The physical state of matter, whether a substance is a solid, liquid, or gas, is determined by the silent tug-of-war between molecules known as intermolecular forces.
In this comprehensive guide, we will explore the fundamental nature of these forces, with a specific focus on the unique mechanics of hydrogen bonding in H₂O vs H₂S.
We will examine why water exists as a life-sustaining liquid at room temperature while its chemical “cousin,” hydrogen sulphide, remains a toxic gas, despite their similar molecular structures.
Along the way, we will cover the different types of intermolecular attractions, the unique properties they impart to substances, and the biological significance of these “weak” but essential interactions that define the very world around us.
Table of Contents
1. Defining Intermolecular Forces (IMFs)
Intermolecular forces, or IMFs, are the attractive forces that exist between atoms, ions, or molecules, holding them together in a specific physical state.
It is crucial to distinguish these from intramolecular forces, which are the strong chemical bonds (such as covalent or ionic bonds) that hold the atoms together within a single molecule.
A helpful way to remember this distinction is through a sports analogy: intercollegiate sports are played between different colleges, whereas intracollegiate (or intramural) sports are played within a single college.
While intramolecular bonds are incredibly strong, intermolecular forces are typically significantly weaker; a hydrogen bond is only about one-tenth as strong as a covalent bond.
Despite their relative weakness, IMFs are responsible for the boiling points, melting points, and viscosities of all substances.
2. The Spectrum of Intermolecular Attractions
To understand why substances behave differently, we must categorise the six different types of IMFs based on the arrangement and movement of their electrons.
London Dispersion Forces (LDF)
London Dispersion Forces are the weakest type of IMF and exist between all molecules, including non-polar ones and noble gases. These forces arise due to the constant movement of electrons. [Periodic Table Trends].
At any given instant, a non-polar molecule might have more electrons on one side than the other, creating a temporary dipole. This temporary dipole then induces a dipole in a neighbouring molecule, leading to a weak attraction.
The strength of LDFs increases as the size of a molecule increases. This is because larger molecules have more electrons and a more polarisable electron cloud, meaning the cloud is more easily distorted to create a dipole.
Dipole-Dipole Interactions
These attractions occur between polar molecules, those with a permanent partial positive (δ+) end and a partial negative (δ-) end.[Fajans’ Rule].
The opposite partial charges on neighbouring molecules attract each other. The more polar a molecule is, the stronger the dipole-dipole interaction, which leads to higher boiling and melting points.
Ion-Dipole and Ion-Ion Interactions
- Ion-Dipole Forces: These exist when an ion is attracted to a polar molecule, such as when common salt (NaCl) dissolves in water. These are generally stronger than standard dipole-dipole or hydrogen bonds.
- Ion-Ion Interactions: These are the strongest intermolecular forces, occurring in ionic compounds. Because these electrostatic forces are so powerful, most ionic compounds are solids at room temperature with extremely high melting points.
3. The Mechanics of Hydrogen Bonding
Hydrogen bonding is not a true chemical bond but an unusually strong type of dipole-dipole attraction.
It occurs only when a hydrogen atom is directly bonded to one of the three most electronegative and small elements: Fluorine (F), Oxygen (O), or Nitrogen (N), often remembered by the mnemonic “H-Phone”.
Because F, O, and N are so “electron-greedy” (electronegative), they pull the shared electrons away from the hydrogen atom.
This leaves the hydrogen with a significant partial positive charge, which is then strongly attracted to the lone pairs of electrons on the F, O, or N atom of a neighbouring molecule.
Types of Hydrogen Bonding: Inter vs Intra
- Intermolecular Hydrogen Bonding: This occurs between separate molecules. This is the force that holds water molecules together in a liquid state.
- Intramolecular Hydrogen Bonding: This occurs within a single molecule, such as in ethylene glycol or the base pairs of DNA, where it holds the double helix together.
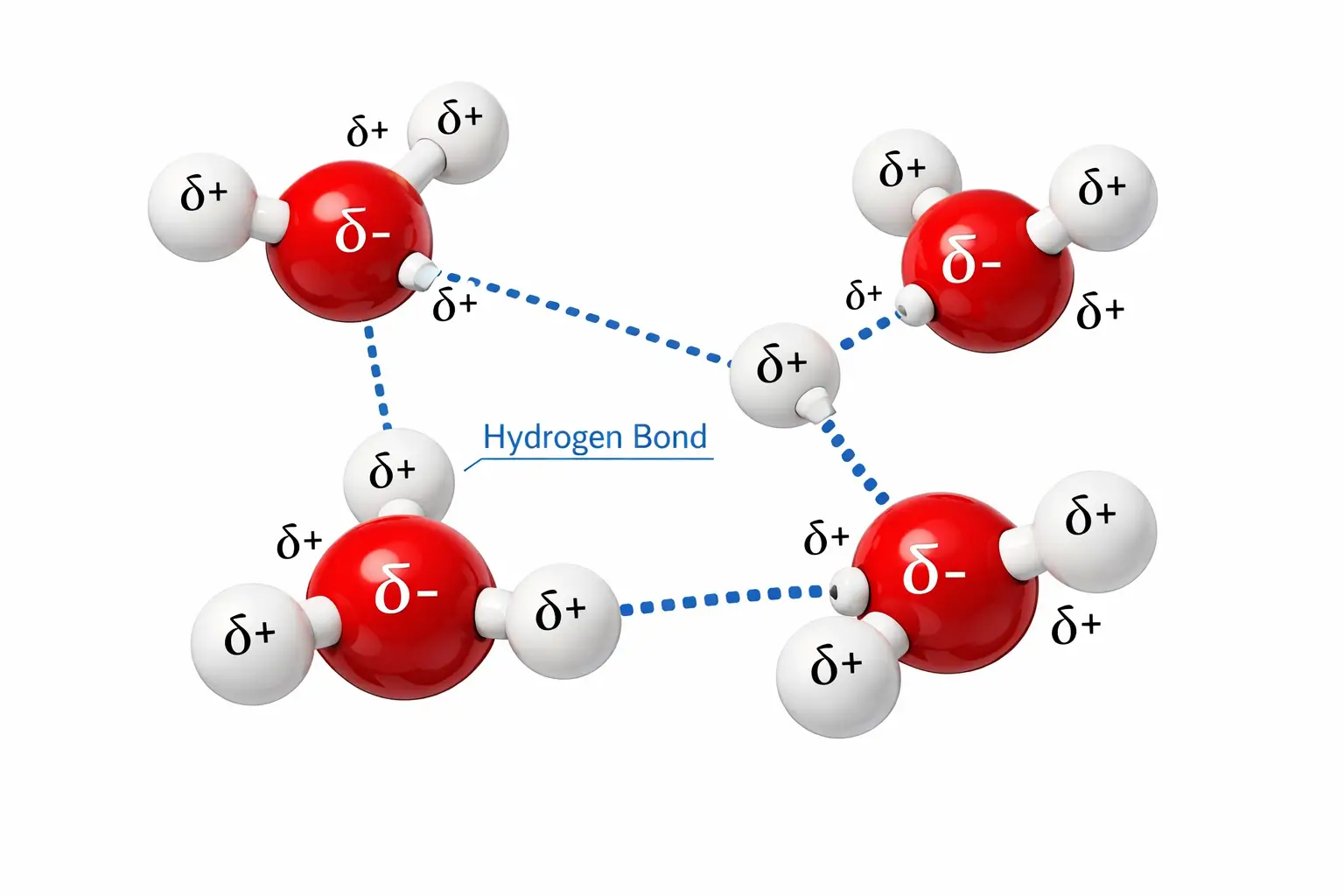
4. The Big Mystery: H₂O vs. H₂S
One of the most frequently asked questions in chemistry is: Why is H₂O a liquid while H₂S is a gas? Both molecules belong to Group 16 of the periodic table and share a similar “bent” molecular geometry.
The Case for Water (H2O)
In a water molecule, hydrogen is bonded to oxygen, which has a very high electronegativity and a small atomic size. This creates a highly polar O-H bond, allowing for extensive intermolecular hydrogen bonding. [VSEPR Theory Guide].
Each water molecule can form an average of two hydrogen bonds with neighbouring molecules. These strong attractions require significant energy (heat) to break, resulting in a high boiling point of approximately 100°C.
The Case for Hydrogen Sulphide (H₂S)
Sulphur is located directly below oxygen on the periodic table. Although it has the same number of valence electrons, sulphur is much larger and significantly less electronegative than oxygen.
Because the electronegativity difference is low, S-H groups are unable to form hydrogen bonds. Instead, H₂S molecules are held together only by weak van der Waals forces and dipole-dipole interactions.
With such weak “molecular glue,” H₂S requires very little energy to separate, boiling at -60°C and existing as a gas at room temperature.
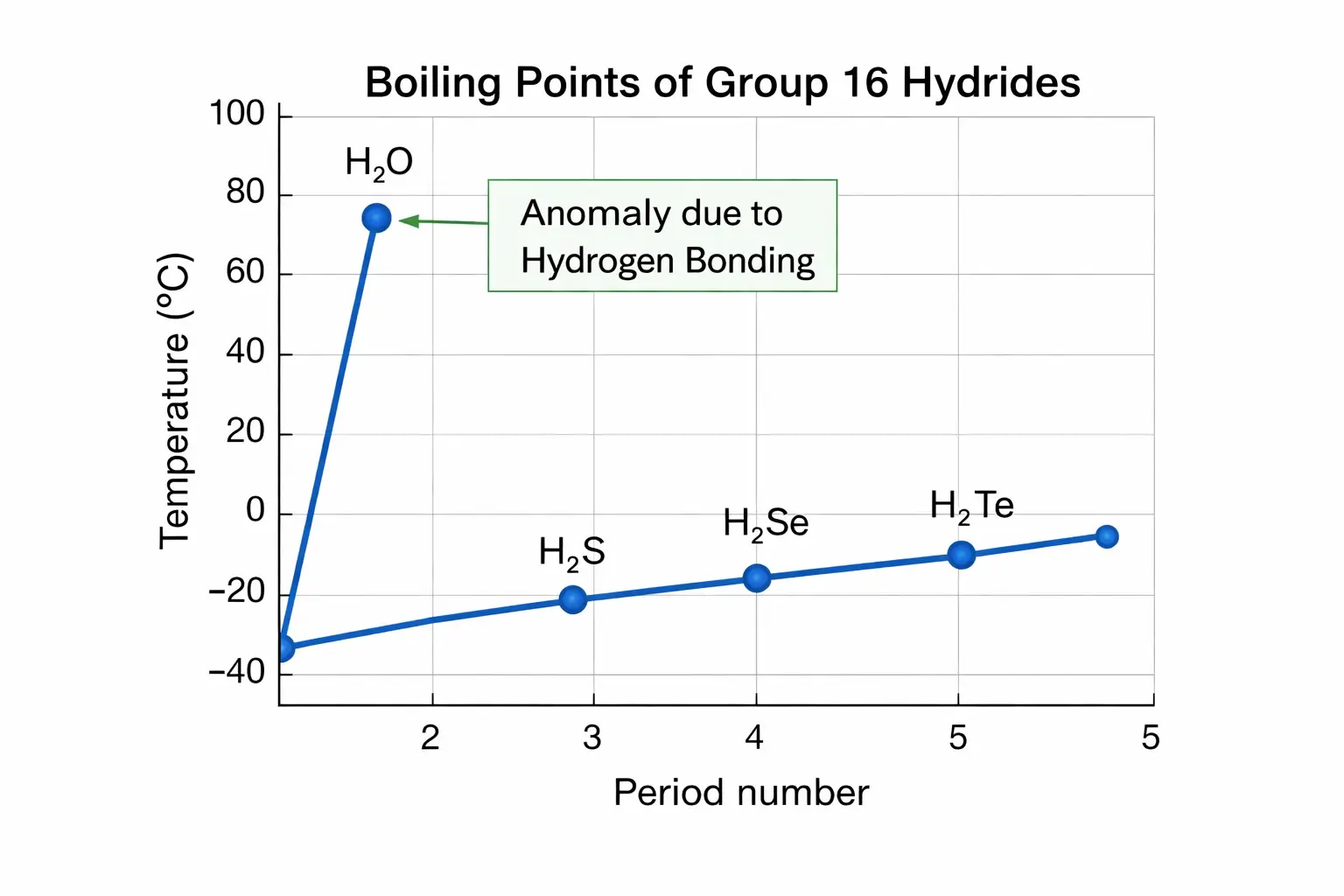
5. Comparison with Ammonia
To further illustrate the power of these forces, we can look at ammonia. Nitrogen is located right next to oxygen on the periodic table. Like water, ammonia can form hydrogen bonds because hydrogen is bonded to nitrogen.
However, ammonia is a gas at room temperature with a boiling point of -33.34°C. Why is it a gas while water is a liquid?
- Hydrogen Bond Density: Each ammonia molecule has one lone pair of electrons and three hydrogen atoms, meaning it can only form an average of one hydrogen bond per molecule.
- Water’s Advantage: Water has two lone pairs and two hydrogens, allowing it to form two hydrogen bonds per molecule. Because water has more hydrogen bonds per molecule, more energy is needed to separate them, resulting in a much higher boiling point.
6. Practical Consequences of IMFs
Intermolecular forces dictate nearly all the physical properties we observe in the laboratory.
- Boiling and Melting Points: Stronger IMFs (like hydrogen bonding) lead to higher boiling and melting points.
- Viscosity: This is a measure of a liquid’s resistance to flow. Viscosity increases with stronger IMFs; for example, honey has a higher viscosity than water because it has stronger intermolecular attractions.
- Vapour Pressure: This is the pressure exerted by a gas in equilibrium with its liquid phase. Substances with strong IMFs have lower vapour pressures because the molecules are too strongly attracted to the liquid to easily escape into the gas phase.
7. The Ice Anomaly: Why Ice Floats
Hydrogen bonding is also responsible for the unusual density of ice. In most substances, the solid phase is denser than the liquid phase.
However, as water freezes, the hydrogen bonds stabilise and orient the molecules into a rigid, hexagonal “open-cage” structure.
This arrangement keeps the water molecules further apart in the solid state than they are in the liquid state. Because the same mass of water occupies a larger volume as ice, its density decreases, causing ice cubes and icebergs to float.
This anomaly is vital for life; a layer of floating ice insulates the water below, allowing aquatic life to survive even when surface temperatures are below freezing.
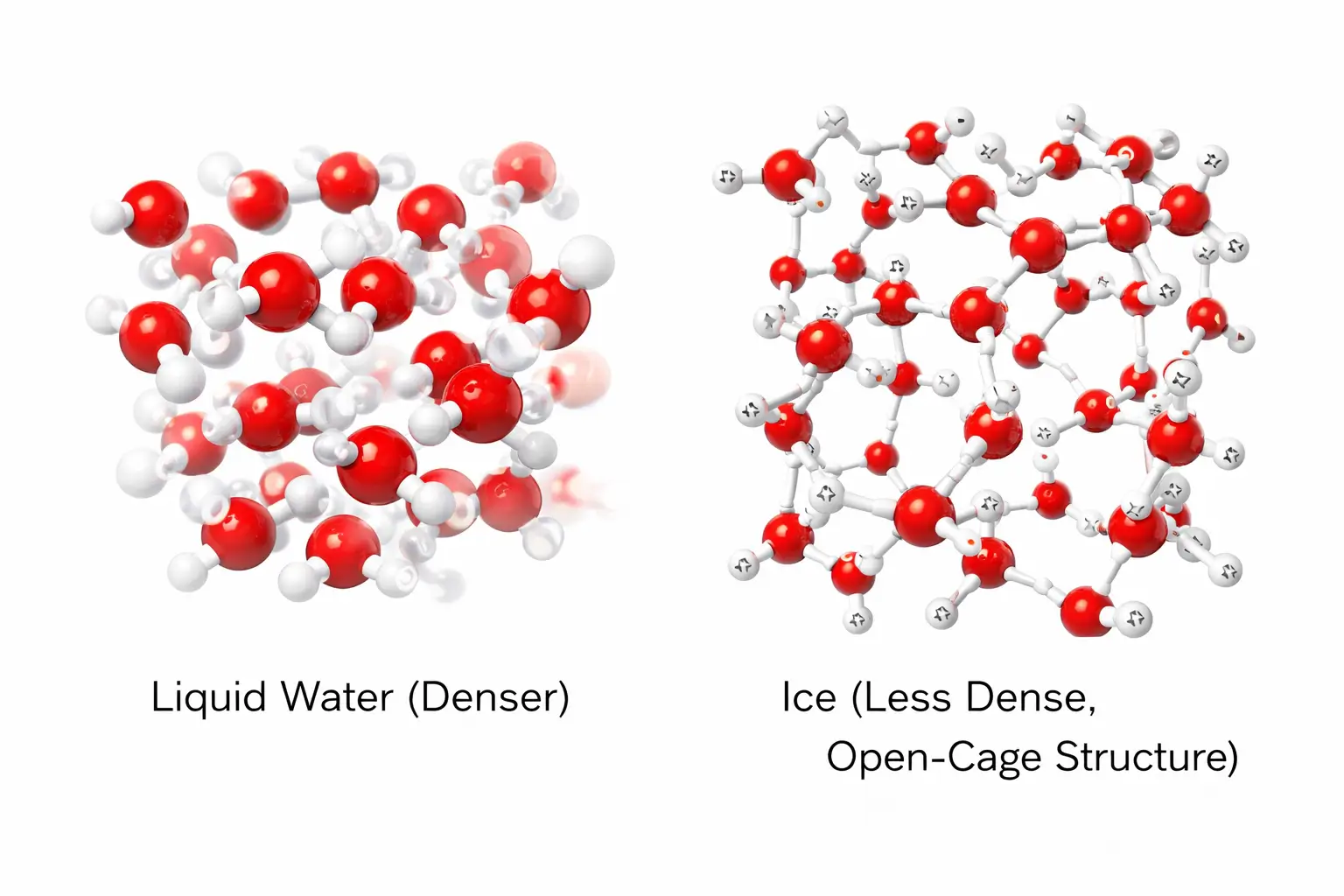
8. Biological and Chemical Importance
Without hydrogen bonding, life as we know it would not exist. The human body is 60% to 70% water, and most cellular metabolism occurs in a water-based “goo” called cytosol.
- DNA Stability: Hydrogen bonds are responsible for holding the two strands of the DNA double helix together. They are strong enough to keep the genetic code secure but weak enough to be “unzipped” during replication.
- Solvent Properties: Water is often called the “universal solvent” because its polarity and ability to form hydrogen bonds allow it to dissolve many polar and charged molecules. Substances that interact well with water are termed hydrophilic (“water-loving”), while non-polar molecules like oils are hydrophobic (“water-fearing”).
9. Summary: Predicting Molecular Behaviour
To determine which type of IMF a substance will exhibit, one must examine the types of atoms present and the molecular polarity.
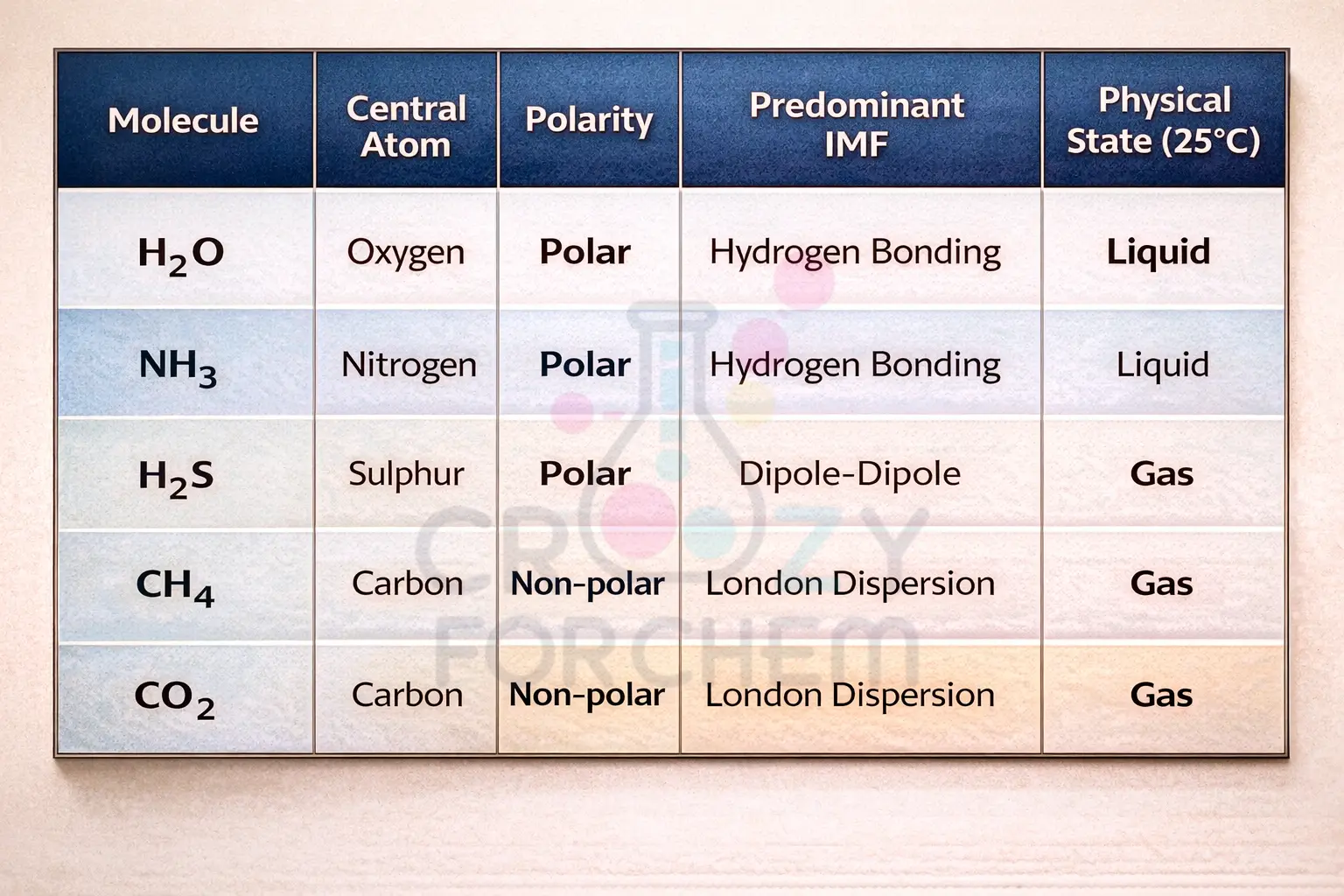
Conclusion: Hydrogen Bonding in H₂O vs H₂S
The reason water is a liquid while hydrogen sulphide is a gas comes down to the exceptional strength of hydrogen bonding in H₂O vs H₂S.
While sulphur and oxygen sit close to each other on the periodic table, the high electronegativity and small size of oxygen allow it to form a dense network of intermolecular attractions that sulphur simply cannot match.
By mastering these forces, we can predict the behaviour of everything from the air we breathe to the DNA that defines our existence.
💡 CrazyForChem Fact: While we often think of hydrogen bonding as a “water thing,” it is also what makes your hair change shape when it gets wet! Water molecules disrupt the hydrogen bonds between protein chains in your hair, allowing it to be restyled until it dries and the bonds reform.
Frequently Asked Questions
-
Is a hydrogen bond a true chemical bond?
Ans: No, a hydrogen bond is not a true chemical bond like a covalent or ionic bond. It is an exceptionally strong intermolecular force of attraction. While a covalent bond involves sharing electrons within a molecule, a hydrogen bond is an electrostatic attraction between the molecules themselves. It is typically about 10 times weaker than a covalent bond.
-
Why is H₂O a liquid while H₂S is a gas at room temperature?
Ans: The difference lies in Hydrogen Bonding. Oxygen is small and highly electronegative, allowing water molecules to form strong hydrogen bonds that keep them in a liquid state. Sulphur is larger and less electronegative; therefore, H₂S molecules are only held by weak van der Waals forces, which are easily broken at room temperature, making it a gas.
-
Which is stronger: a Hydrogen bond or a Van der Waals force?
Ans: A Hydrogen bond is significantly stronger than standard Van der Waals forces (like London Dispersion Forces or Dipole-Dipole interactions). This is because the partial charges involved in H-bonding (on F, O, or N) are much more concentrated and intense.
-
Why does Ice float on Water?
Ans: Ice floats because it is less dense than liquid water. Due to hydrogen bonding, water molecules freeze into a rigid, hexagonal “open-cage” structure. This structure pushes the molecules further apart compared to their liquid state, increasing the volume and decreasing the density.
-
Does HCl show hydrogen bonding?
Ans: No, HCl does not show significant hydrogen bonding. Although Chlorine is electronegative, its atomic size is too large. For an effective hydrogen bond, the atom must be small and highly electronegative (like Fluorine, Oxygen, or Nitrogen) to create a high charge density that can strongly attract a hydrogen atom.
-
Why is the boiling point of H₂O higher than NH3 (Ammonia)?
Ans: Even though both have hydrogen bonding, water has a higher boiling point because it has a higher “Hydrogen Bond Density.” Each water molecule can form an average of two hydrogen bonds (due to two lone pairs and two hydrogens), whereas ammonia typically forms only one, as it has only one lone pair.
