Acids and bases are fundamental chemical opposites that influence nearly every aspect of our world, from large-scale industrial manufacturing to the delicate biological systems that keep us alive.
The word “acid” finds its roots in the Latin term acere or acidus, which literally translates to “sour,” a characteristic that has defined these substances for centuries.
In modern chemistry, understanding the interactions between these substances is the key to mastering chemical reactions and industrial engineering
Table of Contents
What are Acids and Bases? (Fundamentals)
To grasp the nature of these substances, one must first understand the concept of ionisation. When certain compounds dissolve in water, they break down into charged particles called ions.
The Concept of Ionisation
An ion is an electrically charged atom or molecule that forms when a neutral substance dissociates in a solvent. In the context of acids and bases, this process determines whether a solution is acidic or alkaline based on the specific ions released.
- Acids: These are substances that act as H⁺ emitters, releasing hydrogen ions (H⁺) or hydronium ions (H₃O⁺) when in solution. Because a bare proton cannot survive alone in water, it quickly attaches to a water molecule to form the hydronium ion (H₃O⁺). The hydronium ion is the standard representation of an aqueous acid’s strength.
- Bases: These are defined as substances that accept hydrogen ions or release hydroxide ions (OH⁻) into a solution. They are the chemical opposites of acids.
- Alkalis: It is a common mistake to use the terms “base” and “alkali” interchangeably. A crucial distinction is that an alkali is specifically a base that is soluble in water.
Core Properties of Acids and Bases
Physical Properties
We interact with acids and bases daily through our senses.
- Taste: Acids are famously sour, as experienced in lemons (citric acid) or vinegar (acetic acid). Conversely, bases have a distinctly bitter taste, such as that found in baking soda.
- Texture: Bases are characterised by a slippery or soapy feel. This occurs because bases react with the oils on your skin to turn them into small amounts of soap.
Chemical Properties
- Conductivity: Both acids and bases act as electrolytes. When dissolved in water, they produce mobile ions that allow the solution to conduct electricity.
- Corrosiveness: Strong versions of both substances are highly corrosive and can cause severe damage to organic tissues and materials like metal or stone.
- Indicators: The most common way to test these substances in a lab is using the litmus test. Acids turn blue litmus paper red, while bases turn red litmus paper blue.
Beyond litmus paper, chemists use a variety of indicators to detect pH levels. Synthetic indicators like Phenolphthalein turn vivid pink in basic solutions while remaining colourless in acids. Meanwhile, natural indicators such as turmeric or red cabbage juice can also be used; for instance, turmeric changes from yellow to deep red when exposed to an alkaline substance.
For more on how these properties lead to different reactions, see our guide on Types of Chemical Reactions.
Three Major Theories of Acids and Bases
Over time, our scientific understanding of acids and bases has evolved through three primary frameworks.
1. Arrhenius Theory (1883)
Proposed by Svante Arrhenius, this theory is the most restrictive and focuses entirely on water-based (aqueous) solutions. He defined acids as substances that increase the H⁺ concentration and bases as those that increase OH⁻ concentration.
2. Brønsted-Lowry Theory (1923)
This theory expanded the definition to any solvent. It defines acids as proton donors and bases as proton acceptors. This theory introduced Conjugate Acid-Base Pairs; when an acid like HCl loses a proton, it becomes its conjugate base, Cl⁻. (Since a neutral Hydrogen atom consists of only one proton and one electron, losing that electron to become H⁺ leaves behind nothing but a bare nucleus; hence, H⁺ is chemically referred to as a proton.)
3. Lewis Theory (1923)
The broadest and most “expert level” definition was provided by Gilbert Lewis. He looked at electron-pair transfer rather than protons. A Lewis acid is an electron-pair acceptor, while a Lewis base is an electron-pair donor.
Comparison Table (The Featured Snippet Magnet)
| Theory | Acid Definition | Base Definition | Scientific Focus |
| Arrhenius | H⁺ Producer | (OH⁻) Producer | Aqueous (Water) Solutions |
| Brønsted-Lowry | Proton (H⁺) Donor | Proton (H⁺) Acceptor | Proton Transfer |
| Lewis | Electron-pair Acceptor | Electron-pair Donor | Electron Transfer |
The Amphoteric Nature of Water
It is fascinating to note that some substances can act as both an acid and a base, depending on the reaction environment. Water (H₂O) is the most common example of such an amphoteric substance; it can donate a proton to become a hydroxide ion (OH⁻) or accept one to form a hydronium ion (H₃O⁺).
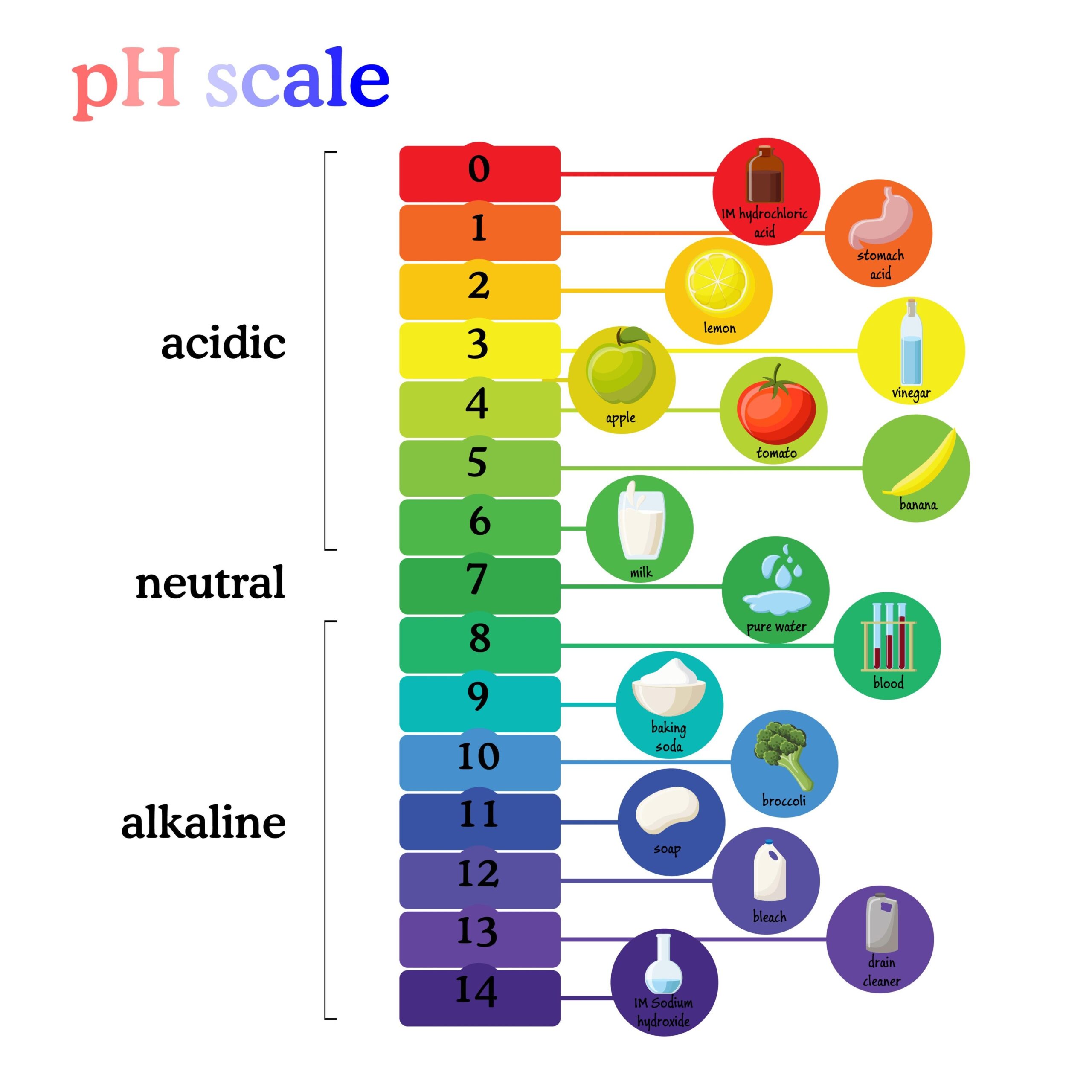
The pH Scale: Measuring Acidity and Alkalinity
The pH scale measures the “potential of hydrogen” in a solution. It is a logarithmic scale, meaning each unit represents a tenfold (10x) difference in hydrogen ion concentration.
- pH < 7 (Acidic): Solutions like gastric acid (pH 1.5–2.0) fall here.
- pH 7 (Neutral): Pure distilled water is the point of neutrality.
- pH > 7 (Basic/Alkaline): Household ammonia (pH 11.0–11.5) is a common example of a base.
Strong vs Weak Acids and Bases
The “strength” of these chemicals refers to their dissociation percentage in water.
- Strong Acids/Bases: These ionise or dissociate 100% in water. Common examples include Hydrochloric Acid (HCl) and Sodium Hydroxide (NaOH).
- Weak Acids/Bases: These only partially ionise, often less than 5%. Common examples include Acetic Acid (vinegar) and Ammonia.
- Constants (Ka and Kb): Scientists use the Acid Dissociation Constant (Ka) and Base Dissociation Constant (Kb) to provide a numeric value for how strong an acid or base is.
Learn the specifics of these constants in our Deep Dive into Strong vs Weak Acids.
Neutralisation: When Opposites React
When an acid and a base meet, they undergo a neutralisation reaction. The standard formula is: Acid + Base →Salt + Water.
Everyday Examples:
- Antacids: These are bases that neutralise excess hydrochloric acid in your stomach to relieve indigestion.
- Agriculture: Farmers use lime (calcium hydroxide) to neutralise acidic soil, ensuring optimal conditions for crop growth.
Acids and Bases in Living Organisms
Your body is a complex chemical factory that relies on precise pH levels.
- Digestion: The stomach uses strong hydrochloric acid to activate the enzyme pepsin, which breaks down proteins. To prevent this acid from damaging the small intestine, the pancreas secretes alkaline substances to neutralise it.
- Blood pH: For survival, human blood must be maintained at a very narrow range between 7.35 and 7.45. The body uses sophisticated buffer systems to resist changes in this pH.
Safety Guidelines and Best Practices
Working with acids and bases requires extreme caution.
- The Golden Rule: The “AA” rule stands for Always Add Acid to water. Never add water to concentrated acid, as it can cause a violent exothermic reaction and spattering.
- PPE: Always wear Personal Protective Equipment, including safety goggles and chemical-resistant gloves, especially when handling concentrated substances.
Conclusion
From the sour taste of a lemon to the vital regulation of your blood pH, acids and bases are foundational to both chemistry and life. Mastering their properties, theories, and reactions is essential for any science student aiming to understand how the world functions at a molecular level.
Ready to test your knowledge? Try our [Acids and Bases Practice Quiz] or explore our next pillar post on Chemical Equations!
Frequently Asked Questions
-
What are 5 examples of acids and bases?
Answer: Common examples of acids include Hydrochloric Acid (stomach acid), Acetic Acid (vinegar), Citric Acid (lemons), Sulphuric Acid (car batteries), and Carbonic Acid (fizzy drinks). Common bases include Sodium Hydroxide (drain cleaner), Magnesium Hydroxide (antacids), Sodium Bicarbonate (baking soda), Ammonia (cleaning products), and Calcium Hydroxide (lime).
-
Why does an acid-base reaction produce water?
Answer: In a neutralisation reaction, the hydrogen ion (
OH⁻) from the acid combines with the hydroxide ion (OH⁻) from the base. Chemically, H⁺ +OH⁻rightarrow H_2O$. This combination results in the formation of water, which effectively neutralises the corrosive properties of both reactants. -
Is milk an acid or a base?
Answer: Milk is slightly acidic, with a pH typically ranging from 6.5 to 6.7. This acidity is primarily due to the presence of lactic acid. However, it is very close to neutral (pH 7) compared to strong acids like lemon juice or vinegar.
-
How can you tell if a substance is a strong or weak base?
Answer: The strength of a base is determined by its degree of ionisation in water. A strong base, like Potassium Hydroxide (KOH), dissociates completely (100%) into ions. A weak base, such as Ammonia (NH₃), only partially reacts with water, leaving most of its molecules intact.
-
What happens when you mix a strong acid with a strong base?
Answer: Mixing a strong acid and a strong base results in a vigorous neutralisation reaction. The process releases significant energy (exothermic) and produces a neutral salt and water. For example, mixing HCl and NaOH produces Sodium Chloride (table salt) and water, usually resulting in a solution with a pH of 7.
-
What is the main difference between the Arrhenius and Brønsted-Lowry theories?
Answer: The primary difference lies in their scope. The Arrhenius theory is restricted to aqueous (water) solutions and defines bases only as
OH⁻producers. The Brønsted-Lowry theory is broader; it applies to any solvent and defines bases as proton (H⁺) acceptors, even if they do not contain hydroxide ions.

