Chemical reactions are the fundamental engines of change in the universe, driving everything from the metabolic processes in our bodies to the industrial production of modern materials.
To master the fundamentals of Chemistry, it is essential to understand the various types of chemical reactions, as they explain how substances transform in the world around us.
At its most basic level, a chemical reaction is a process where the chemical bonds within reactant molecules are broken, and new bonds are formed to create product molecules, resulting in the formation of a new substance.
One of the most critical concepts for any chemistry student to understand is that while substances change, atoms themselves do not.
In a classic chemical reaction, atoms are simply rearranged to form new substances with new properties, but no atom is ever destroyed or created, adhering to the fundamental Law of Conservation of Mass.
Understanding these transformations allows us to categorise the millions of possible chemical interactions into a few manageable types.
Table of Contents
What is a Chemical Reaction? (The Basics)
Before diving into specific categories, one must grasp the core components of a chemical equation. A chemical equation serves as a mathematical and symbolic representation of a reaction, showing how reactants transform into products.
Reactants vs Products
In any chemical process, we start with specific substances known as reactants. These are the starting materials that undergo a chemical change. Once the reaction is complete, the resulting substances are called products.
In a written equation, reactants are placed on the left-hand side, while products appear on the right-hand side, separated by an arrow that signifies the direction of the reaction.
Signs of a Reaction
Chemical reactions are typically accompanied by detectable physical changes that indicate a transformation has occurred. Common signs include:
- Colour change: For instance, when green ferrous sulphate crystals are heated, they change colour as they decompose into ferric oxide.
- Temperature change: Some reactions release heat (exothermic), while others absorb it (endothermic), often making the reaction vessel feel significantly hotter or colder.
- Evolution of gas: The formation of bubbles or a distinct smell often signals that a gas is being produced.
- Formation of a precipitate: This occurs when two clear solutions are mixed and produce an insoluble solid that settles at the bottom.
Quick Reference: Chemical Reaction Summary Table
The following table provides a quick reference to help you categorise the primary types of chemical reactions discussed in this guide.
| Reaction Type | Reactants | Products | Key Feature |
|---|---|---|---|
| Combination | 2 or more | 1 single | Synthesis |
| Decomposition | 1 single | 2 or more | Breakdown |
| Displacement | Element + Compound | Compound + Element | Reactivity based |
| Neutralisation | Acid + Base | Salt + Water | pH balance |
The 7 Essential Types of Chemical Reactions.
1. Combination Reaction
A combination reaction, also known as a synthesis reaction, occurs when two or more reactants interact to form a single, more complex product. This type of reaction is essentially a “building” process where simpler substances merge into one.
- General Formula: A + B → AB.
- Key Characteristics: These reactions are often exothermic because forming new chemical bonds generally releases energy into the surroundings.
- Example: A classic demonstration involves Magnesium ribbon burning in the presence of Oxygen. When ignited, the magnesium combines with oxygen from the air to produce a dazzling white flame and a white powdery substance known as Magnesium Oxide.
- Equation: 2Mg(s) + O₂(g) → 2MgO(s)
2. Decomposition Reaction
A decomposition reaction is the functional opposite of a combination reaction. In this process, a single compound breaks down into two or more simpler substances.
Because breaking chemical bonds requires energy, most decomposition reactions are endothermic and require an input of heat, light, or electricity to proceed.
- General Formula: AB → A + B.
- Example: The Limestone Reaction: A vital industrial example is the thermal decomposition of Calcium Carbonate (Limestone). When limestone is heated, it breaks down into Calcium Oxide (Quicklime) and Carbon Dioxide gas. Calcium oxide is a primary ingredient in the production of cement.
- Equation: CaCO₃(s) →[Δ] CaO(s) + CO₂(g)
3. Single Displacement (Replacement) Reaction
In a single displacement reaction, a more reactive element displaces a less reactive element from its compound. This reaction is governed by the “Reactivity Series,” which dictates that a stronger, more “aggressive” element will essentially kick out a weaker one to take its place in a molecule.
- General Formula: A + BC → AC + B.
- Example: When Zinc metal reacts with Hydrochloric Acid, the zinc displaces the hydrogen because zinc is more reactive. This results in the formation of aqueous zinc chloride and the release of hydrogen gas bubbles.
- Equation: Zn(s) + 2HCl(aq) → ZnCl₂(aq) + H₂(g)
4. Double Displacement Reaction
A double displacement reaction, sometimes called a metathesis reaction, involves two compounds exchanging ions to produce two entirely new compounds. Think of this as a “partner swap” between two ionic substances usually in an aqueous solution.
- General Formula: AB + CD → AD + CB.
- Example: Barium Sulphate Precipitate: One of the most visually clear examples occurs when Sodium Sulphate solution is mixed with Barium Chloride solution. The barium ions and sodium ions swap partners, resulting in the formation of Barium Sulphate, which is an insoluble white solid (precipitate).
- Equation: Na₂SO₄(aq) + BaCl₂(aq) → BaSO₄(s) + 2NaCl(aq)
5. Combustion Reaction
A combustion reaction occurs when a substance reacts rapidly with Oxygen (O₂), releasing a significant amount of energy in the form of heat and light. This is what we commonly refer to as “burning”.
- Key Fact: When hydrocarbons (compounds containing only carbon and hydrogen) undergo combustion, the products are always Carbon Dioxide (CO₂) and Water (H₂O).
- Example: The burning of Methane gas, which is the primary component of natural gas, provides heat for cooking and heating.
- Equation: CH₄(g) + 2O₂(g) → CO₂(g) + 2H₂O(g) + Energy.
6. Oxidation-Reduction (Redox) Reactions
A Redox reaction is a process involving the transfer of electrons between chemical species. It consists of two simultaneous processes: Oxidation, where a substance loses electrons, and Reduction, where a substance gains electrons.
- Example: The rusting of iron is a slow redox reaction where iron reacts with oxygen and moisture to form iron oxide. Another example is the reaction between Zinc and Copper Sulphate, where Zinc is oxidised, and Copper ions are reduced to solid copper.
- Equation: Zn(s) + CuSO₄(aq) → ZnSO₄(aq) + Cu(s)
7. Neutralisation (Acid-Base) Reaction
A neutralisation reaction is a specific type of double displacement reaction that occurs between an acid and a base. The primary outcome is the formation of a Salt and Water, which effectively “neutralises” the acidic and basic properties, aiming for a pH balance.
Equation: HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
General Formula: Acid + Base → Salt + Water.
Example: The reaction between Hydrochloric Acid (HCl) and Sodium Hydroxide (NaOH) results in the formation of common table salt (Sodium Chloride) and water.
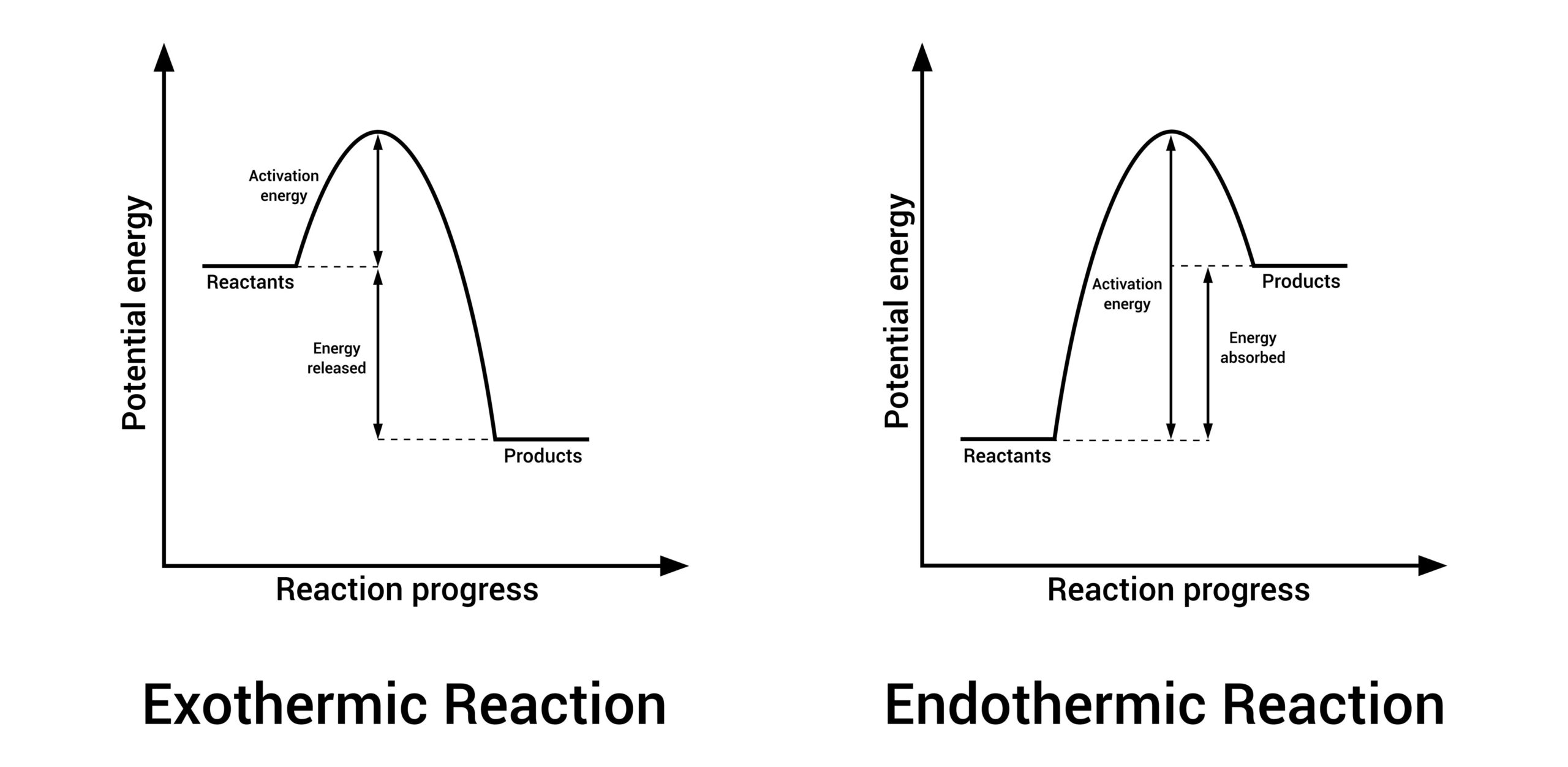
Exothermic vs Endothermic Reactions
Every chemical reaction involves an exchange of energy with the surrounding environment.
- Exothermic Reactions: These reactions release energy, usually as heat or light. Examples include combustion, neutralisation, and many combination reactions. If you touch a test tube during an exothermic reaction, it will feel warm or hot.
- Endothermic Reactions: These reactions absorb energy from the surroundings. Most decomposition reactions, such as the breakdown of limestone, require a constant supply of heat to continue. These reactions can make the environment feel colder.
Special Category: Precipitation Reactions
While often classified under double displacement, precipitation reactions specifically focus on the formation of an insoluble solid from the mixture of two soluble aqueous solutions. The insoluble solid is called the precipitate. These are crucial in laboratory chemistry for identifying specific ions present in a solution.
How to Identify Reaction Types? (Quick Cheat Sheet)
Identifying different types of chemical reactions becomes much simpler once you learn to look for specific patterns in the reactants and products.
- Count the Reactants and Products: If you see two or more reactants turning into one single product, it is a Combination reaction.
- Look for Breakdown: If one single reactant splits into multiple products, it is a Decomposition reaction.
- Search for Oxygen: If Oxygen (O₂) is a reactant and heat is produced, you are likely looking at a Combustion reaction.
- Check for “Partner Swapping”: If you see two compounds and their ions appear to have exchanged places, it is a Double Displacement reaction.
Real-Life Examples of Chemical Reactions
Chemical reactions are not just for the lab; they are the basis of life:
- Photosynthesis: Plants use a complex series of reactions to convert carbon dioxide and water into glucose and oxygen using sunlight.
- Respiration: The process by which our bodies break down glucose to release energy is a form of redox and decomposition reaction.
- Rusting: The oxidation of iron structures like bridges and railings over time.
- Digestion: The breakdown of complex food molecules into simpler nutrients in our stomach is a series of decomposition reactions facilitated by enzymes.
Conclusion
Mastering these 7 types of chemical reactions is essential for any student looking to excel in chemistry.
Understanding these patterns does not just help with passing exams; it provides the tools needed for balancing complex equations and predicting the products of unknown reactions based on the properties of the reactants.
Whether it is the synthesis of new medicines or the neutralisation of acidic soil, these reactions are the building blocks of our scientific understanding.
Frequently Asked Questions
-
What are the 5 main types of chemical reactions?
Answer: In Chemistry, there are five primary categories: Combination, Decomposition, Single Displacement, Double Displacement, and Combustion. Additionally, Redox and Neutralisation reactions are also essential patterns that often fall within these categories.
-
How do you identify a redox reaction?
Answer: The easiest way to identify a redox reaction is by looking for changes in Oxidation Numbers. If one element loses electrons (Oxidation) and another gains them (Reduction), it is a redox reaction. A helpful mnemonic to remember this is OIL RIG: Oxidation Is Loss, Reduction Is Gain.
-
Is neutralisation a double displacement reaction?
Answer: Yes, neutralisation is a specific type of double displacement reaction. In this process, the H+ ions from the acid and the OH- ions from the base “swap partners” to form water, H₂O and a salt.
-
What is the difference between exothermic and endothermic reactions?
Answer: Exothermic reactions release heat into the surroundings (such as combustion), which often makes the reaction vessel feel warm. Endothermic reactions absorb energy from the surroundings (such as thermal decomposition), requiring a constant external energy source to proceed.
-
Can a reaction be both a combination and a redox?
Answer: Absolutely! Many combination reactions are also redox reactions. For instance, when Magnesium reacts with Oxygen to form Magnesium Oxide, the Magnesium is oxidised while the Oxygen is reduced.
-
Why is the burning of methane classified as a combustion reaction?
Answer: The burning of methane is a combustion reaction because it reacts rapidly with Oxygen (O₂), releasing a significant amount of heat and light (energy) while producing Carbon Dioxide and water vapour.

