Organic chemistry is the scientific study of hydrocarbons and materials primarily composed of them, often incorporating oxygen, nitrogen, halogens, sulfur, and phosphorus.
While the sheer number of chemical transformations can seem daunting, the foundational key to mastery lies in understanding Organic Reaction Mechanisms.
By moving beyond simple rote learning and focusing on how and why electrons move, students can unlock the logic behind biological pathways and synthetic materials alike.
Table of Contents
Why Study Organic Chemistry? Understanding the Fundamentals
The Centrality of Carbon
Organic molecules are the essential building blocks of life, revolving around the unique ability of carbon to form stable covalent bonds.
Carbon’s versatility allows it to create complex chains and rings, forming the basis of everything from perfumes and cosmetics to fertilisers and food preservatives.
Understanding these structures is the vital first step toward comprehending the change in functional groups that leads to the formation of new substances.
Beyond Memorisation
Shifting from simple memorisation to a logical framework is essential for any aspiring chemist.
By learning the fundamental patterns of how reactions occur such as proton transfers, dissociations, and nucleophilic attacks you can predict products for reactions you have never encountered before.
Mastering Organic Reaction Mechanisms allows you to comprehend the three-dimensional orientation of atoms, also known as stereochemistry, which determines the physical and chemical properties of the resulting products.
This depth of understanding provides the “hypothetical motion picture” of participating atoms during a transformation.
What is an Organic Reaction Mechanism?
Defining the Mechanism
An overall description of how a chemical transformation takes place is called a reaction mechanism.
It serves as a detailed, step-by-step account of exactly what takes place at each stage of a process, specifying which bonds are broken and formed and in what precise order.
A complete mechanism must account for all reactants used and every product formed, including the role of any catalysts present.
Furthermore, a well-defined mechanism allows for the deduction of rate laws and the relative speeds of each individual step.
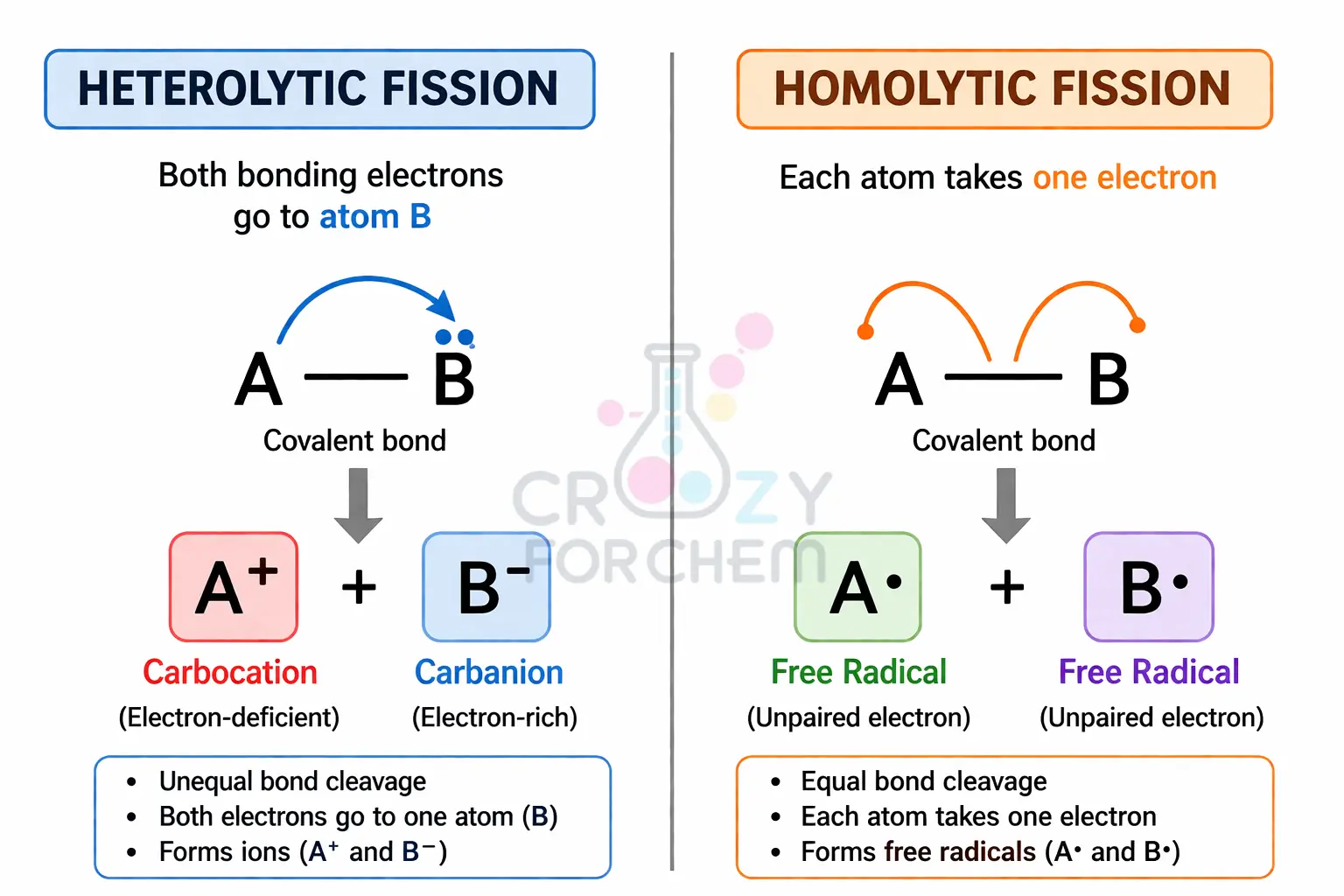
Homolytic vs Heterolytic Fission: How Bonds Break
- Heterolytic Fission: One atom takes both electrons, creating ions (Cation and Anion). This is the basis of most Nucleophilic Substitutions.
- Homolytic Fission: Each atom takes one electron, creating Free Radicals. Usually triggered by heat or UV light.
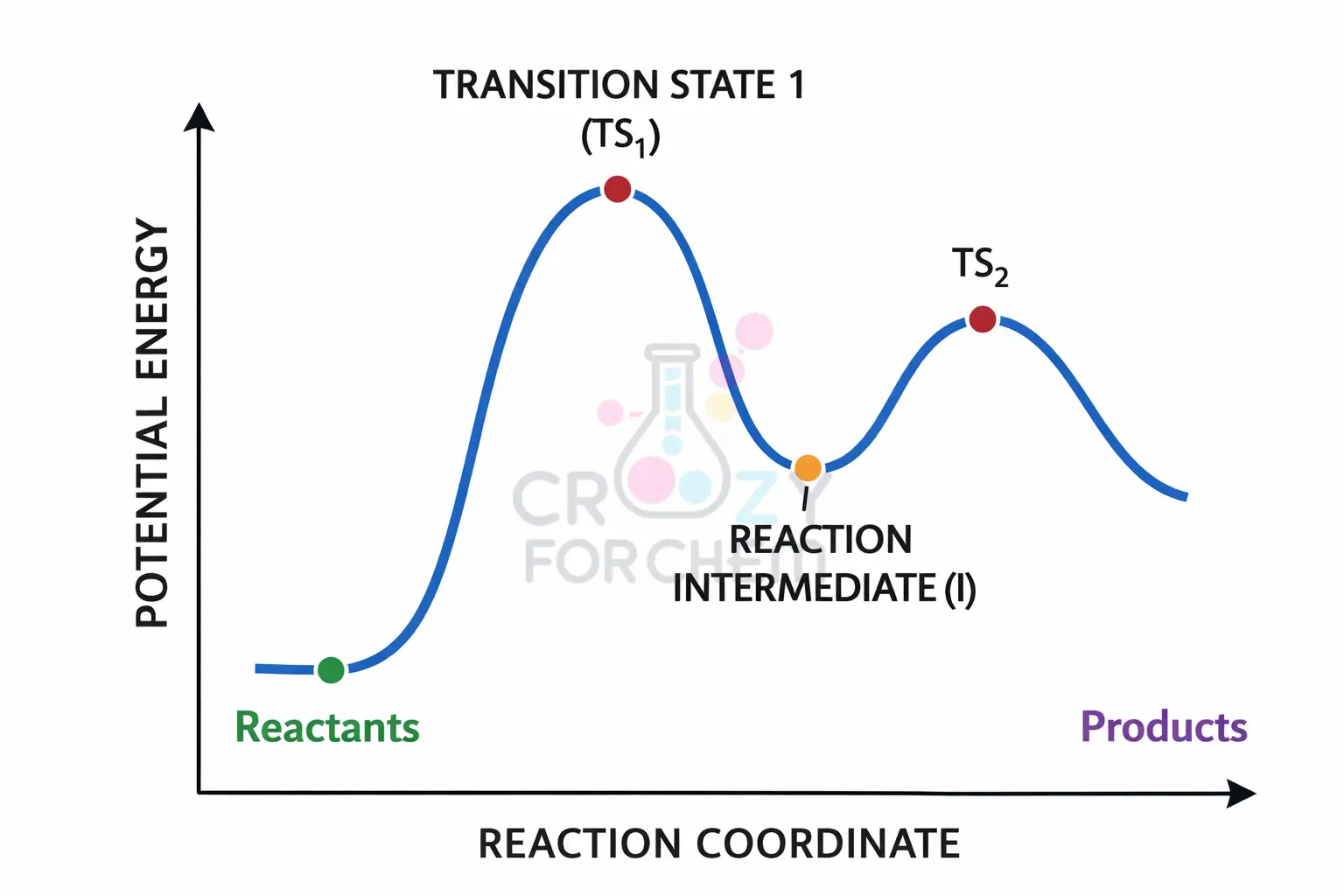
Transition State vs Reaction Intermediate (The Energy Gap)
It is crucial to distinguish between these two for JEE/NEET:
- Transition State (TS): A high-energy “peak” on the reaction coordinate. It cannot be isolated and lasts for only a vibration (~10⁻¹³ s).
- Reaction Intermediate: A “valley” or local minimum on the energy graph. It is a real chemical species (like a Carbocation) that exists for a short time.
The Role of Energetics
Every chemical reaction involves significant energy changes that dictate its feasibility and direction.
These are often visualised using potential energy diagrams, where local energy maxima represent transition states and local minima represent reaction intermediates.
Transition states are unstable molecular states with unusual numbers of bonds or unstable geometry that exist only momentarily at the peak of an energy barrier.
In contrast, intermediates are actual chemical species that are short-lived and unstable but represent a distinct stage in the reaction pathway.
The Driving Forces: Inductive, Resonance, and Hyperconjugation
Before drawing arrows, it is vital to understand the electronic effects that dictate electron density. These are the “silent directors” of every mechanism:
| Effect | Nature | Key Characteristic |
| Inductive Effect (I) | Permanent | Displacement of σ-electrons along a saturated carbon chain. |
| Resonance (M/R) | Permanent | Delocalisation of π-electrons or lone pairs in conjugated systems. |
| Hyperconjugation | Permanent | Interaction of σ-electrons of C-H bond with adjacent empty p-orbital. |
| Electromeric (E) | Temporary | Complete transfer of π-electrons in the presence of an attacking reagent. |
Mastering the Tools: The Language of Curved Arrows
The Motion of Electrons
Curved arrow notation is the graphical language used to depict the redistribution of electrons during a chemical process.
A vital rule for every student is that these arrows represent the motion of electrons, never the motion of atoms.
Electrons move from an electron “source,” such as a lone pair or a pi bond, to an electron “sink,” which is typically an electronegative atom or a positive charge.
Partial charges within a molecule often provide clues about this electron flow, as nucleophilic sites attack electrophilic centres.
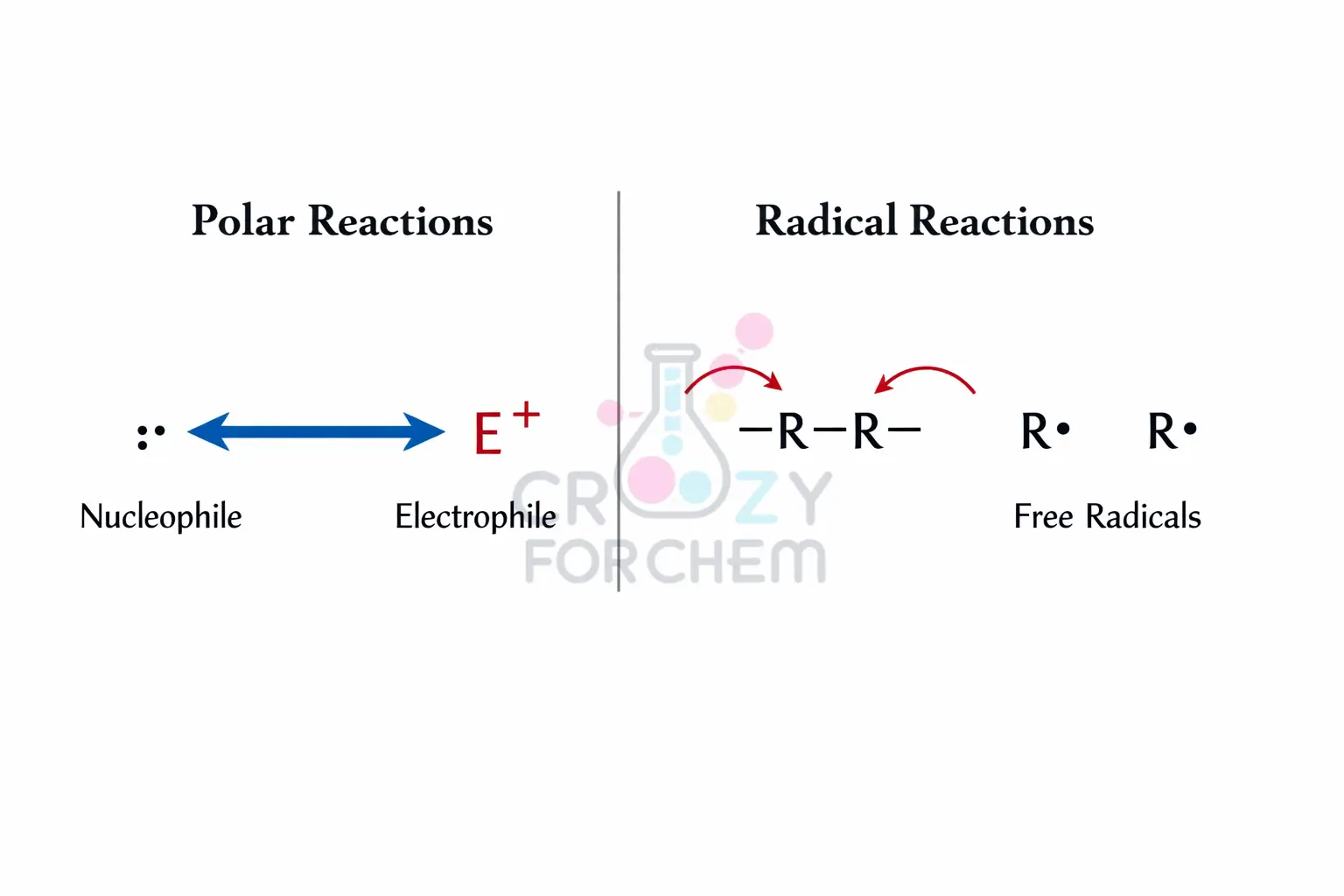
Double-Headed vs Fishhook Arrows
- Double-headed arrows indicate the movement of an electron pair. This is characteristic of polar reactions involving heterolytic bond cleavage, where one atom retains both electrons from a shared pair to form an anion, while the other becomes a cation.
- Fishhook arrows (single-headed) depict the movement of a single electron. This is used in radical reactions involving homolytic cleavage, where a bond splits evenly, and each atom takes away one electron to form highly reactive free radicals.
| Arrow Type | Meaning | Common Reaction Example |
| Full Head (Double-barbed) | Movement of an electron pair (2e⁻) | SN1, SN2, E2 (Polar Reactions) |
| Half Head (Fishhook) | Movement of a single electron (1e⁻) | Free Radical Halogenation |
| Curved Double-Headed | Resonance/Mesomerism | Showing canonical structures |
Key Components of Every Reaction
Substrates and Reagents
In organic chemistry, a reaction typically involves an organic molecule, known as the substrate, interacting with a suitable attacking reagent.
The substrate is generally the reactant that contributes carbon to the new bond being formed or is the primary focus of the study. If both reactants contribute carbon, the choice of which is the substrate is often arbitrary.
Reaction Intermediates
A vast number of reactions proceed via the formation of reaction intermediates, which are temporary reactants or products neither present in the initial starting materials nor the final products.
- Carbocations: Structure: sp² hybridised, trigonal planar.
- Stability Order: Tertiary (3°) > Secondary (2°) > Primary (1°).
- Stabilised by: +I effect and Hyperconjugation.
- Carbanions: These carry a negative charge and a lone pair on a carbon atom. They generally adopt a planar structure if the carbon bearing the negative charge is adjacent to multiple bonds, allowing for stabilisation through charge dispersal.
- Free Radicals: These species possess an unpaired electron and are typically generated through homolytic cleavage. Like carbocations, they are often stabilised by resonance and hyperconjugation.
- Carbenes and Nitrenes: Highly reactive neutral species containing only six valence electrons. Carbenes can exist in singlet or triplet states and are often involved in cyclopropanation reactions.
Nucleophiles and Electrophiles
Reactions are fundamentally driven by the attraction between electron-rich and electron-poor species.
Nucleophiles (“nucleus-loving” Lewis bases) are electron-rich entities attracted to positive centres.
Electrophiles (“electron-loving” Lewis acids) are electron-poor species that seek electrons to complete or stabilise their valence shells.
Some versatile molecules, like those containing carbonyl groups, can act as both, a property known as being amphiphilic.
The Four Major Classes of Organic Reactions
Substitution Reactions
In these reactions, one atom or group is replaced by another.
The SN1 mechanism (Unimolecular Nucleophilic Substitution) is a stepwise process where the leaving group departs first to form a carbocation intermediate, followed by a fast nucleophilic attack.
This pathway is common in tertiary substrates and is favoured by polar protic solvents.
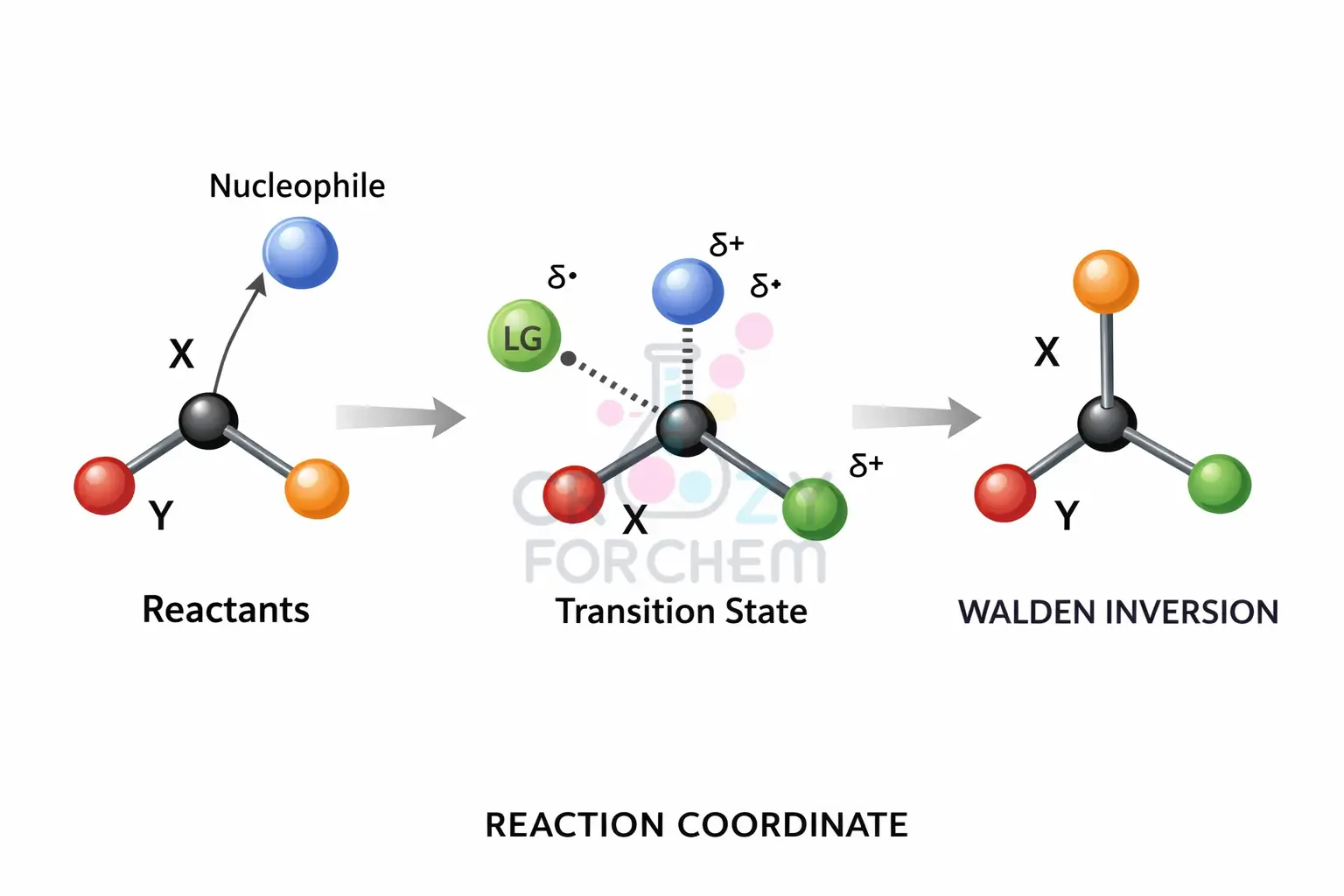
In contrast, the SN2 mechanism (Bimolecular Nucleophilic Substitution) occurs in a single, concerted step.
The nucleophile attacks the substrate from the side directly opposite the leaving group, leading to an inversion of configuration known as a Walden inversion.
To remember the difference, imagine two cats and a comfy chair: in SN2, one cat kicks the other off simultaneously; in SN1, the first cat waits for the second to leave before sitting down.
Addition Reactions
Characteristic of unsaturated molecules like alkenes and alkynes, addition reactions involve breaking a pi bond to add new atoms or groups.
This process increases the number of groups attached to the substrate and decreases its unsaturation.
Electrophilic addition is typical for alkenes, often following Markownikoff’s rule, where the electrophile adds to the carbon bearing the most hydrogens to form the most stable intermediate.
Nucleophilic addition is more common in carbonyl chemistry, where a nucleophile attacks the partially positive carbon of a polar C=O bond.
Elimination Reactions
Elimination reactions are essentially the reverse of addition, removing two atoms or groups to form a double or triple bond.
- E1 Reactions: These are two-step processes involving the formation of a carbocation intermediate. Acid-catalysed dehydration of tertiary alcohols typically proceeds via this pathway.
- E2 Reactions: These occur in a single concerted step where a strong base removes a proton as the leaving group departs. Reagents such as phosphorus oxychloride (POCl3) in pyridine are used to promote E2 mechanisms to avoid carbocation rearrangements. Elimination reactions generally follow Zaitsev’s rule, favouring the formation of the more highly substituted, stable alkene.
Rearrangement Reactions
These involve the migration of an atom or a group within a molecule to form a more stable or functional structure.
Common examples include 1,2-hydride or alkyl shifts, where a substituent moves to an adjacent carbon to transform a secondary carbocation into a more stable tertiary one.
A famous example used in synthesis is the Benzil-Benzilic Acid rearrangement, where a 1,2-diketone is converted into an alpha-hydroxy-carboxylic acid under the influence of a base.
| Feature | SN1 Mechanism | SN2 Mechanism |
| Steps | Two-step (via Carbocation) | One-step (Concerted) |
| Kinetics | First order: Rate = k[Substrate] | Second order: Rate = k[Substrate][Nu] |
| Stereochemistry | Racemisation (Partial) | Walden Inversion (100%) |
| Preferred Solvent | Polar Protic (e.g., H2O, EtOH) | Polar Aprotic (e.g., DMSO, Acetone) |
Scope of Organic Chemistry in Competitive Exams
Organic Chemistry for JEE Main & Advanced
Mastering JEE Organic Chemistry requires a deep understanding of named reactions and the ability to propose valid Organic Reaction Mechanisms based on experimental evidence, like rate laws and stereochemistry.
Advanced students must be able to interpret how changes in temperature, solvent, or catalysts dramatically alter the course of a reaction.
Organic Chemistry for NEET
For NEET Organic Chemistry, the focus is often on high-yield topics and the stability of reaction intermediates.
Students must be proficient in identifying reaction types, substitution, addition, elimination, or rearrangement based on given reactants and conditions.
Mastery of the sp³ to sp² hybridisation shifts during transition states is also a frequently tested concept.
University-Level Chemistry
At the university level, students explore advanced concepts such as pericyclic reactions, which are concerted processes governed by orbital symmetry.
This includes the Woodward-Hoffmann rules, which predict whether a reaction is thermally or photochemically allowed based on the approach and overlap of molecular orbitals.
Such advanced study explores the “symmetry-imposed barriers” to reactions, which are fundamental to complex natural product synthesis.
Real-World Applications of Organic Mechanisms
Pharmaceutical Synthesis
A primary area for organic chemistry applications is drug development.
Understanding mechanistic nuances is critical for controlling regioselectivity and avoiding unwanted side products during the synthesis of complex molecules.
For example, the detailed electron flow in the HIV protease reaction is a cornerstone of modern medicinal chemistry, allowing for the design of specific inhibitors.
Synthetic Polymers
Mechanisms also explain the creation of modern materials that define our world.
The radical-based addition reactions of alkenes, such as the polymerisation of ethene, are used to produce a vast array of synthetic polymers.
By mastering the chain propagation steps of these free radical mechanisms, chemists can tailor the properties of plastics and other essential materials.
Conclusion: Your Roadmap to Mastery
Mastering Organic Reaction Mechanisms is not about memorising an endless list of unrelated facts; it is about learning a logical, universal language.
By focusing on the flow of electrons, the stability of intermediates, and the energetics of transition states, you can unlock the complexities of both competitive exams and real-world chemical innovation.
Whether you are preparing for JEE Organic Chemistry or researching new pharmaceuticals, the mechanism remains the ultimate guide to understanding how the molecular world transforms.
Frequently Asked Questions
-
What is the fundamental role of an organic reaction mechanism?
An organic reaction mechanism provides a theoretical, step-by-step description of how a chemical transformation occurs. It details the exact sequence of bond-breaking (fission) and bond-making, the movement of electrons using curved arrow notation, and the formation of short-lived reaction intermediates such as carbocations or free radicals.
-
Why do curved arrows always point from a nucleophile to an electrophile?
In organic chemistry, curved arrows represent the movement of electron pairs. Therefore, the arrow must start at an electron-rich source (a nucleophile’s lone pair or pi bond) and point towards an electron-poor sink (an electrophile). This visual tool helps chemists predict the outcome of polar reactions accurately.
-
What is the difference between a transition state and a reaction intermediate?
A transition state is a high-energy, unstable arrangement of atoms at the peak of a reaction’s energy barrier that cannot be isolated. In contrast, a reaction intermediate (such as a carbocation, carbanion, or radical) is a distinct chemical species that exists at a local energy minimum. Intermediates are short-lived but have a finite lifetime and can sometimes be detected experimentally.
-
How does Walden Inversion affect the outcome of an SN2 reaction?
Walden Inversion is the 100% reversal of the stereochemical configuration at a chiral centre. During an SN2 mechanism, the nucleophile attacks from the side opposite to the leaving group (backside attack). This results in the final product having a spatial arrangement of atoms that is the exact opposite of the starting substrate, similar to an umbrella being blown inside out by the wind.
-
Which is more stable: a primary, secondary, or tertiary carbocation?
A tertiary carbocation is the most stable, followed by secondary, and then primary 3° > 2° > 1°. This stability trend is governed by the Inductive Effect (+I) of alkyl groups and Hyperconjugation, both of which help disperse the positive charge on the carbon atom, lowering the overall energy of the intermediate.
-
What are the 4 main types of organic reactions found in JEE and NEET exams?
The four primary classes of organic reactions are:
Substitution: Replacing one atom/group with another (e.g., SN1, SN2).
Addition: Adding atoms across a double or triple bond (e.g., Electrophilic Addition).
Elimination: Removing groups to form unsaturation (e.g., E1, E2 following Zaitsev’s Rule).
Rearrangement: Structural reordering to form more stable structures (e.g., 1,2-Hydride Shift).

