For thousands of years, the onion (Allium cepa) has been a staple of human civilisation.
The ancient Egyptians valued them so highly that they were used as currency to pay the workers who built the pyramids, and they were even placed in the eye sockets of mummies, such as King Ramesses IV, as a symbol of eternity.
The Romans used them for therapeutic applications ranging from treating dog bites to hair loss. However, despite this long history, one mystery remained for ages: the science of onion tears.
Whether you are preparing a French onion soup or a simple salad, the stinging sensation and subsequent “waterworks” are almost universal.
This is not an emotional response but a complex chemical defence mechanism that the onion has evolved over millions of years to survive.
In this comprehensive guide, we will explore the molecular biology of the onion and the chemistry of the “tear gas” it produces and provide the ultimate science-backed strategies on how to cut onions without crying.
Table of Contents
1. The Evolutionary Shield: Why Onions “Fight Back”
To understand the science of onion tears, we must first look at where the onion grows.
As a perennial bulb that matures underground, the onion is a prime target for soil-dwelling predators like voles, insects, and microbes.
Because the onion cannot run away, it has developed a sophisticated system of “chemical warfare”.
Sulfur Absorption from the Soil
Onions are grown in sulphur-rich soil. Sulphur is an essential nutrient for the plant; it absorbs this element and converts it into amino acids and proteins, storing it in the form of sulphur-containing organic molecules.
These molecules are the building blocks of the onion’s flavour and its defensive capabilities.
Cellular Compartmentalisation
The ingenious design of an onion cell keeps its defensive weapons in separate “rooms”.
Inside the cell, fluids hold sulphur compounds (amino acid sulfoxides), while a specific enzyme trigger called ‘alliinase’ is held separately in a storage area called a ‘vacuole’.
As long as the onion remains intact, these two components never meet, and the onion remains relatively odourless.
2. The Chemical Cascade: How the “Gas” is Formed
The moment you slice, dice, or bite into an onion, you rupture these cellular barriers.
This allows the previously separated enzymes and sulphur compounds to mix, initiating an instant cascade of chemical processes.
- Enzymatic Activation: The alliinase enzyme immediately begins breaking down the amino acid sulfoxides into 1-propenesulfenic acid.
- The LFS Enzyme Discovery: For a long time, scientists thought alliinase was solely responsible for the tears. However, the science of onion tears took a major leap forward in 2002 when Japanese scientists discovered a second enzyme specifically responsible for the lachrymatory effect: lachrymatory-factor synthase (LFS).
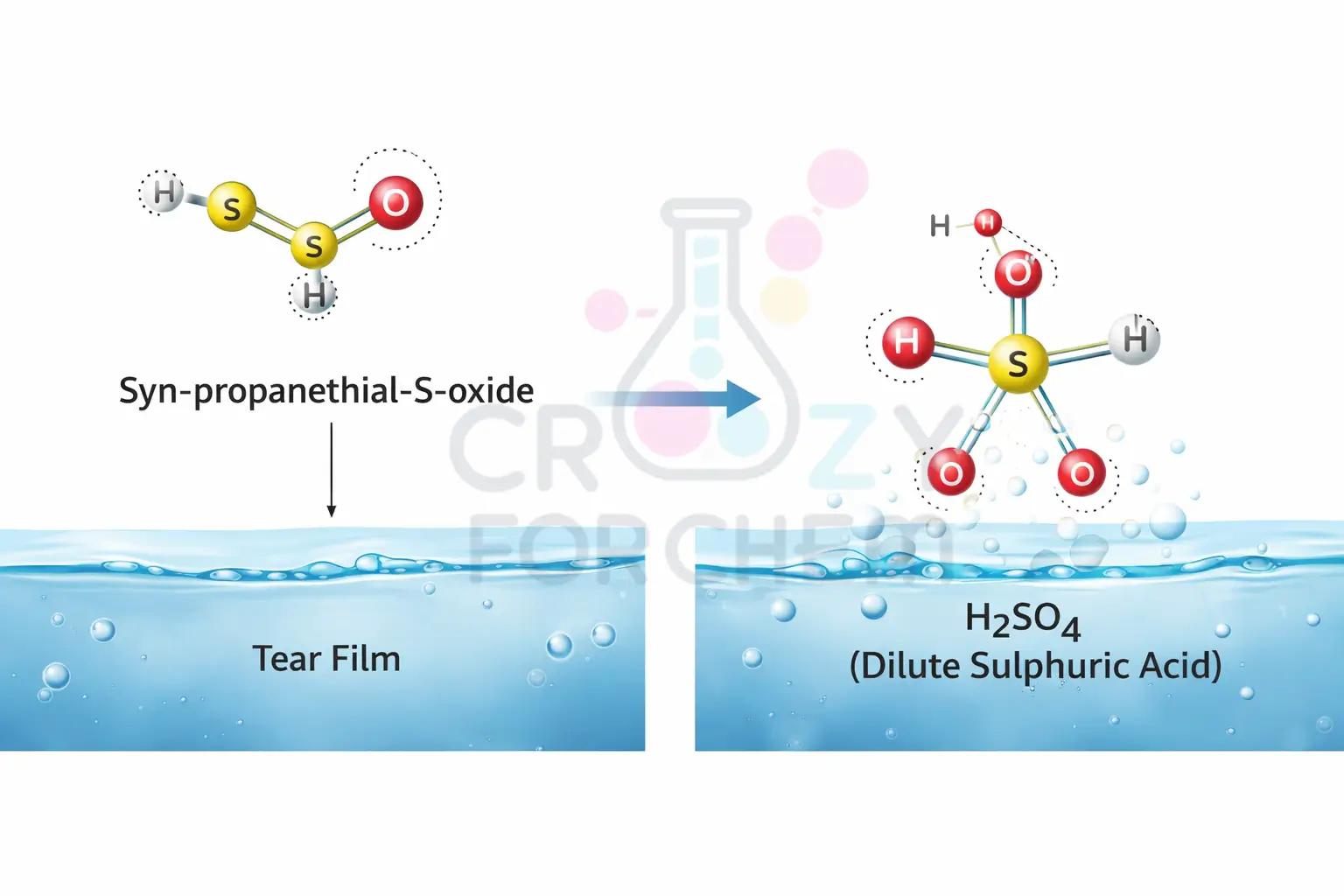
- The Final Product: LFS takes the 1-propenesulfenic acid and rearranges it into a volatile gas called syn-propanethial S-oxide.
This gas is a “lachrymatory agent”, meaning it is specifically designed to generate tears when it touches an animal’s eye.
Because the molecules are incredibly small and light, they quickly aerosolise at room temperature and float up towards your face.
3. Why It Stings: The Sulphuric Acid Reaction
Once the syn-propanethial-S-oxide gas reaches your eyes, it interacts with the natural water layer (the tear film) that covers and protects your eyeballs. This is where the chemistry becomes particularly aggressive.
The gas reacts with the moisture in your eye to form a very dilute version of sulphuric acid (H₂SO₄). While it is extremely dilute, it is still a corrosive irritant that attacks the nerve endings in your cornea directly.
Your brain detects this acid as a threat to your vision and sends an emergency message to your lacrimal glands (tear ducts). These glands go into overdrive, producing reflexive tears to flush the irritant away and dilute the acid. This is the fundamental reason why do onions make you cry.
4. Comparing Onion Varieties: Ranking the Pungency
Not all onions are equally offensive. The levels of sulphur absorbed from the soil and the concentration of enzymes vary between varieties.

Table 1: Pungency Ranking of Common Onions
| Onion Type | Tear Potential | Scientific Reasoning |
|---|---|---|
| Yellow Onions | Very High | High concentration of sulphur compounds, the most pungent. |
| Red Onions | High | Rich in sulphur and flavonoids (anthocyanins); very irritating. |
| White Onions | High | Standard sulphur levels frequently cause significant tearing. |
| Sweet Onions | Medium/Low | Often grown in low-sulphur soil (e.g., Vidalia), making them milder. |
| Green Onions | Low | Contain less sulphur and produce fewer tears for most people. |
| Sunions | Zero | A specific cross-bred “tearless” variety that is sweet and non-pungent. |
5. Contact Lenses vs. Glasses: The Protective Barrier
One of the most debated topics in the science of onion tears is whether eyewear helps. Experiments by optometrists and home cooks have yielded interesting results.
“As a chemistry professional, I always recommend safety goggles if you’re sensitive, but for home cooks, the hacks in Section 6 are your best bet.”
- Contact Lenses: Many wearers report being “immune” to onion eyes. This is because a contact lens acts as a physical shield over the cornea, covering the sensitive nerves and pain receptors that would otherwise react to the gas.
- The Downside of Contacts: However, lenses can also disrupt the tear film or lead to dry eyes, which can actually make the irritation worse if the gas gets behind the lens, as it becomes harder to flush away.
- Glasses: Standard glasses provide a very slight baffle against the air but do not seal the eyes, so the gas can easily float around the lenses and reach the eye.
6. How to Cut Onions Without Crying: Science-Backed Hacks
There are countless “grandmother’s remedies” for onion tears, but which ones actually hold up to scientific scrutiny? We tested the most popular theories.
Table 2: Effectiveness of Popular Onion-Cutting Hacks
| Hack | Effectiveness | Scientific Verdict |
|---|---|---|
| Chilling/Freezing | High | Cold temperatures slow down enzymatic reactions and make gas less volatile. |
| Sharp Knife | High | Cleanly slices cells instead of crushing them; releases significantly less gas. |
| Ventilation/Fan | Very High | Physically removes the volatile molecules from your air space. |
| Submerged in Water | High | Water neutralises the gas before it reaches your eyes but makes onions soggy. |
| Bread in Mouth | Zero | The bread does not absorb the gas; it only masks the smell for your nose. |
| Candle Light | Zero | Lighting a candle might mask the odour, but it does nothing to stop the acid formation in your eyes. |
Pro-Tips for the Kitchen:
- The Sharpness Rule: As emphasised by professional chefs, a dull knife is your enemy. Use a sharp blade and a “rock and roll” cutting technique to minimise cell damage.
- Avoid the Root: The root end of the onion contains the highest concentration of sulphur compounds. Leave the root intact until the very last slice to keep the irritants contained.
- The Freezer Trick: Popping an onion in the freezer for 30 minutes (or the fridge for 2 hours) is one of the most practical ways to reduce kitchen tears.
7. Safety and Health: Onions Beyond the Kitchen
While the science of onion tears is mostly about temporary irritation, onions are a powerhouse of nutrition and have significant health impacts.
- For humans, onions are rich in antioxidants, vitamin C, and flavonoids like quercetin. Clinical trials have even suggested they can help reduce blood glucose concentrations in diabetic patients.
- The “Reflex” Protection: Crying is actually healthy. It is your body’s way of lubricating and protecting the eye from dust, smoke, and acid.
- 🚨 PRO-TIP: Warning for Pets: While healthy for us, onions are poisonous to dogs and cats. Ingesting onions can cause life-threatening anaemia and bloody urine in pets. Never feed onion scraps to your animals.
8. The Future: Genetically Modified and Tearless Onions
The quest to master the science of onion tears has led to the development of the Sunion, a tearless onion variety created through decades of natural cross-breeding.
By selecting plants with lower levels of lachrymatory-factor synthase, farmers in Nevada and Washington have produced an onion that actually gets sweeter and less pungent the longer it is stored.
Scientists are also exploring genetic modification to “silence” the LFS gene entirely, which could make the standard tear-inducing onion a thing of the past.
Conclusion
Understanding why onions make you cry transforms a kitchen annoyance into a fascinating lesson in chemistry.
The next time you feel that familiar sting, remember that it is just a tiny amount of Sulphuric Acid triggering your body’s brilliant defence mechanism.
By keeping your knives sharp, your onions cold, and your kitchen well-ventilated, you can enjoy all the culinary benefits of this “majestic bulb” without the tears.
Frequently Asked Questions
-
Why do onions make you cry?
The science of onion tears involves a chemical reaction. When you cut an onion, you break its cells, releasing an enzyme called alliinase and sulphur compounds. These react to form a volatile gas called syn-propanethial-S-oxide. When this gas hits the moisture in your eyes, it turns into a very dilute sulphuric acid, triggering your tear ducts to flush the irritant away.
-
How can I cut onions without crying?
To stop the stinging, you must slow down the chemical reaction or move the gas away. The most effective methods are chilling the onion in the fridge for an hour, using a very sharp knife to minimise cell damage, and working in a well-ventilated area or near a kitchen fan.
-
Does the “bread in mouth” hack actually work?
No. Scientific testing shows that holding a piece of bread in your mouth is a myth. The bread does not absorb the sulphur gas before it reaches your eyes. Chilling and ventilation remain the only science-backed strategies for a tear-free experience.
-
Are some onions less likely to cause tears?
Yes. Sweet onions, such as Vidalia, are often grown in low-sulphur soil and are much milder. Recently, cross-bred varieties like Sunions have been developed to be completely tearless, actually becoming sweeter and less pungent the longer they are stored.
-
Is the gas from onions harmful to your eyes?
While the sensation is painful, it is not harmful. The sulphuric acid produced is extremely dilute, and the “reflex tears” produced by your body are a brilliant natural defence mechanism designed to protect your vision and clean the surface of the eye.
-
Why doesn’t garlic make you cry as onions do?
Although garlic is a close relative of the onion and also contains sulphur, it lacks the specific enzyme called lachrymatory-factor synthase (LFS). Without this enzyme, the chemical reaction cannot produce the volatile “tear gas” that onions are famous for.
