In the intricate world of organic chemistry, the reactivity of a molecule is rarely a matter of chance. Instead, it is governed by the distribution of electron density across its framework.
One of the most fundamental concepts used to describe these electronic shifts is the Organic Reaction Mechanisms framework, specifically the Inductive Effect.
This phenomenon involves the transmission or communication of electric charge through a chain of atoms, resulting in a permanent polarity within the molecule.
By understanding how electrons are “pushed” or “pulled” through sigma bonds, students and researchers can predict everything from the stability of carbocations to the acidic strength of carboxylic acids.
Table of Contents
What is the Inductive Effect?
Definition and Origin
The Inductive Effect is defined as the phenomenon in which a permanent dipole arises in a given molecule due to the unequal sharing of bonding electrons.
This unequal sharing is primarily driven by the difference in electronegativity between the two bonded atoms.
When an atom or a group of atoms with a different electronegativity than carbon is introduced into a chain, it causes a slight displacement of electrons, leading to a permanent dipole.
The Concept of Permanent Dipole
Unlike temporary effects that only manifest in the presence of an external reagent, the Inductive Effect represents a permanent phenomenon.
It results in a molecule where one end becomes slightly positively charged (Δ) and the other slightly negatively charged (Δ).
This tiny imbalance of charge can affect the behaviour of the entire molecule, influencing its physical properties such as dipole moments and its chemical reactivity.
Essential Characteristics of the Inductive Effect
A Permanent Electronic Phenomenon
The Inductive Effect is a long-lasting, permanent displacement of electrons along a covalent bond. It is inherent to the molecule’s ground state and does not require an attacking reagent to manifest.
While it is a permanent effect, it is generally considered a weak electronic force compared to stronger influences such as resonance (mesomeric) or hyperconjugation.
Transmission through Sigma Bonds
The mechanism of the Inductive Effect involves the sigma bond electron displacement.
This is a critical distinction in organic chemistry: while the electromeric effect only occurs in pi (π) bonds, the Inductive Effect operates exclusively through sigma σ bonds.
The electrons involved are tightly held within these single bonds, and the effect is relayed from one atom to the next along the carbon chain.
Distance Dependency in Organic Chemistry
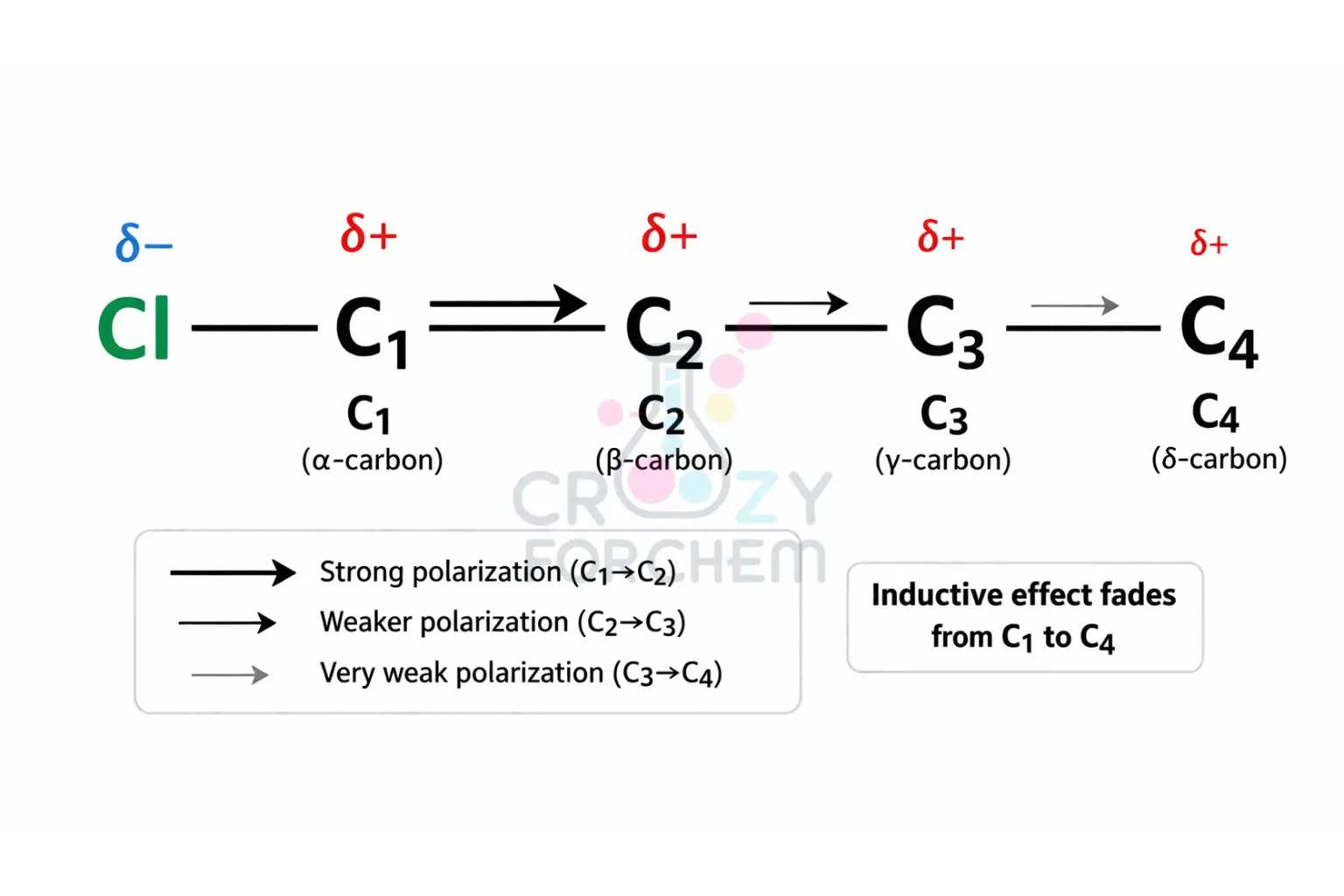
Perhaps the most significant feature of this effect is its distance dependency in organic chemistry.
The induced change in polarity is greatest at the bond closest to the electronegative group and weakens rapidly as it moves further along the chain.
Practically, the effect is observed to be maximum for the first two carbons, becomes negligible after the third carbon C₃, and is almost insignificant after four bonds from the active group.
Experimental data suggest that only about 10% of the effect is transferred to the next atom in the sequence.
Types of Inductive Effects: +I and -I
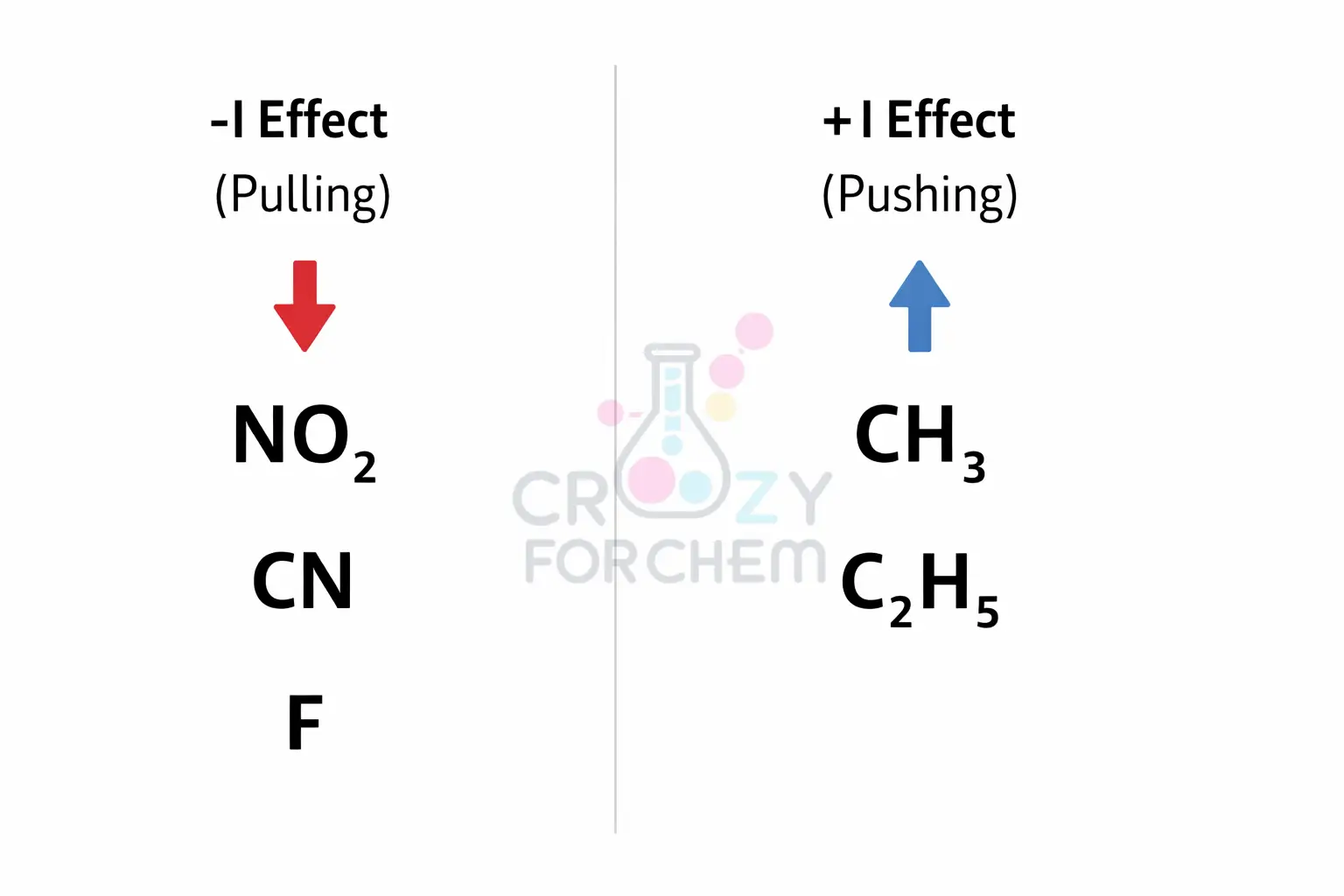
Groups are classified by their ability to either donate or withdraw electron density compared to a hydrogen atom, which is used as the standard reference point.
Negative Inductive Effect (-I effect)
The -I effect is caused by electron-withdrawing groups (EWG). These are atoms or groups with an electron-withdrawing ability stronger than that of hydrogen.
They pull electron density away from the rest of the molecule through the sigma bonds, creating a partial positive charge on adjacent atoms.
-I effect examples include:
- Halogens: Fluorine (-F), Chlorine (-Cl), Bromine (-Br), and Iodine (-I).
- Nitro groups: NO₂.
- Carbonyl and Carboxyl groups: -CHO, -COOH, and -CO.
- Cyano groups: -CN.
- Sulphonic acid groups: -SO₃H.
The relative order of the -I effect for some common groups is: NH₃⁺ > NO₂ > CN > SO₃H > CHO > CO > COOH > F > Cl > Br > I > OH > OR > NH₂ > C₆H₅ > H
Positive Inductive Effect (+I effect)
The +I effect is caused by electron-donating groups (EDG). These groups have a stronger ability to release electrons than hydrogen.
They push electron density toward the rest of the molecule, slightly increasing the electron density on nearby atoms.
+I effect examples include:
- Alkyl groups: Methyl, Ethyl, and Isopropyl.
- Negatively charged groups: -O⁻ and -COO⁻.
- Alkali metals: Highly electropositive atoms.
The relative order of the +I effect for alkyl groups is: -C(CH₃)₃ > -CH(CH₃)₂ > -CH₂CH₃ > -CH₃. This order exists because more substituted alkyl groups provide greater electron-donating support.
Inductive Effect vs Resonance: The Battle of Dominance
In many organic molecules, the Inductive Effect and the Resonance (Mesomeric) Effect operate simultaneously, often in opposite directions. Understanding which one “wins” is the key to predicting reactivity.
The General Rule: Resonance Wins
Generally, the Resonance Effect is stronger than the Inductive Effect. This is because resonance involves the actual delocalisation of π-electrons or lone pairs over a larger molecular area, whereas induction only involves the slight shifting of σ-electrons.
- Example: In Aniline (-NH₂ on Benzene), the nitrogen has a -I effect (withdrawing) but a powerful +R effect (donating). Since resonance dominates, the ring becomes electron-rich.
The Halogen Exception (Crucial for JEE/NEET)
Halogens (F, Cl, Br, I) are the most famous exception to the general rule. When a halogen is attached to a benzene ring:
- Inductive Effect (-I): They withdraw electrons strongly via sigma bonds.
- Resonance Effect (+R): They donate their lone pair via pi bonds.
The Outcome: For halogens, the Inductive Effect (-I) dominates over the Resonance Effect (+R) in terms of overall reactivity.
This is why Chlorobenzene is less reactive than Benzene toward electrophilic substitution. However, the resonance effect still dictates the orientation (ortho/para directing).
Quick Comparison Table: Dominance at a Glance
| Situation | Dominant Effect | Reason |
| General Organic Compounds | Resonance (R/M) | π-electron delocalisation is more effective than σ-shift. |
| Halogens on Benzene | Inductive (-I) | Poor 2p-3p (or higher) orbital overlap weakens resonance. |
| Distance > 3 Carbons | None | Inductive effect vanishes; Resonance only works if conjugated. |
Practical Applications in Organic Chemistry
Acidic Strength of Carboxylic Acids
The Inductive Effect is a primary tool for predicting the acidic strength of carboxylic acids. Acidity is essentially the ability of a molecule to donate a proton (H⁺).
When an acid loses a proton, it forms a conjugate base (RCOO⁻). The stability of this conjugate base determines the strength of the acid.
Electron-withdrawing groups (-I) increase acidity by pulling electron density away from the carboxylate group, which stabilises the negative charge through delocalisation.
Conversely, electron-donating groups (+I) decrease acidity because they push electron density toward the already negative conjugate base, causing destabilisation through inter-electronic repulsions.
Example Cases:
- Formic Acid vs Acetic Acid: Formic acid is more acidic than acetic acid because the methyl group in acetic acid has a +I effect, which destabilises the conjugate base.
- Halogenated Acids: Fluoroacetic acid is a stronger acid than chloroacetic acid, which in turn is stronger than bromoacetic acid. This follows the electronegativity of the halogens (F > Cl > Br).
- Substitution Levels: The acidity of chloroacetic acids increases with the number of chlorine atoms: trichloroacetic acid > dichloroacetic acid > monochloroacetic acid.
Stability of Reaction Intermediates
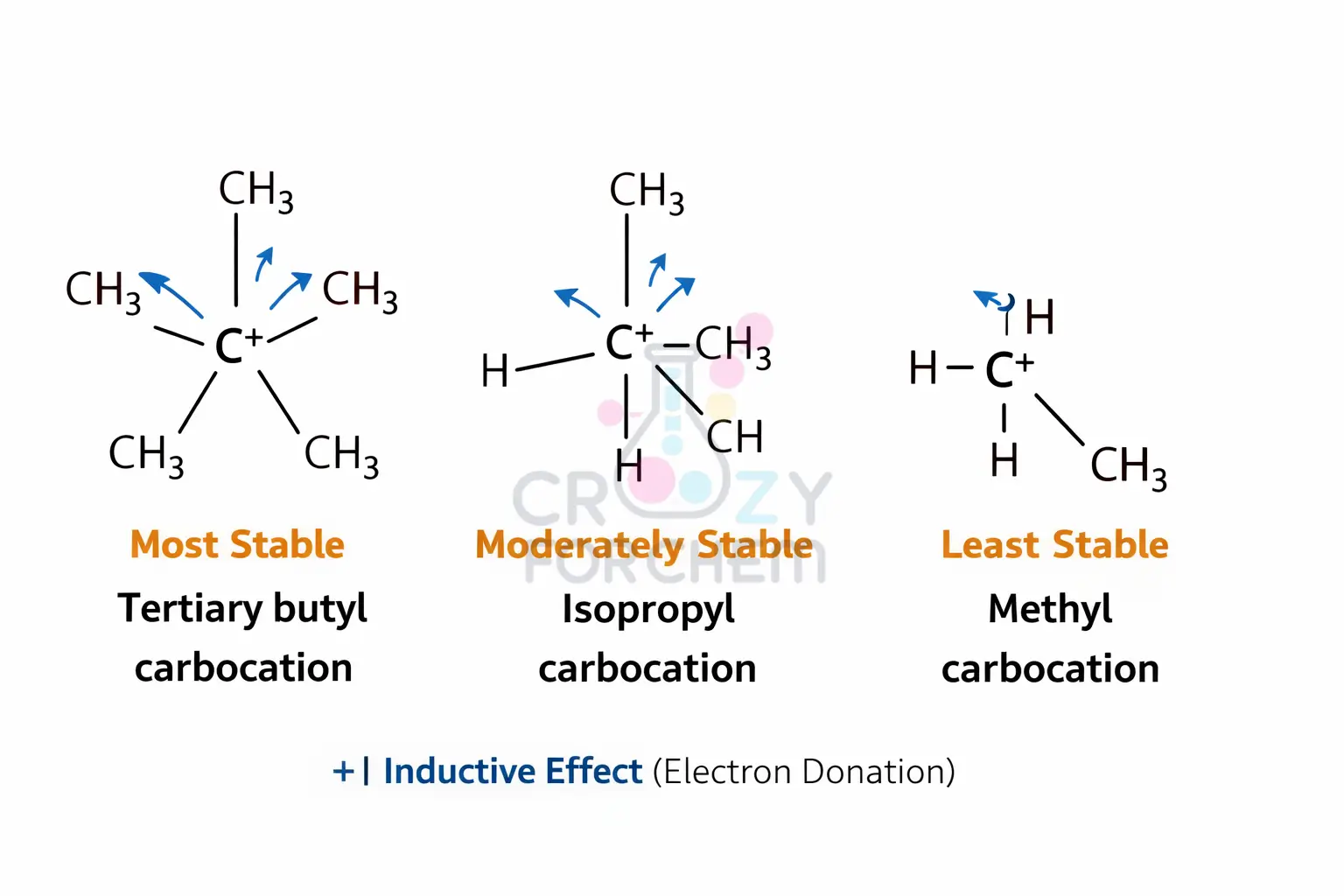
The stability of carbocations and carbanions is heavily influenced by induction. The Inductive Effect stabilises Reaction Intermediates (like carbocations) by dispersing the charge, which lowers the overall potential energy of the system.
- Carbocations: These are positively charged, electron-deficient carbon species. Alkyl groups stabilise carbocations via the +I effect by donating electron density toward the positive centre, thereby reducing the overall positive charge. Consequently, a tertiary carbocation is more stable than a secondary or primary one.
- Carbanions: These negatively charged species are stabilised by electron-withdrawing groups (-I), which help reduce the excess negative charge. They are destabilised by +I groups.
- Transition State vs Intermediate: While the Inductive Effect works on intermediates, it also influences the energy of the Transition State. By stabilising the species that forms during the reaction, induction lowers the activation energy, thereby increasing the reaction rate.
Basicity of Amines
The Inductive Effect also determines the basicity of compounds like amines. Basicity depends on the availability of a lone pair of electrons on the nitrogen atom for protonation.
- +I groups (like alkyl groups) increase basicity by pushing electron density toward the nitrogen, making the lone pair more available. For example, isopropylamine is more basic than ethylamine, which is more basic than ammonia.
- -I groups (like chlorine) reduce basicity by pulling electron density away from the nitrogen, making it harder for the molecule to donate its lone pair.
Inductive Effect vs Electromeric Effect
It is crucial to differentiate the Inductive Effect from other electronic phenomena like the electromeric effect.
| Complete transfer of an electron pair | Inductive Effect | Electromeric Effect |
| Bond Involvement | Sigma (σ) bonds only | Pi (π) bonds only |
| Nature | Permanent | Temporary (occurs in presence of reagent) |
| Electron Shift | Partial displacement | Complete transfer of electron pair |
| Charge Produced | Partial charges (δ+, δ-) | Full formal charges (+, -) |
Conclusion: The Foundation of Electronic Effects
The Inductive Effect serves as a vital logical framework for navigating the complexities of organic synthesis.
By mastering how permanent polarity arises from sigma bond electron displacement, students can move away from rote memorisation toward a predictive understanding of molecular behaviour.
Whether evaluating the acidic strength of carboxylic acids, the basicity of amines, or the stability of carbocations, the Inductive Effect remains an indispensable tool.
Acknowledging its distance dependency in organic chemistry ensures a precise application of this concept, allowing chemists to account for the subtle electronic nuances that define chemical reactivity.
Frequently Asked Questions (FAQs)
-
Why is the inductive effect considered a distance-dependent property?
The inductive effect is a distance-dependent phenomenon because it operates through the polarisation of σ-bonds. Since electrons in single bonds are tightly held by atomic nuclei, the displacement of electron density diminishes rapidly as it is transmitted along the carbon chain. Experimental evidence shows the effect is strongest at the first carbon and becomes negligible after the third carbon (
C₃) atom. -
Which electronic effect is stronger: Inductive or Resonance?
In almost all organic systems, the Resonance (Mesomeric) Effect is significantly stronger than the inductive effect. Resonance involves the delocalisation of π-electrons over a larger molecular framework, whereas induction only involves a slight shift of σ-electrons. However, a notable exception occurs with halogens (like Chlorine) attached to a benzene ring, where the electron-withdrawing -I effect dominates the overall reactivity, even though the +R effect dictates the orientation.
-
How does the -I effect increase the acidic strength of carboxylic acids?
An electron-withdrawing group (-I effect), such as Chlorine in chloroacetic acid, pulls electron density away from the O-H bond. This polarization weakens the bond, making it easier to release a proton (H⁺). Furthermore, the -I effect stabilises the resulting carboxylate anion (conjugate base) by dispersing its negative charge, which fundamentally increases the acidity of the molecule.
-
Is the inductive effect a permanent or temporary phenomenon?
The inductive effect is a permanent electronic effect. It is an inherent property of the molecule’s ground state, arising solely from the differences in electronegativity between bonded atoms. Unlike the electromeric effect, it does not require the presence of an external attacking reagent to manifest.
-
How do alkyl groups stabilise carbocations via the +I effect?
Alkyl groups (such as methyl or ethyl groups) possess a positive inductive effect (+I), meaning they are electron-donating. They “push” electron density towards the positively charged, electron-deficient carbon atom of a carbocation. This dispersal of the positive charge lowers the potential energy of the intermediate, making tertiary carbocations more stable than primary ones (3° > 2° > 1°).

