The study of organic chemistry is essentially a study of the redistribution of shared or lone pairs of electrons.
While many electronic factors, such as the inductive effect, resonance, and hyperconjugation, govern the physical characteristics and chemical reactivity of molecules, the Electromeric Effect is unique in its dynamic nature.
It serves as a vital theoretical framework for understanding the “on-demand” shifts that occur during a chemical transformation.
Table of Contents
What is the Electromeric Effect?
Definition and Origins
The Electromeric Effect (denoted by the symbol ‘E’) is defined as the complete transfer of a shared pair of π-electrons from a multiple bond to one of the bonded atoms, typically the more electronegative one, in response to the requirement of an attacking reagent.
Historically, early organic chemists observed consistent behaviours in addition reactions to unsaturated compounds that could not be fully explained by existing theories, primarily the Inductive Effect.
While the Inductive Effect accounts for a permanent state of polarisation due to electronegativity differences, it was often found to be too weak to explain the dramatic and immediate electron redistributions observed at the moment of a reaction.
Consequently, the Electromeric Effect was conceptualised to describe how a molecule instantaneously redistributes its electron density to facilitate a reaction upon the approach of an external force.
The “On-Demand” Electron Shift
To visualise this temporary electronic effect, one might use the analogy of a sleeping dog.
When the dog hears the sound of a cat, it instantly wakes up and prepares to attack; without the sound, the dog remains in its resting state.
Similarly, an alkene or a carbonyl compound remains in its unpolarised ground state until an attacking reagent approaches, creating an electronic demand.
This “on-demand” shift is possible because π-electrons, compared to σ-electrons, are more loosely held and thus highly polarisable.
Visual Representation
In chemical shorthand, the Electromeric Effect is depicted using curved arrow notation.
These arrows represent the movement of electrons from their source (a multiple bond or a lone pair) to the electron-deficient site or atom that accepts them.
The tail of the arrow begins at the π-bond, and the head points toward the atom that acquires the pair of electrons, resulting in the development of unit positive and negative charges within the molecule.
Key Characteristics of the Electromeric Effect
A Temporary and Reversible Phenomenon
One of the most essential permanent vs temporary effects distinctions is that the Electromeric Effect is purely transient. It is not an inherent property of the molecule but is fully induced by the presence of an attacking reagent.
This phenomenon has been compared to a student giving an exam: a temporary uneasiness is observed only when the invigilator approaches, but the student returns to their normal state once the invigilator leaves.
Likewise, once the reagent is removed from the reaction environment, the polarised molecule reverts to its original, unpolarised state.
Requirement of Multiple Bonds
This effect is exclusively observed in organic compounds that contain at least one double or triple bond, such as alkenes, alkynes, and carbonyl groups.
Saturated hydrocarbons, such as alkanes, lack these loosely held π-electrons and therefore do not exhibit this specific type of polarisation.
Complete Transfer of Pi Electrons
Unlike the Inductive Effect, which involves only a slight or partial displacement of σ-electrons, the Electromeric Effect involves a pi-electron transfer of the entire shared pair.
This complete relocation of electrons leads to the development of distinct positive and negative poles, effectively turning the molecule into an “instant dipole”.
This high degree of polarisation increases the attraction between the substrate and the reagent, thereby favouring the formation of new chemical bonds.
Types of Electromeric Effects: +E and -E
The classification of this effect is based on the direction of electron transfer relative to the atom to which the attacking reagent eventually attaches.
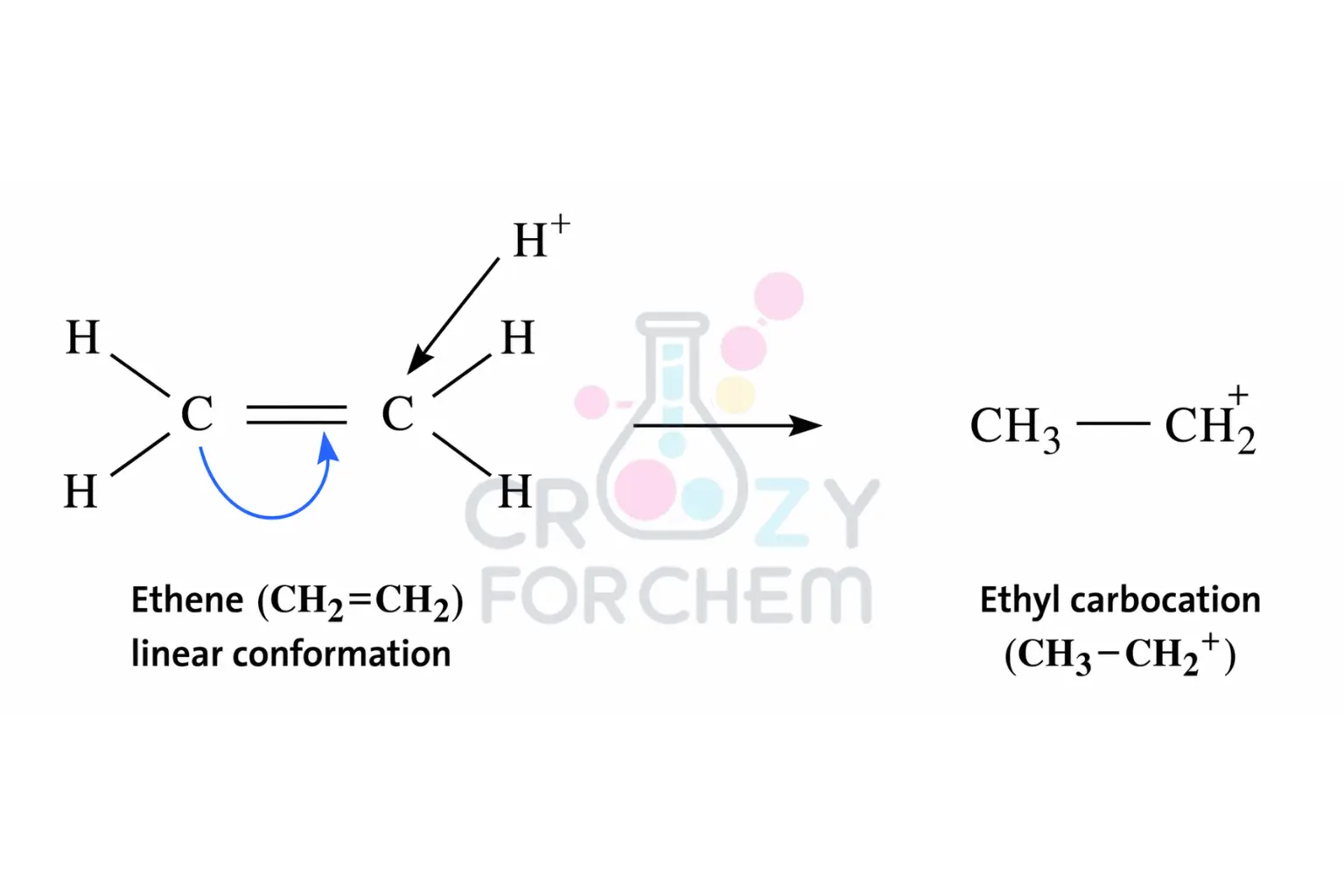
Positive Electromeric Effect (+E)
In a +E effect, the π-electrons of the multiple bond are transferred to the specific atom that the attacking reagent bonds with.
This is most commonly seen in electrophilic addition reactions. Because the reagent (the electrophile) is “electron-loving,” it attracts the electron pair toward the site where it is attacking.
A classic example is the protonation of ethene, where the electrons move toward the carbon atom that accepts the H⁺ ion.
Negative Electromeric Effect (-E)
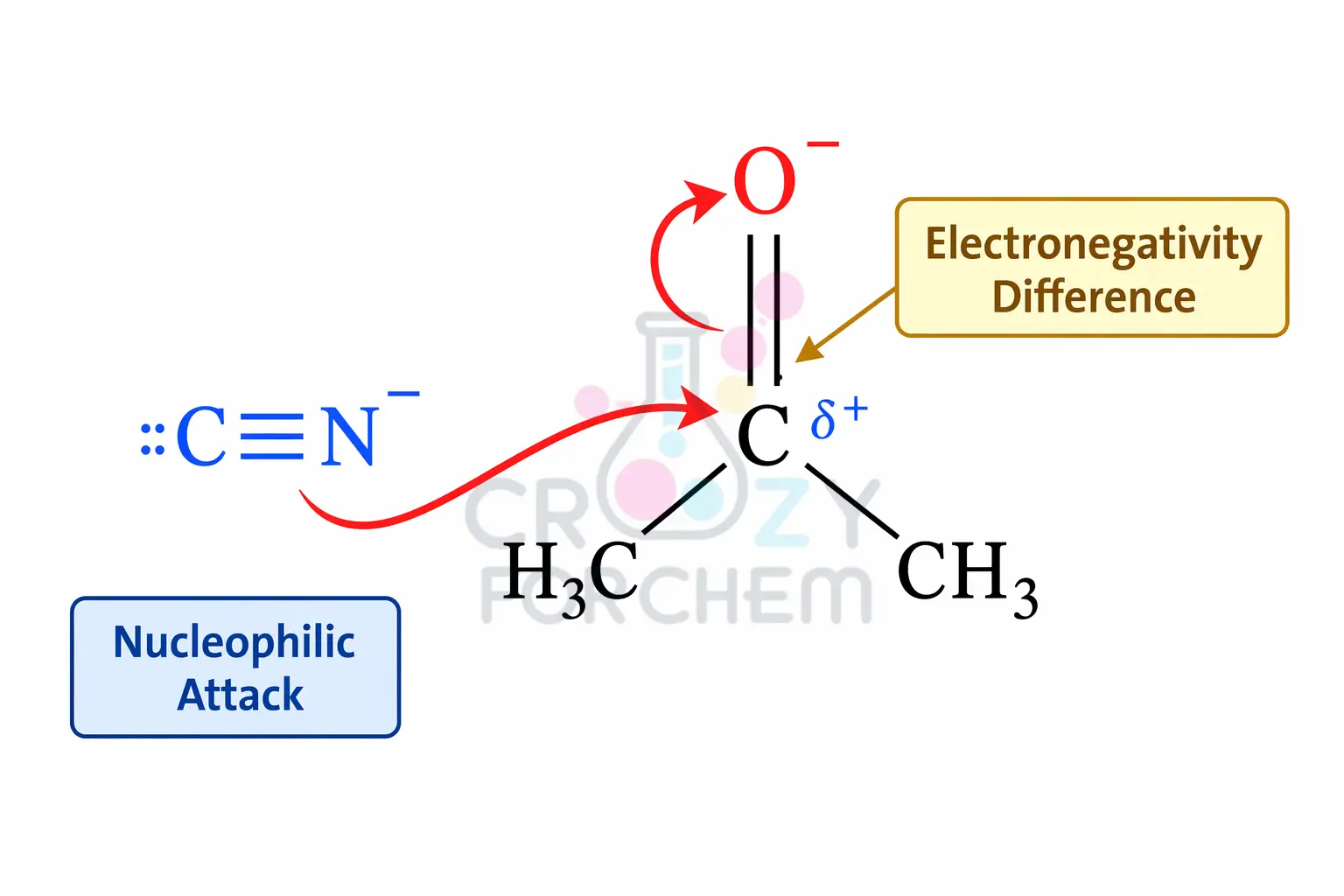
Conversely, the -E effect occurs when the π-electrons are transferred to an atom that the attacking reagent does not attach to. This effect is typical of a nucleophilic attack, particularly in carbonyl chemistry.
In such cases, the attacking reagent (the nucleophile) is “positive charge loving” and carries a negative charge or excess electrons. It repels the π-electron pair away from the site of attack.
For instance, during the addition of a cyanide ion (CN⁻) to a carbonyl group, the electrons shift toward the oxygen atom, while the cyanide bonds with the carbon.
Electromeric vs Inductive Effect
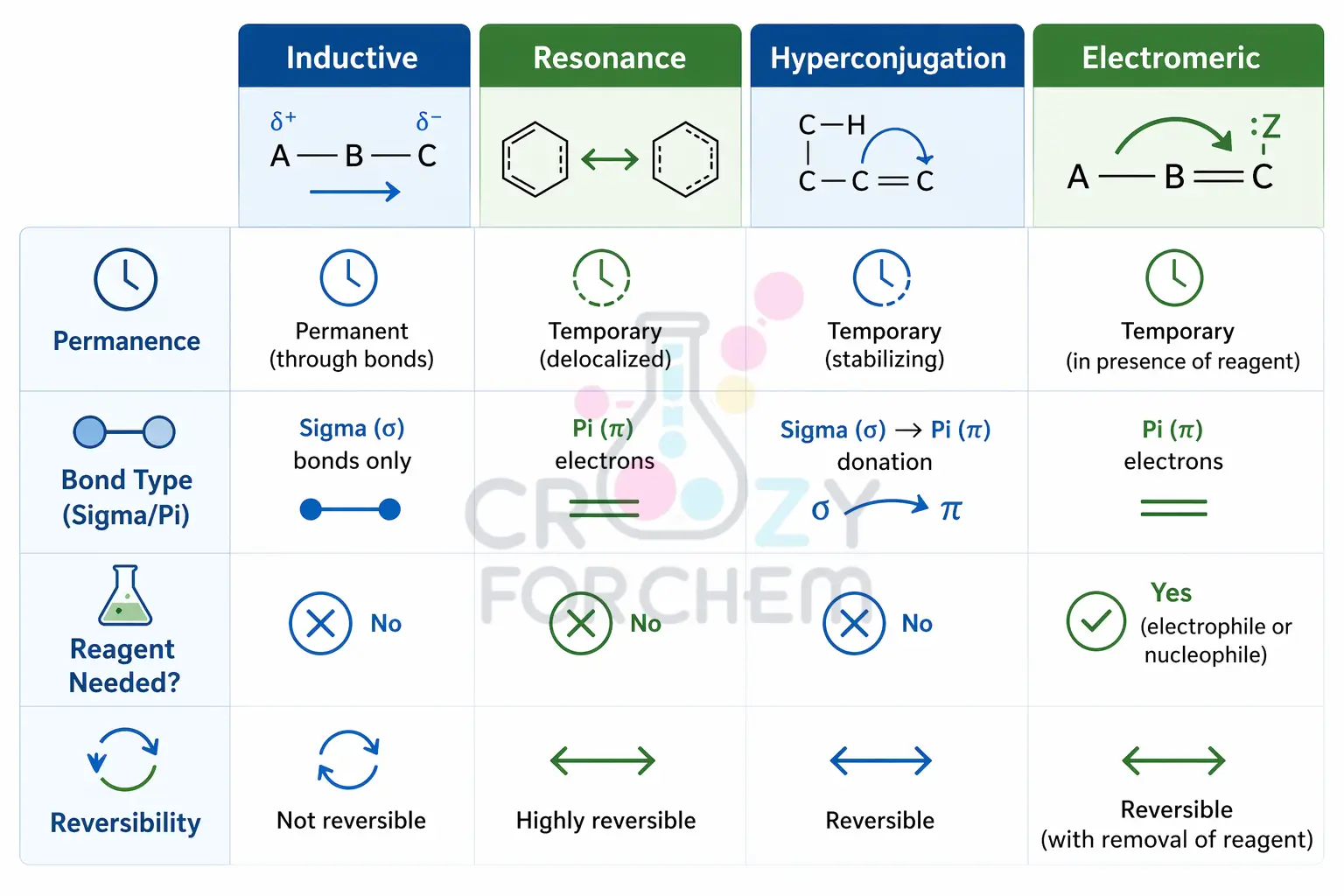
Permanent vs Temporary
The primary difference lies in their permanence: the Inductive Effect is a permanent feature inherent to the molecule’s structure, whereas the Electromeric Effect is a temporary response to a reagent. While the Inductive Effect is always “on,” the E-effect is only “on-demand”.
Bonding and Mechanism
The Inductive Effect involves the displacement of σ-electrons and is distance-dependent, usually fading after the second or third carbon atom in a chain.
In contrast, the Electromeric Effect involves the transfer of π-electrons and remains limited to the atoms involved in the multiple bond.
Strength and Predominance
Because it involves the complete transfer of a pair of electrons rather than a slight shift, the Electromeric Effect is significantly stronger than the Inductive Effect.
When both effects operate simultaneously in a reaction but act in opposite directions, the Electromeric Effect typically overrides the Inductive Effect. This makes it a decisive factor in predicting the site of attack and the stability of intermediate species.
Example of Competition: In the reaction of Vinyl Chloride with an electrophile, the Inductive effect (-I) of Chlorine pulls electrons away, but the Electromeric effect (+E) can push π-electrons towards the carbon.
In such cases, the Electromeric effect usually predominates because it involves a complete transfer of electrons rather than a mere shift.
| Feature | Mesomeric (Resonance) Effect | Electromeric (E) Effect |
| Nature | Permanent effect: It is an inherent property of the molecule. | Temporary effect: It occurs only on the demand of an attacking reagent. |
| Condition | Occurs in conjugated systems (alternating single/double bonds). | Occurs in any molecule with a multiple bond (double or triple). |
| Requirement | No external reagent is required for this effect to exist. | Strictly requires the presence of an attacking reagent to trigger. |
| Electron Type | Involves delocalisation of π-electrons or lone pairs. | Involves the complete transfer of a shared pair of π-electrons. |
| Reversibility | Static state; the molecule remains in resonance hybrid form. | Reversible; the molecule returns to its ground state if the reagent is removed. |
| Representation | Static state: the molecule remains in resonance hybrid form. | Shown by curved arrows indicating electron shift during a reaction. |
Practical Examples in Organic Mechanisms
Addition Reactions in Alkenes: The +E effect in hydrohalogenation
A standard illustration of the Electromeric Effect is the addition of HBr to an unsymmetrical alkene like propene. When the H⁺ electrophile approaches the double bond, it induces a +E effect.
The π-electrons shift toward the terminal carbon atom to facilitate the bond formation with H⁺, which results in the formation of a secondary carbocation.
According to Markovnikov’s Rule, the H⁺ adds to the carbon with more hydrogens, which the Electromeric Effect facilitates by guiding the electron shift toward the most stable intermediate.
Nucleophilic Addition in Carbonyls: The -E effect in carbon-oxygen bonds
Carbonyl compounds, such as acetone, possess an inherent partial polarity because oxygen is more electronegative than carbon. However, when a nucleophile like a cyanide ion (CN⁻) approaches, this polarity is greatly enhanced by the -E effect.
On the demand of the CN⁻ ion, the shared pair of π-electrons completely shifts to the oxygen atom. The CN⁻ ion then attaches to the now electron-deficient carbonyl carbon. This pi-electron displacement away from the carbon makes it highly susceptible to nucleophilic attack.
Real-World Applications and Industrial Importance
Predicting Regioselectivity
While the Electromeric Effect is a theoretical concept, its practical utility lies in predicting regioselectivity, the preference of a reaction to occur at one specific carbon atom over another.
This is a cornerstone of the petrochemical industry. For example, in the industrial production of alcohols, when water is added to propene in the presence of an acid catalyst, the E-effect helps predict that the hydroxyl group will attach to the central carbon to yield propan-2-ol rather than propan-1-ol.
Explaining Markovnikov’s Rule
The Electromeric Effect provides a mechanistic explanation for Markovnikov’s Rule. By identifying which electron shift leads to the more stable carbocation intermediate (such as a tertiary carbocation over a primary one), chemists can rationalise why specific products are formed in high yields during electrophilic additions.
Synthesis of Pharmaceuticals and Polymers
Precise control over product formation is vital for designing efficient synthesis pathways for pharmaceuticals and polymers.
By understanding how π-electrons shift upon a reagent’s approach, industrial chemists can ensure that the desired product is formed with minimal unwanted byproducts, thus making processes more cost-effective.
Conclusion: Mastering Reaction Dynamics through the E-Effect
The Electromeric Effect remains an indispensable tool for understanding the dynamic nature of organic reactions.
By characterising it as a temporary electronic effect driven by the needs of an attacking reagent, we can accurately describe transformations that static models cannot.
Whether it is predicting the regioselectivity of an alkene addition or the path of a nucleophilic attack on a carbonyl, the ability to trace pi-electron transfer through curved arrows allows chemists to master the underlying mechanisms of molecular transformation.
Through examples of the electromagnetic effect, students and professionals alike gain the insight needed to navigate the complex world of organic synthesis with precision.
Frequently Asked Questions
-
Why is the electromeric effect considered a temporary effect?
The electromeric effect is a temporary phenomenon because it only occurs when a molecule with a multiple bond is approached by an attacking reagent (an electrophile or nucleophile). The polarisation is an “on-demand” response; as soon as the attacking reagent is removed, the π-electrons return to their original positions, and the molecule reverts to its unpolarised ground state.
-
What is the difference between +E and -E effects?
The distinction lies in the direction of the electron shift relative to the attacking site:
Positive Electromeric (+E) Effect: The π-electrons transfer to the atom where the attacking reagent eventually bonds (e.g., H⁺ addition to alkenes).
Negative Electromeric (-E) Effect: The π-electrons transfer away from the atom where the reagent bonds (e.g.,CN⁻ addition to a carbonyl group). -
Which is stronger: the inductive effect or the electromeric effect?
In a chemical reaction where both effects operate simultaneously but in opposite directions, the electromeric effect typically outweighs the inductive effect. This is because the electromeric effect involves the complete transfer of a shared pair of π-electrons, creating full charges, whereas the inductive effect only involves a partial displacement of σ-electrons.
-
Can the electromeric effect occur in alkanes?
No, the electromeric effect cannot occur in saturated hydrocarbons like alkanes. This effect strictly requires the presence of a multiple bond (double or triple bond) containing loosely held π-electrons that are capable of being displaced to an adjacent atom.
-
How does the electromeric effect influence Markovnikov’s Rule?
The electromeric effect provides the mechanistic basis for Markovnikov’s Rule. In the presence of an electrophile (H⁺), it facilitates the shift of π-electrons toward the specific carbon atom that results in the formation of the most stable carbocation intermediate, thereby determining the regioselectivity of the reaction

