose an electron.
- Trend: It increases down a group and decreases across a period. The most metallic and reactive metals (like Francium) are in the bottom-left, while the most reactive non-metals (like Fluorine) are in the top-right.
- Reactivity “Why”: For metals, reactivity increases as atoms get larger because it becomes easier to lose valence electrons. For non-metals, reactivity increases as atoms get smaller because they can more easily attract and “strip” electrons from other atoms.
Summary Table: Periodic Table Trends at a Glance
| Property | Across Period (L to R) | Down Group (T to B) | Primary Reason | Key Exception |
|---|---|---|---|---|
| Atomic Radius | Decreases | Increases | Proton count vs. Shell count | Ga < Al (d-contraction) |
| Ionisation Energy | Increases | Decreases | Distance and Zeff | N > O (half-filled shell) |
| Electronegativity | Increases | Decreases | Ability to attract bond pairs | Noble Gases (usually zero) |
| Electron Affinity | Increases | Decreases | Small size repulsion (Halogens) | Cl > F (size/repulsion) |
| Metallic Character | Decreases | Increases | Ease of losing electrons | Francium (most metallic) |
Check Your Knowledge: Solved Examples of Periodic Trends
To ensure you’ve mastered these concepts, let’s look at two quick Solved Examples:
Example 1: Arrange O, F, and N in increasing order of their first Ionisation Energy.
- Answer: O < N < F.
- Reason: Nitrogen beats Oxygen due to its stable half-filled 2p subshell, but Fluorine is highest because it is furthest to the right.
Example 2: Which has a larger radius: Mg or Mg²⁺?
- Answer: Mg.
- Reason: Cations are always smaller than their parent atoms because losing electrons increases the Effective Nuclear Charge Zeff on the remaining electrons.
Conclusion
Understanding Periodic Table Trends is the key to mastering inorganic chemistry.
By looking past the simple directions on a chart and focusing on the underlying “Why”, the balance between effective nuclear charge, shielding, and orbital stability, you can predict the properties of elements with remarkable accuracy.
Whether it is the d-block contraction in Gallium or the orbital symmetry in Nitrogen, these exceptions are what make the study of the elements a fascinating journey into the architecture of matter.
Frequently Asked Questions
-
Which element has the highest Ionisation Energy in the periodic table?
Ans: Helium (He) has the highest first ionisation energy. This is because Helium is a very small atom with a full valence shell (stable configuration), and its electrons are very close to the nucleus, making them extremely difficult to remove.
-
Why does Chlorine have a higher Electron Affinity than Fluorine?
Ans: Although Fluorine is more electronegative, Chlorine has a higher electron affinity. Fluorine is a very small atom, and adding an electron to its crowded 2p orbital causes significant inter-electronic repulsion. Chlorine is larger and can accommodate the incoming electron more easily.
-
What are the three main factors that affect periodic trends?
Ans: The three primary factors are:
Nuclear Charge (Proton count): Pulls electrons closer.
Shielding Effect: Inner electrons blocking the nuclear pull.
Atomic Radius: The distance between the nucleus and valence electrons. -
Why is the Ionisation Energy of Nitrogen higher than that of oxygen?
Ans: This is a classic exception due to orbital stability. Nitrogen has a half-filled 2p subshell (2p³), which is exceptionally stable. Oxygen (2p⁴) has two electrons paired in one orbital; the repulsion between these paired electrons makes it easier to remove one compared to Nitrogen.
-
What is the trend for Metallic Character in the periodic table?
Ans: Metallic character decreases down a group (as atoms get larger and lose electrons easily) and decreases across a period from left to right (as atoms hold onto their electrons more tightly due to increased nuclear charge).

