In the complex landscape of electronic effects, Hyperconjugation in Organic Chemistry stands as a pivotal concept for explaining the unexpected stability of certain molecular frameworks.
Often described as a general stabilising interaction, it bridges the gap between simple Lewis structures and the more sophisticated molecular orbital theory.
By understanding how sigma (σ) electrons delocalise, chemists can rationalise the behaviour of reactive intermediates, the orientation of elimination reactions, and the physical dimensions of molecules themselves.
Table of Contents
What is Hyperconjugation?
Definition and Fundamentals
Hyperconjugation, also frequently referred to as σ-conjugation or no-bond resonance, is defined as the delocalisation of electrons through the participation of bonds that are primarily σ in character.
In traditional resonance theory, electron delocalisation was thought to occur only via the parallel overlap of p-orbitals.
However, hyperconjugation posits that delocalisation can also occur via the parallel overlap of hybridised orbitals participating in sigma bonds with adjacent empty p-orbitals or antibonding π orbitals.
This interaction results in the formation of extended molecular orbitals, allowing electron density to be shared over a larger number of atoms.
Why “No-Bond Resonance”?
The term no-bond resonance is a descriptive label arising from the specific way these interactions are represented on paper.
In the contributing canonical structures, the sigma bond between the carbon and the α-hydrogen appears to be broken.
This suggests that there is no formal bond between these two atoms in the resonance form, although the hydrogen atom remains in close proximity.
This broken connection is what allows the delocalisation to be visualised using standard resonance arrows.
The Baker-Nathan Effect
Historically, this phenomenon is often used synonymously with the Baker-Nathan effect.
It is named after the researchers who applied the concept to specific chemical reactions where the observed stability trends of alkyl groups contradicted predictions based purely on the inductive effect.
In modern chemistry, while “hyperconjugation” is the broader term for the electronic effect itself, “Baker-Nathan effect” remains a common historical reference in many textbooks and competitive exam syllabi.
Hyperconjugation vs Resonance vs Inductive Effect
Understanding the differences between the three major electronic effects—Inductive, Resonance, and Hyperconjugation is vital for any student of organic chemistry.
- Inductive Effect: This is a permanent effect involving the partial displacement of electron density along a σ-bond due to differences in electronegativity. Unlike hyperconjugation, it does not involve the sharing of electron clouds between different types of orbitals; it is merely a pull through the existing bond framework.
- Resonance (Mesomeric Effect): This involves the delocalisation of π-electrons or lone pairs through alternating single and double bonds or p-orbitals. It is generally a much stronger effect than hyperconjugation because it involves the full movement of electrons within a pi-system.
- Hyperconjugation: This occupies a middle ground, involving the delocalisation of σ-electrons into adjacent empty or partially filled p-orbitals or π-systems. It is a permanent effect that is generally more pronounced than induction but less so than full resonance.
The “Strength Hierarchy”: Which effect wins?
In a competition between these effects to determine the stability or reactivity of a molecule, a clear hierarchy of strength is observed: Resonance > Hyperconjugation > Inductive Effect.
This means that if a carbocation can be stabilised by both resonance and hyperconjugation, the resonance stabilisation will contribute the most to its overall energy reduction.
However, Hyperconjugation in Organic Chemistry remains a significant factor because it often provides the crucial margin of stability in systems where resonance is not possible.
| Feature | Inductive Effect | Hyperconjugation | Resonance |
| Bonds Involved | σ-bonds only | σ to p or π* | π-bonds or Lone Pairs |
| Distance | Decreases rapidly | Permanent & Constant | Permanent & Constant |
| Mechanism | Electronegativity pull | Orbital Overlap | Delocalisation |
| Strength | Weakest | Intermediate | Strongest |
Essential Conditions for Hyperconjugation
Not every molecule can exhibit hyperconjugation; specific structural requirements must be met for this interaction to occur.
The Requirement of Alpha (α) Hydrogens
The most critical requirement is the presence of an α-hydrogen.
This is a hydrogen atom attached to an sp³ hybridised carbon (the α-carbon) that is directly adjacent to an unsaturated system, such as a double bond, or an electron-deficient centre, like a carbocation.
The electrons within these sp³ C-H sigma bonds serve as the “source” for delocalisation. Without α-hydrogens, hyperconjugation simply cannot occur, as seen in ethylene, which lacks adjacent alkyl groups.
Orbital Alignment
Effective delocalisation requires proper orbital alignment. The σ-orbital of the C-H bond must be “more or less parallel” to the adjacent empty p-orbital or π-system to allow for meaningful overlap.
This parallel orientation facilitates the maximum transfer of electron density.
While free rotation around C-C single bonds means that only one C-H bond may be perfectly aligned at any given moment, the rotation is fast enough that all available α-hydrogens can contribute to the overall stabilisation over time.
Structural Representation & Drawing Structures
How to draw hyperconjugating structures
Drawing hyperconjugating structures involves representing the flow of sigma electrons using curved arrow notation.
In a molecule like propene, an arrow is drawn from a C-H sigma bond of the methyl group to the adjacent C-C single bond.
Simultaneously, the π-bond of the double bond shifts to the terminal carbon, which then carries a negative charge. The hydrogen from the broken sigma bond is represented as H⁺ with no formal bond to the carbon.
Counting the number of structures: (n + 1) rule
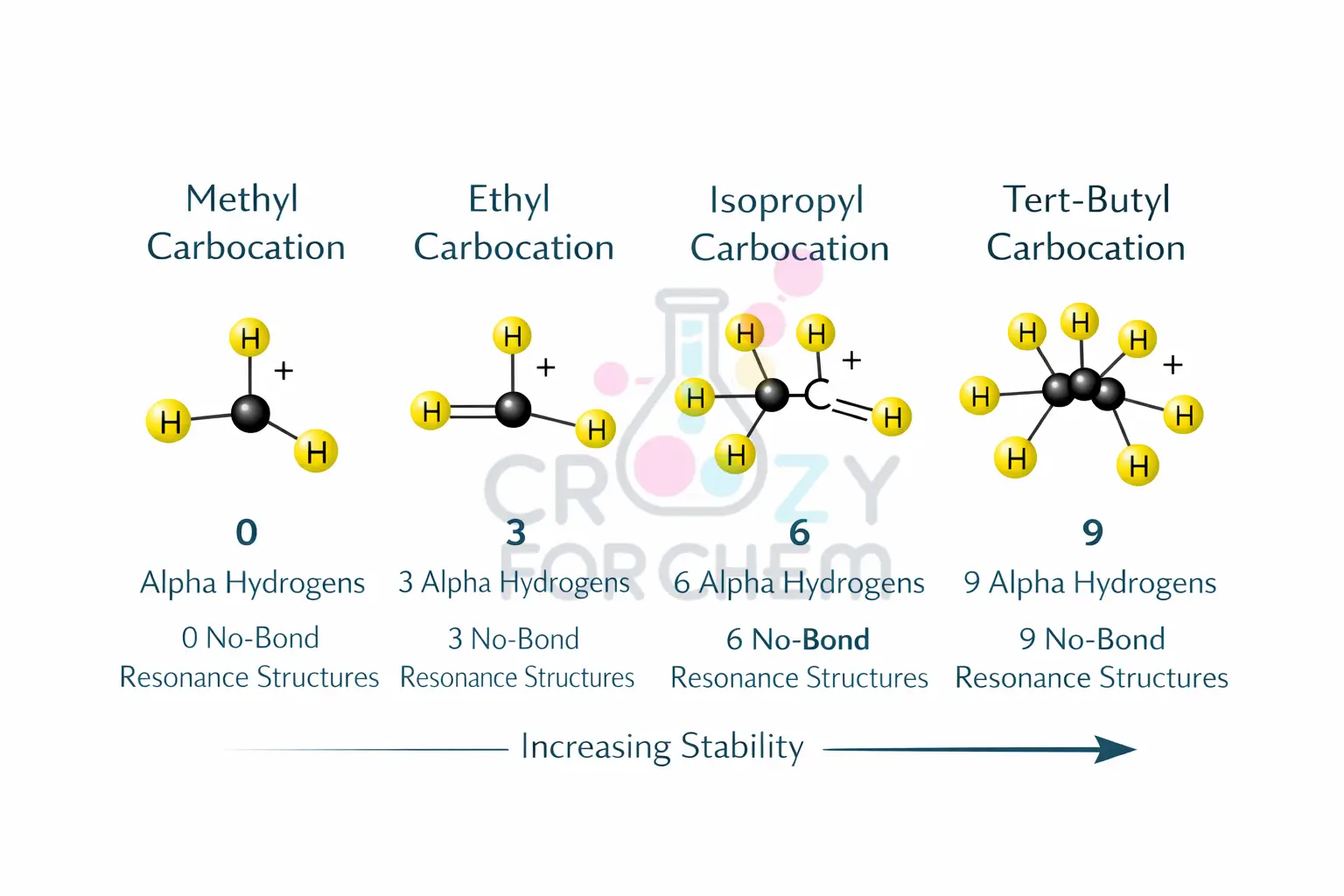
The total number of contributing resonance forms for a molecule is determined by the (n + 1) rule, where n represents the number of available α-hydrogens. For example:
- Ethyl carbocation: Has three α-hydrogens, leading to 3 + 1 = 4 structures.
- Isopropyl carbocation: Has six α-hydrogens, leading to 6 + 1 = 7 structures.
- Tert-butyl carbocation: Has nine α-hydrogens, leading to 9 + 1 = 10 structures. The more structures that can be drawn, the greater the dispersal of charge and the more stable the molecule becomes.
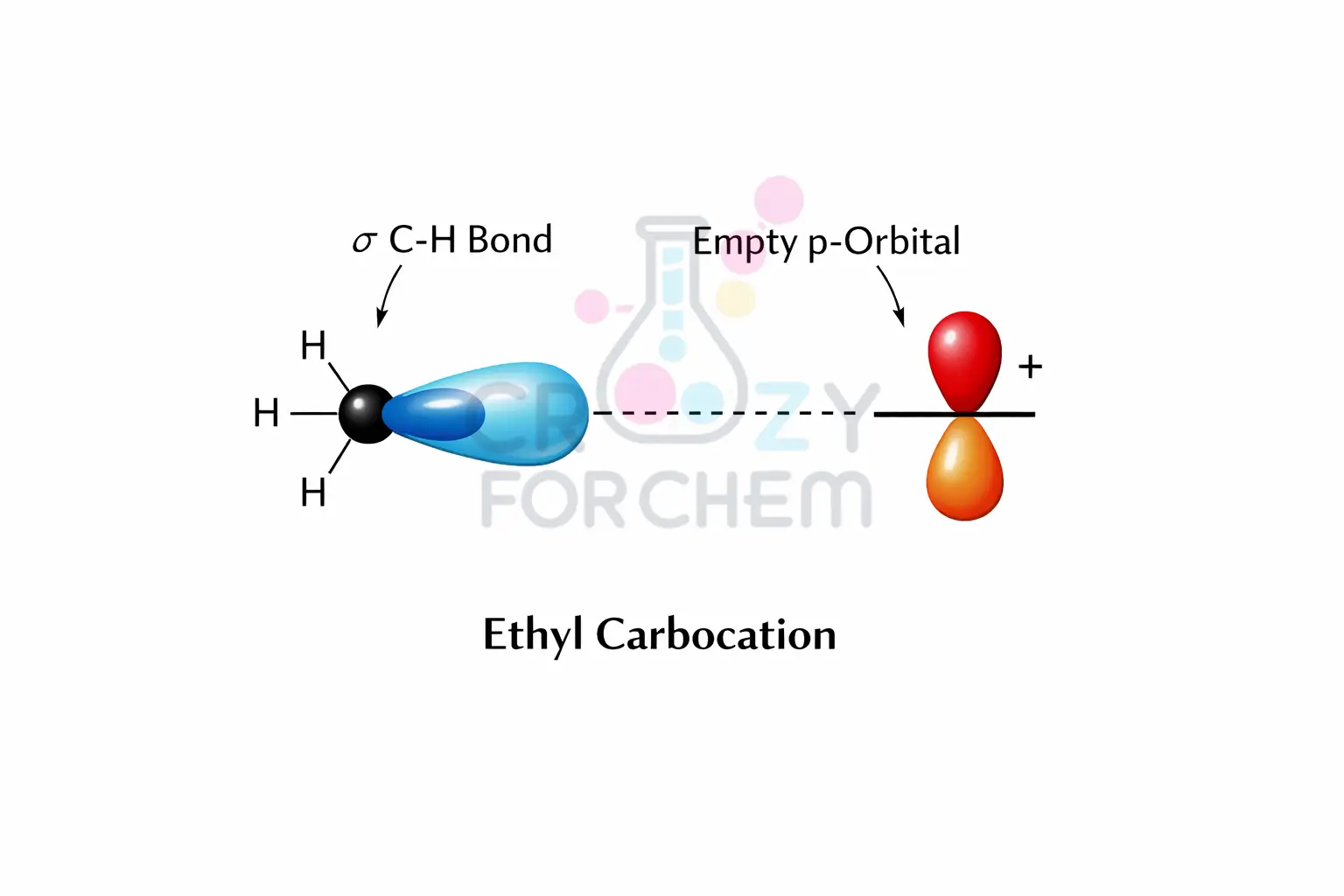
The Mechanism of Sigma Electron Delocalisation
The fundamental mechanism of hyperconjugation is sigma electron delocalisation. To understand this, one can use the analogy of a box with two compartments filled with gas.
If gas is at a high concentration on one side and a low concentration on the other, opening a valve allows the gas to flow and spread out until equilibrium is reached.
In a similar fashion, electrons act like waves or clouds rather than fixed sticks. The C-H sigma bonds around an alkyl group represent an area of high electron density.
When adjacent to an empty p-orbital (low density), these “electron clouds” spread out to fill the deficiency.
This spreading out of the electron density effectively disperses the positive charge over a larger number of atoms, lowering the potential energy and increasing the carbocation stability.
Impact on the Stability of Reactive Intermediates
Stability of Carbocations
Hyperconjugation is the primary factor used to explain the relative carbocation stability of different alkyl ions.
In a tertiary carbocation, nine α-hydrogens are available to share their electron density with the positive centre, dispersing the charge over a total of ten atoms. In a primary ethyl carbocation, only three α-hydrogens contribute.
This results in the well-known stability order: Tertiary > Secondary > Primary > Methyl.
Stabilisation of Free Radicals
The same logic applies to the stability of free radicals. In a radical, the p-orbital is only partially filled with a single unpaired electron.
Hyperconjugation allows electron density from adjacent C-H bonds to be shared with this half-filled orbital, providing stabilisation.
Consequently, tertiary radicals are more stable than secondary or primary ones due to the increased number of available α-hydrogens.
Hyperconjugation in Alkenes and Unsaturated Systems
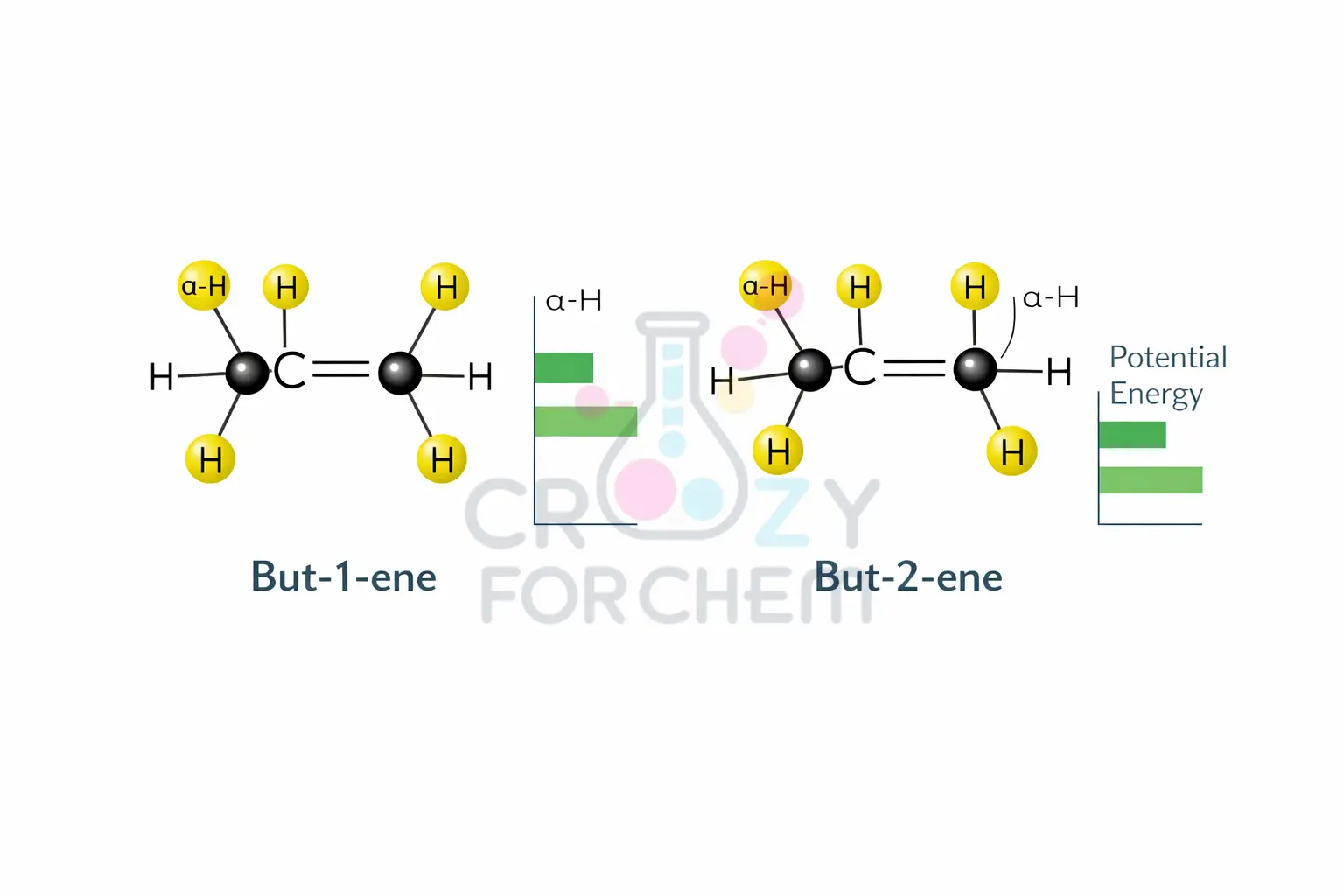
Alkene Stability Trends (Saytzeff’s Rule)
In unsaturated compounds, Hyperconjugation in Organic Chemistry explains why more substituted double bonds are thermodynamically more stable.
The interaction occurs between the filled C-H sigma bond and the empty antibonding π orbital of the alkene (σ C-H → π*). Because highly substituted alkenes have more α-hydrogens, they enjoy greater delocalisation.
This is the underlying electronic basis for Saytzeff’s Rule (or Zaitsev’s Rule), which states that the more highly substituted alkene product is favoured in elimination reactions.
Influence on Heat of Hydrogenation
The stability provided by hyperconjugation is directly reflected in the heat of hydrogenation.
A more stable alkene has a lower potential energy and therefore releases less heat when it reacts with hydrogen to become an alkane.
Experimental data confirms that as the number of α-hydrogens increases (e.g., from ethylene to propene to 2-butene), the heat of hydrogenation per double bond decreases, illustrating the increased alkene stability provided by the effect.
Physical Consequences of Hyperconjugation
The effects of hyperconjugation are not merely theoretical; they can be measured through various physical properties of molecules.
Why is Staggered Ethane more stable?
For years, it was taught that the staggered conformation of ethane is more stable due to less steric hindrance. However, advanced computational chemistry suggests that Hyperconjugation is the primary reason.
In the staggered form, the filled σ C-H orbital is perfectly aligned with the empty σ* C-H orbital of the adjacent carbon, allowing for stabilising delocalisation that is not possible in the eclipsed form.
Bond Length Alterations
One of the most striking applications is explaining bond length variations. In molecules like propene, the C-C single bond adjacent to the double bond is roughly 1.46 Å, significantly shorter than the standard 1.54 Å single bond found in saturated hydrocarbons.
This is because hyperconjugation gives the single bond partial double-bond character. Conversely, the C=C double bond in such systems can be slightly longer than expected because its pi electrons are being delocalised away into the sigma framework.
Dipole Moment Changes
Since hyperconjugation involves the movement of electron density and can cause partial charge separation, it significantly affects the dipole moment of a molecule.
For example, propene has a measurable dipole moment whereas ethylene does not. This is attributed to the methyl group donating electron density toward the double bond via the hyperconjugation mechanism, creating a polarity that wouldn’t exist based on simple covalent bonding alone.
Advanced Concepts: Negative Hyperconjugation
While standard hyperconjugation involves the donation from a filled sigma orbital, negative hyperconjugation describes the opposite flow.
This occurs when electron density is donated from a filled π or p-orbital (such as a lone pair) into a nearby empty antibonding σ orbital.
This effect is particularly common when the σ orbital is located on highly electronegative atoms like fluorine or oxygen, as these low-lying antibonding orbitals are better energy matches for the filled orbitals.
Negative hyperconjugation can stabilise certain transition states and lengthen specific sigma bonds.
Conclusion: The Power of Sigma Delocalisation
The concept of Hyperconjugation in Organic Chemistry is an essential logical tool for explaining the subtle nuances of molecular structure and reactivity.
Whether it is justifying the stability of carbocations and free radicals, predicting the major product of an elimination reaction through Saytzeff’s Rule, or accounting for deviations in bond length and dipole moments, hyperconjugation provides the necessary explanation.
By moving beyond rigid bond representations and viewing electrons as delocalised waves, we gain a far more accurate and predictive understanding of the organic world.
Frequently Asked Questions
-
Why is hyperconjugation called no-bond resonance?
Hyperconjugation is termed no-bond resonance because, in its contributing canonical structures, there is no formal covalent bond between the α-carbon and the α-hydrogen. Although the σ-bond appears broken in these diagrams, the proton (H⁺) remains in close spatial proximity to the π-system or carbocation, allowing for effective electron delocalisation.
-
How does hyperconjugation in organic chemistry affect bond length?
Hyperconjugation leads to significant bond length alterations. It imparts partial double-bond character to adjacent single bonds, making them shorter than usual (e.g., in propene). Conversely, it can make existing double bonds slightly longer as the π-electron density is partially delocalised into the neighbouring sigma framework.
-
Is hyperconjugation a permanent or temporary electronic effect?
Hyperconjugation is a permanent electronic effect. It is an inherent structural property that exists in the molecule’s ground state. Unlike the electromeric effect, which requires an attacking reagent, hyperconjugation is always present and consistently influences the molecule’s stability and physical properties.
-
How do you count alpha (α) hydrogens for hyperconjugation?
To determine the extent of hyperconjugation, identify the carbon atoms directly attached to the unsaturated system or carbocation (the α-carbons). Count all hydrogen atoms bonded to these sp³ hybridised α-carbons. The more α-hydrogens present, the greater the number of hyperconjugating structures and the higher the molecular stability.

