“Mixing bleach and ammonia is dangerous because it triggers a rapid chemical reaction that releases toxic chloramine gas (NH₂Cl). This gas causes immediate respiratory distress, lung damage, and in severe cases, can be fatal.”
“As a chemist, I don’t see cleaning products; I see reactive reagents stored in a kitchen cupboard.”
In a modern quest for a sterile environment, many people mistakenly believe that combining common household cleaners will result in a more powerful disinfectant.
However, this practice is a significant risk factor for dangerous chemical mixes at home, as the resulting household chemical reactions can lead to immediate and life-threatening consequences.
The most notorious of these toxic cleaning combinations is the interaction between sodium hypochlorite and ammonium hydroxide.
Mixing bleach and ammonia is an incredibly hazardous practice because it triggers a cascade of molecular changes that produce volatile, poisonous vapours.
This comprehensive bleach safety guide explores the molecular science behind these interactions, the physiological symptoms of exposure, and the essential protocols required to maintain chloramine gas safety in the domestic environment.
Table of Contents
1. The Chemistry of a Deadly Brew: What Happens on a Molecular Level?
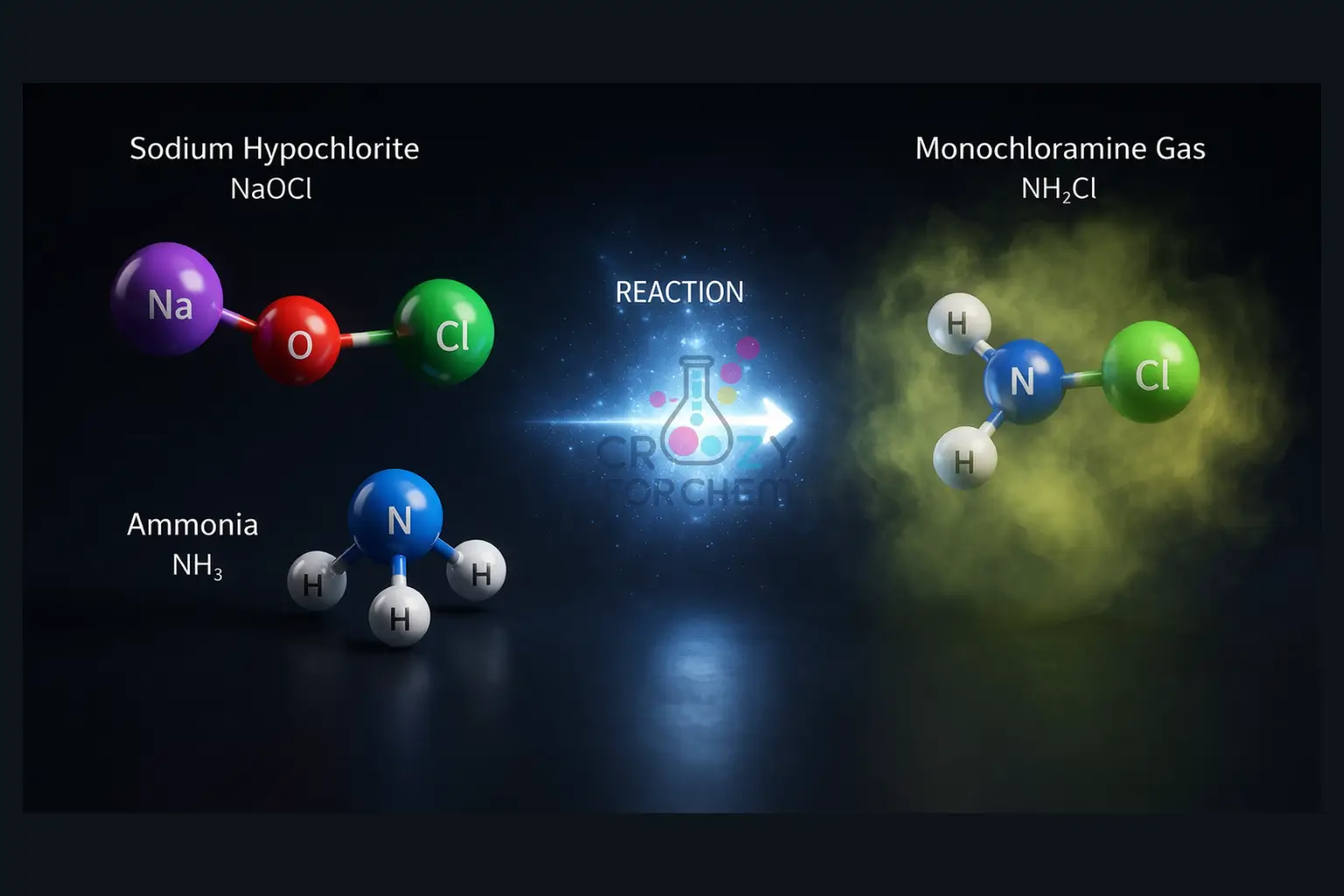
Understanding household chemical reactions is the first step in identifying potential hazards. Household bleach is generally a 3–5% solution of sodium hypochlorite (NaOCl). It is a powerful oxidiser used for disinfecting and laundry.
Ammonia, often found in glass cleaners, floor polishes, and certain paints, is its primary chemical antagonist.
The Formation of Toxic Chloramine Gas
When these two substances are combined, the substance’s chemical identity changes because the atoms rearrange themselves, breaking strong covalent or ionic bonds to form new molecules. The primary result of mixing bleach and ammonia is the production of toxic chloramine gas (NH₂Cl).
The reaction proceeds in stages:
- Initial Decomposition: Bleach first decomposes to form hydrochloric acid (HCl).
- NaOCl → NaOH + HOCl
- HOCl → HCl + O
- The Synthesis of Chloramines: The resulting hydrochloric acid reacts with ammonia to produce monochloramine (NH₂Cl) fumes.
- NaOCl + 2HCl → Cl₂ + NaCl + H₂O
- 2NH₃ + Cl₂ → 2NH₂Cl
This toxic chloramine gas is a powerful irritant and can be further categorised alongside dichloramine and nitrogen trichloride
The Hydra of Hydrazine
In scenarios where ammonia is present in excess, the reaction can progress to a more hazardous stage, forming hydrazine.
Hydrazine is a toxic, not only a respiratory irritant, but also potentially explosive gas that may boil and spray hot, chemically toxic liquid if the mixture is agitated or heated. Sodium chloride and water are formed as by-products of this reaction.
2. Symptoms of Exposure: Identifying the Hazard
Exposure to the fumes generated by mixing bleach and ammonia affects multiple systems in the body, ranging from mild irritation to fatal respiratory failure
The Dangerous household chemical mixes found in modern cleaning kits can cause a range of physiological responses. These are often divided into immediate distress and more insidious, delayed complications.
Immediate Respiratory and Ocular Irritation
Exposure to toxic chloramine gas often triggers instant reactions. Victims may experience:
- Respiratory: Violent coughing, shortness of breath, wheezing, and chest pain.
- Ocular: Burning, watery eyes and redness.
- Gastrointestinal: Nausea and irritation of the nose and throat.
The Silent Threat: Delayed Pulmonary Oedema
A critical component of safety awareness is the distinction between acute and delayed symptoms. While initial irritation may seem to subside, serious conditions can develop hours later.
Chemical Pneumonitis: Inhaling toxic material can cause severe lung inflammation. Acute pneumonitis occurs suddenly, but long-term complications from high concentrations can lead to pneumonia, coma, or death
Pulmonary Oedema: This is a condition where water fills the lungs, often not becoming manifest until several hours of rest and medical observation have passed.
Briefly explain why it’s delayed.- “Chloramine gas is less soluble in water than chlorine, meaning it penetrates deeper into the lower respiratory tract before reacting with moisture in the alveoli, causing a slow build-up of fluid.”
3. Beyond Ammonia: The Forbidden Bleach Combinations
While mixing bleach and ammonia is a primary concern, other Dangerous household chemical mixes must be avoided to prevent different toxic gases from being released
- Bleach + Acids (e.g., Vinegar): Mixing bleach with acidic liquids like vinegar, toilet bowl cleaners, or rust removers converts sodium hypochlorite into deadly chlorine gas (Cl₂). This gas reacts with water in the lungs to produce hydrochloric acid, causing severe tissue damage.
- Bleach + Rubbing Alcohol: This combination synthesises chloroform, a highly toxic substance that can cause dizziness and respiratory failure.
- Bleach + Hydrogen Peroxide: Combining these two substances releases highly flammable oxygen gas, creating a significant fire hazard in the home.
- Bleach + Pet Urine: Urine contains natural ammonia. Using bleach to clean litter boxes or diaper pails can inadvertently release toxic chloramine gas.
- Bleach + Drain Cleaners: Because drain cleaners contain a variety of strong, reactive chemicals, they should never be mixed with bleach, as they can release chlorine gas or other hazardous by-products
Table 1: Summary of Toxic Cleaning Combinations
| Mixture | Resulting Chemical | Principal Health Hazard |
|---|---|---|
| Bleach + Ammonia | Chloramines | Chest pain, shortness of breath, pneumonia |
| Bleach + Vinegar | Chlorine Gas | Severe mucous membrane irritation, death |
| Bleach + Rubbing Alcohol | Chloroform | High toxicity, nervous system depression |
| Hydrogen Peroxide + Vinegar | Peracetic Acid | Highly corrosive to skin and eyes |

4. The Science of Neutralisation: Safe Disposal
Chemical safety does not end with use; it extends to disposal.
Bleach neutralisers are chemicals used to deactivate the harmful effects of sodium hypochlorite before disposal, preventing damage to plumbing and the environment.
Chemists recommend neutralising bleach before sink disposal to mitigate its harmful effects.
Common Bleach Neutralisers
Several agents can be used to safely deactivate sodium hypochlorite:
- Sodium Metabisulfite (Na₂S₂O₅): Frequently used in water treatment and swimming pools. Adding 2.2 grams (one teaspoon) to 2.5 gallons of water effectively neutralises bleach residue.
- Sodium Thiosulfate (Na₂S₂O₃): Commonly known as “Fixer” in photography, it is a valuable neutraliser for lowering chlorine levels in spas.
- Sodium Sulfite (Na₂SO₃): An effective and inexpensive option typically sold under trade names like “De-Chlor”.
- Ascorbic Acid (Vitamin C): Neutralises residual bleach in seconds and is often used for water storage tanks.
- 3% Hydrogen Peroxide: One cup per gallon of water can also serve as a neutraliser.
Note: You must always wear protective clothing, gloves, and goggles when handling these substances
5. Professional Safety: Understanding the Safety Data Sheet (SDS)
In professional and industrial settings, the Safety Data Sheet (SDS) is the primary reference source for managing hazardous chemicals.
All SDSs must comply with specific regulatory guidelines (such as the MSIHC Rules in India or OSHA in the US) and are divided into 16 standardised sections.
Key Sections to Review on an SDS:
- Section 2: Hazards Identification: Lists GHS label elements and pictograms (e.g., corrosive or toxic symbols).
- Section 4: First-aid Measures: Describes acute and delayed symptoms and necessary treatments.
- Section 10: Stability and Reactivity: Lists incompatible materials, such as ammonia and acids, for bleach products.
6. Emergency Protocol and Crisis Management
If an accident involving mixing bleach and ammonia occurs, the first step is always to be prepared. The most critical action is to remove the individual from further exposure to the hazardous substances.
The “METHANE” Reporting Format
When reporting a major chemical incident to emergency services, professionals use the METHANE mnemonic to provide accurate information:
- M: Major Incident (Has one been declared?).
- E: Exact Location (Where did the spill happen?).
- T: Type of Incident (Chemical spill/gas release).
- H: Hazards (What fumes are present?).
- A: Access (Best entry route for responders).
- N: Number of Casualties (How many people are affected?).
- E: Emergency Services (Which ones are already on-site?).
The STARC Principle for Communication
When informing family members or neighbours of a chemical event, follow the STARC principle:
- Simple: Use words people understand.
- Timely: Provide information as soon as possible.
- Accurate: Ensure the facts are correct.
- Relevant: Answer the public’s immediate questions.
- Credible: Openness is key to trust.
7. Step-by-Step Emergency Actions
If you smell a pungent odour or experience dizziness while cleaning, follow this protocol immediately:
- EVACUATE: Leave the contaminated area immediately to find fresh air. Do not stay to “finish the job” or clean up the spill.
- REMOVE CONTAMINANTS: If chemicals have splashed on your skin or eyes, irrigate with clean water or 0.9% saline for at least 15–20 minutes. Remove contaminated clothing and seal it in a plastic bag for disposal.
- VENTILATE: Only return to the area once it is safe to do so. Open all windows and doors, and use fans to dissipate remaining toxic chloramine gas.
- SEEK MEDICAL HELP: Call 999 (UK) or 911 (US) if someone is unconscious, has chest pain, or is struggling to breathe. For minor exposure, contact a Poison Information Centre for specific treatment advice.
8. Lessons from History: Case Studies
The dangers of mixing incompatible chemicals extend beyond the kitchen sink to large-scale disasters.
- Toulouse, France (2001): An explosion at a chemical factory involving ammonia and ammonium nitrate resulted in 31 deaths and thousands of injuries, highlighting the catastrophic potential of stored reactive agents.
- Neishapur, Iran (2004): A train derailment caused the pooling and mixing of incompatible flammable chemicals, leading to a massive explosion that destroyed an entire town.
- Mumbai, India (2010): A chlorine gas leak during the disposal stage of the chemical life cycle required mass evacuation and emergency medical response.
- Bhopal Gas Disaster (1984): While involving Methyl Isocyanate, this remains the ultimate lesson in the need for strict adherence to safety standards and the devastating impact of toxic gas on a populated area.
9. Conclusion: Respecting the Chemistry in Your Cupboard
The Chemistry of Indian spices or the fermentation of milk into curd are beautiful domestic chemical transformations, but the cleaning cupboard represents a different, more volatile side of science.
By understanding the household chemical reactions that occur when you mix bleach and ammonia, you transform cleaning from a mundane chore into a disciplined safety practice.
The Golden Rule remains: Bleach should only ever be mixed with plain water.
By spending 30 seconds reading an ingredient label and identifying “Sodium Hypochlorite” or “Ammonium Hydroxide,” you move from being a potential victim of a chemical accident to an informed, safe consumer. Remember, in the world of reactive reagents, “Hope for the best, but plan for the worst”.
Frequently Asked Questions (FAQs)
-
What happens if I accidentally mix bleach and ammonia?
Mixing bleach and ammonia creates toxic chloramine gases (NH₂Cl). Exposure causes immediate symptoms like violent coughing, nausea, and shortness of breath. If you smell a sharp, acrid odour after mixing cleaners, leave the room immediately and seek fresh air. As a chemist, I advise remaining under medical observation for 48 hours to monitor for delayed pulmonary oedema.
-
Is it safe to mix bleach with vinegar for “natural” cleaning?
No, this is extremely dangerous. Bleach is alkaline, and vinegar is acidic. Mixing them triggers a reaction that releases Chlorine gas (Cl₂). Even in small amounts, chlorine gas is highly toxic and can cause severe damage to the respiratory system, eyes, and throat.
-
How long does chloramine gas stay in the air?
Chloramine gas can linger for several hours, depending on your home’s ventilation. Do not re-enter the contaminated area until it has been thoroughly aired out. It is recommended to wait at least 2 to 4 hours with all windows and doors open before returning to the room.
-
Can I use two different cleaners on the same surface?
Only if the first cleaner is completely rinsed away with water and the surface is dry. You must ensure there is no chemical residue left behind. Mixing even small traces of incompatible chemicals on a sponge or a surface can still release hazardous vapours.
-
Is it safe to use bleach in a toilet bowl if I have already urinated?
No, you should always flush the toilet thoroughly before adding bleach. Urine contains naturally occurring ammonia. Adding a bleach-based disinfectant to a bowl containing urine can inadvertently release toxic chloramine gas in the confined space of a bathroom.
-
How do I know if a cleaning product contains ammonia?
Always check the ingredient label for “Ammonia” or “Ammonium Hydroxide”. It is most commonly found in window cleaners, floor polishes, and some heavy-duty degreasers. If a label warns “Do not mix with bleach,” it often contains ammonia or an acid.
-
Why does water help with chemical eye exposure?
Copious amounts of water or saline irrigate the corrosive agent, effectively diluting its concentration. This mechanical rinsing action is critical for stopping the chemical reaction and preventing irreversible tissue damage to the cornea and mucous membranes.
-
What should I look for on a product label to ensure safety?
Look for GHS (Globally Harmonised System) pictograms. Icons showing a “Corrosive” symbol (test tubes pouring on a hand) or a “Skull and Crossbones” (Acute Toxicity) indicate that the product requires extreme caution. Always read the Safety Data Sheet (SDS) if you are unsure about chemical compatibility.

