In the complex landscape of organic chemistry, the reactivity and stability of molecules are governed by the dynamic movement of electrons, known collectively as electronic displacement effects.
For students preparing for rigorous examinations like the JEE Main or NEET, the fundamental “tug-of-war” between various electronic forces is the key to unlocking reaction mechanisms.
This Inductive vs Mesomeric effect guide provides an exhaustive comparison between the Inductive Effect and the Mesomeric Effect, exploring their mechanisms, ranges, and the crucial hierarchy that determines which effect dominates in a given molecular scenario.
Table of Contents
Understanding Electronic Displacement Effects in Organic Chemistry
The Source of Reactivity
Reactivity in organic molecules stems from an imbalance in electron density. While saturated hydrocarbons are often non-reactive due to the equal sharing of electrons in non-polar C-C and C-H bonds, the introduction of electronegative elements or multiple bonds creates polarity.
This partial polarity leads to electron transfer among atoms, creating electron-rich and electron-deficient sites that attract nucleophiles and electrophiles, respectively.
Electronic effects serve as the “silent directors” that stabilise molecules, influence acidity or basicity, and dictate the most likely sites for chemical attack.
Sigma vs Pi Electrons
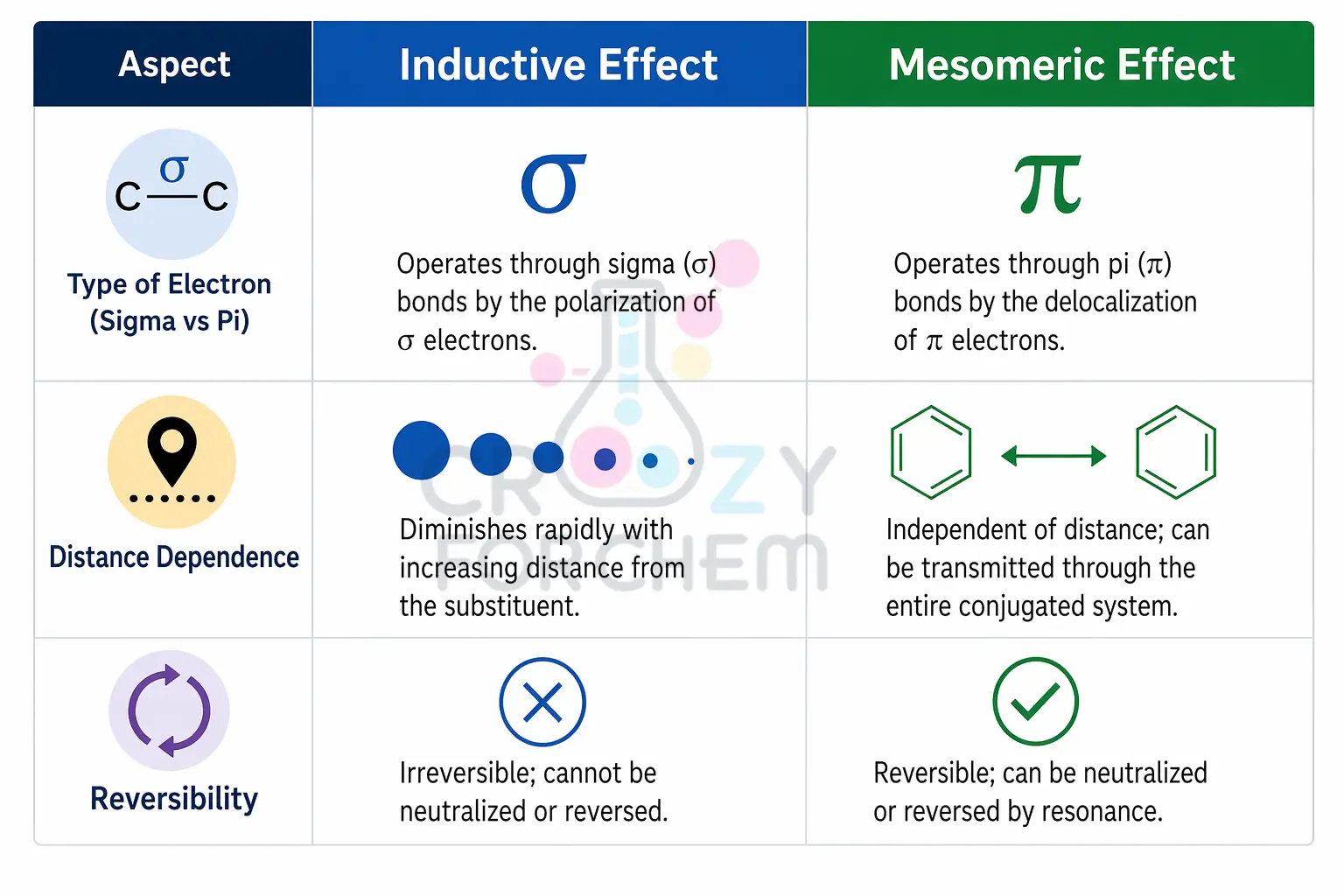
The fundamental distinction between electronic effects lies in the type of electrons involved. The Inductive effect is characterised by the polarisation of sigma (σ) electrons, which are tightly held within single bonds.
In contrast, the Mesomeric effect (often referred to as the Resonance effect) involves the delocalisation or “depolarisation” of pi (π) electrons or lone pairs within a conjugated system.
The Inductive Effect (I-Effect) at a Glance
Mechanism: The Molecular Tug-of-War
The Inductive effect is a permanent state of polarisation caused by differences in electronegativity between bonded atoms.
When two unlike atoms form a σ-bond, the electron density is not uniform; it is more concentrated towards the more electronegative atom.
This creates a permanent dipole where one atom acquires a partial negative charge (δ-) and the other a partial positive charge (δ+).
Classification of Inductive Effects:
- Negative Inductive Effect (-I): Caused by electron-withdrawing groups (EWG) like nitro (-NO₂), halogens (-F, -Cl, -Br, -I), cyano (-CN), and carboxy (-COOH) groups.
- Positive Inductive Effect (+I): Caused by electron-donating or releasing groups (EDG), primarily alkyl groups (-CH₃, -C₂H₅) and metals.
Distance Dependency: The Decay Rule
The most defining feature of the Inductive effect is that it is a distance-dependent phenomenon.
The positive or negative charge induced at the source is relayed to adjacent atoms, but the “pull” or “push” weakens significantly as it travels down the carbon chain.
Generally, the effect vanishes after the second carbon atom and is considered negligible after the third carbon (C₃).
The Mesomeric Effect (M-Effect) at a Glance
Mechanism: Electron Delocalisation
The Mesomeric effect involves the permanent transfer of π-electrons or lone pairs through a conjugated system.
This effect arises from interactions between π-bonds or between a π-bond and a lone pair on an adjacent atom, leading to a redistribution of electron density that cannot be explained by a single Lewis structure.
Instead, the molecule exists as a resonance hybrid, which is a composite of various theoretical resonance structures.
Types of Mesomeric Effects:
- Positive Mesomeric Effect (+M): Occurs when a substituent group donates electrons to the conjugated system through resonance (e.g., -OH, -NH₂ -OR, -NHR). These groups typically have lone pairs that increase the electron density of the system.
- Negative Mesomeric Effect (-M): Occurs when a group withdraws electrons from the conjugated system through resonance (e.g., -NO₂ -CN, -CHO, -COOH, -SO₃H).
Distance Independency: The Long-Range Signal
Unlike the Inductive effect, the Mesomeric effect is distance-independent within the confines of a conjugated system.
As long as the conjugation (alternating multiple bonds or p-orbitals) is maintained, the polarisation is transmitted throughout the entire unsaturated chain without diminishing.
Direct Comparison: Inductive vs Mesomeric Effect
| Feature | Inductive Effect | Mesomeric Effect |
|---|---|---|
| Electron Type | Sigma (σ) electrons | Pi (π) electrons / Lone pairs |
| Transmission | Through σ-bonds down the bond axis | Through conjugated π-systems |
| Polarisation | Partial polarisation; slight shift | Whole/Total polarisation; complete transfer |
| Distance | Distance-dependent; dies after 3 carbons | Distance-independent in conjugation |
| Nature | Permanent | Permanent |
| Alias | Polarisation effect | Polarisability / Resonance effect |
The Hierarchy of Effects: Which One Dominates?
Resonance Beats Induction
When a molecule is subjected to both Inductive and Mesomeric effects, a clear hierarchy dictates the outcome.
In almost all chemical contexts, the Mesomeric effect is more predominant and dominating than the Inductive effect.
This is because the complete delocalisation of π-electrons provides far greater stabilisation or destabilisation than the mere partial displacement of σ-electrons.
Evidence from Acidity and Basicity
The dominance of resonance is best illustrated through acid strength. For example, p-nitrophenol is more acidic than m-nitrophenol.
Although the nitro group (-NO₂) is closer to the hydroxyl group in the meta position (stronger -I effect), the para position allows for an additional electron-withdrawing mesomeric effect (-M) that delocalises the negative charge of the phenoxide ion over the nitro group’s oxygen.
Since this resonance delocalisation is impossible in the meta position, the para isomer is more stable and thus more acidic, proving that the resonance effect overpowers the distance-based inductive effect.
Practical Applications in JEE and NEET Mechanisms
Stabilising Carbocations
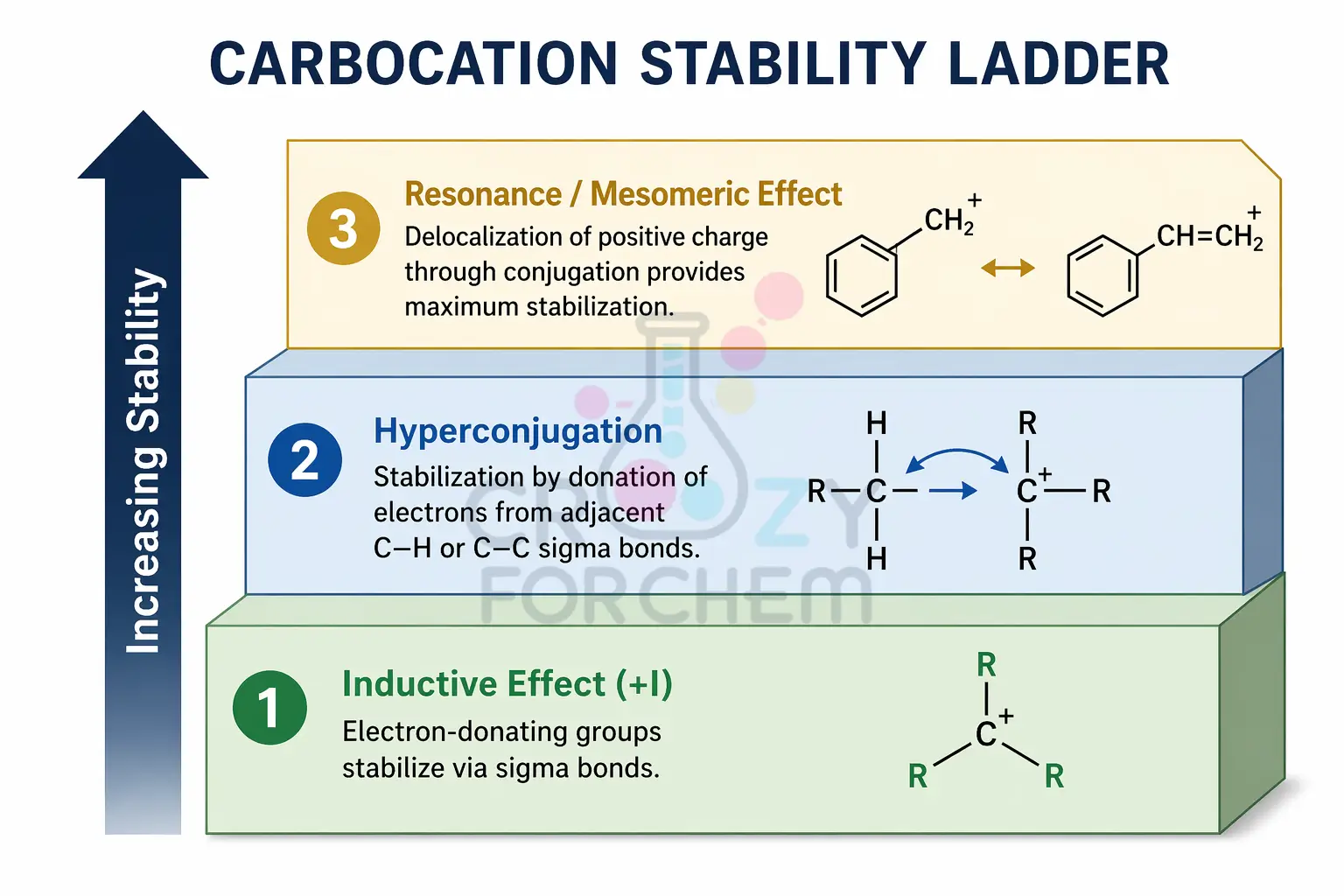
The stability of reaction intermediates, specifically carbocations, is determined by these electronic factors. A carbocation is stabilised by electron-donating groups that reduce the charge disparity.
- Mesomeric Stabilisation: As resonance increases, the stability of a carbocation rises because the electron deficiency is spread across multiple atoms. This is the most effective stabilising force.
- Inductive Stabilisation: Alkyl groups provide stability through a +I effect. This explains the stability order: Tertiary (3°) > Secondary (2°) > Primary (1°).
- Strength Order: Resonance > Hyperconjugation > Inductive Effect.
Directing Effects in Benzene
Electronic effects regulate regioselectivity in electrophilic aromatic substitutions.
- Activating Groups (+M/+I): Groups like methyl (-CH₃, +I) or hydroxyl (-OH, +M) increase the electron density of the benzene ring, making it more reactive and directing incoming electrophiles to the ortho and para positions.
- Deactivating Groups (-M/-I): Groups like nitro (-NO₂) or cyano (-CN) withdraw electron density, making the ring less reactive and directing electrophiles to the meta position.
Exceptions to the Rule: The Halogen Paradox
When -I Outweighs +M
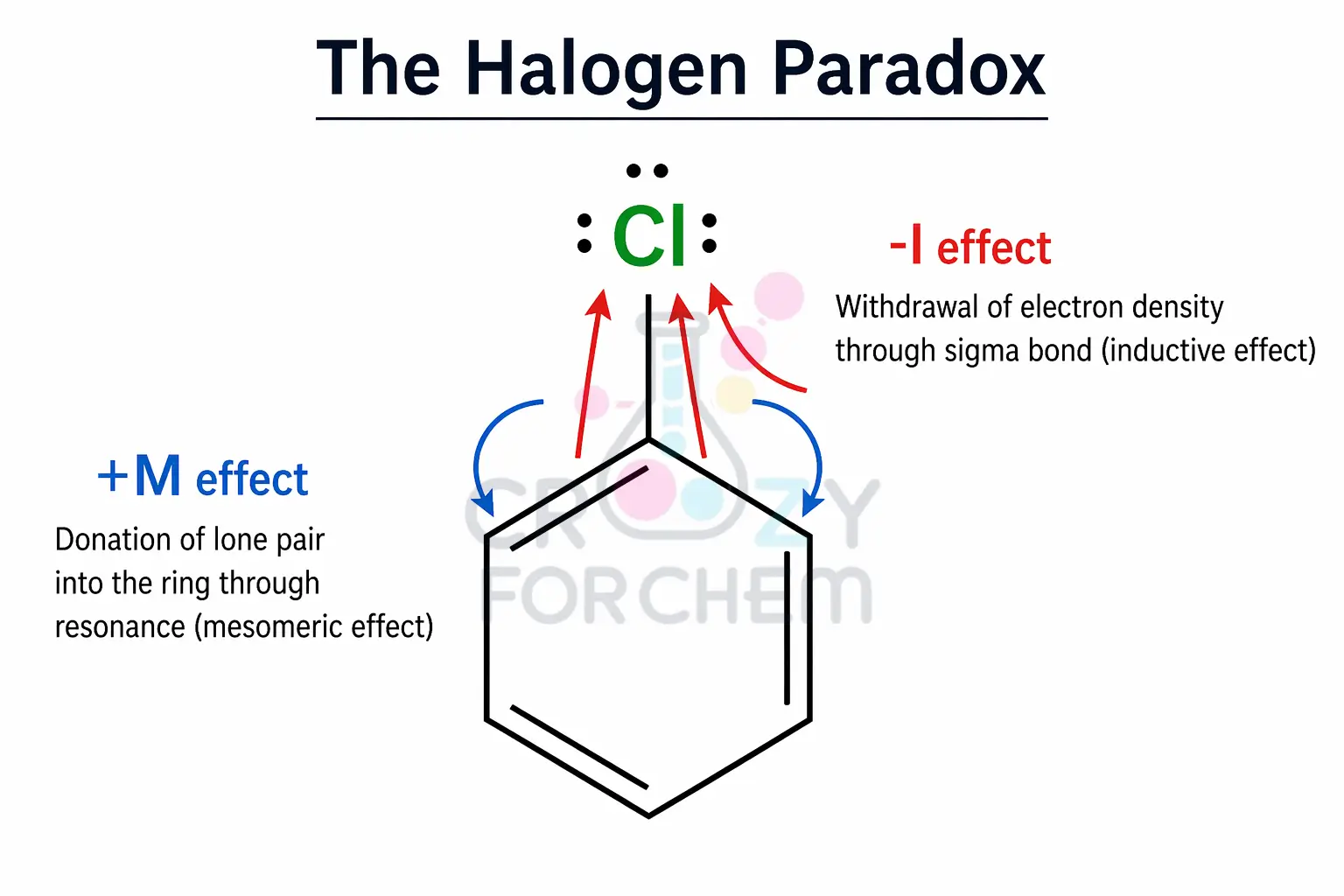
The most critical exception to the “Resonance Dominates” rule occurs with halogens (-F, -Cl, -Br, -I). Halogens present a unique case where two opposite effects operate:
- Inductive Effect (-I): They are highly electronegative and withdraw electron density through σ-bonds.
- Mesomeric Effect (+M): They possess lone pairs that can be donated into a conjugated system via resonance.
In the case of halogens, the negative inductive effect is stronger than the positive mesomeric effect regarding reactivity.
Consequently, halogens are deactivating (making the benzene ring less reactive). However, the Mesomeric effect still dictates the orientation, meaning they remain ortho/para directors because resonance still stabilises the intermediate at those positions.
Conclusion: Mastering the Electronic Hierarchy
Understanding the Inductive vs Mesomeric effect is not merely a task of memorisation but an exercise in logical deduction.
While the Inductive effect is a universal force governed by electronegativity and limited by distance, the Mesomeric effect is a powerful, long-range force restricted to conjugated systems.
By applying the hierarchy where Resonance generally outweighs Induction—with the notable exception of halogens—chemists can accurately predict the acidity, basicity, and reactivity of diverse organic molecules.
This mastery forms the academic core of organic reaction mechanisms, providing the “expert nuance” required for success in higher-level chemistry.
Frequently Asked Questions (FAQs)
-
Which is stronger: the Inductive effect or the Mesomeric effect?
In general, the Mesomeric (Resonance) effect is stronger and dominates over the Inductive effect. This is because the Mesomeric effect involves the complete delocalisation of π-electrons throughout a conjugated system, providing significantly more stabilisation than the partial displacement of σ-electrons involved in the Inductive effect.
-
Does the Inductive effect operate in conjugated systems?
Yes, the Inductive effect operates in all systems regardless of conjugation, but its influence is distance-dependent and diminishes rapidly as it moves down the carbon chain. In contrast, the Mesomeric effect only operates in conjugated systems but transmits electron density without any loss of strength regardless of the distance, provided the conjugation is continuous.
-
Why are halogens ortho/para directing despite being deactivating?
Halogens exhibit a “dual nature.” Their Inductive effect (-I) is stronger than their Mesomeric effect (+M), which causes them to withdraw electron density from the benzene ring, making them deactivating. However, the +M effect (lone pair donation) is more effective at stabilising the intermediate carbocation at the ortho and para positions, which is why they remain ortho/para directing.
-
How can I determine if the Mesomeric effect is stronger than the Inductive effect?
You can determine dominance by analysing the molecular structure: if the molecule contains a conjugated π-system, the Mesomeric effect will almost always prevail over the Inductive effect. If the system is non-conjugated, the Inductive effect becomes the primary factor controlling reactivity and stability.
-
Is the Mesomeric effect reversible like the Electromeric effect?
No, the Mesomeric effect is a permanent property inherent to the molecular structure and does not require an attacking reagent to exist. In contrast, the Electromeric effect is a temporary effect that only occurs upon the demand of an attacking reagent and is fully reversible once that reagent is removed.

