Spices have been woven into the fabric of human civilisation for millennia, with the desire to monopolise the spice trade driving global exploration as early as the 13th century.
India, famously known as the ‘Land of Spices’, grows more than 50 different types, including wide tropical varieties such as black pepper, cardamom, ginger, and turmeric.
Beyond their delightful flavour and pungency, these spices are indispensable because they possess diverse medicinal and pharmacological properties used in traditional Ayurvedic and modern pharmaceutical preparations.
The fundamental chemistry of Indian spices revolves around “secondary metabolites”, organic compounds that do not directly participate in plant growth but provide essential defence mechanisms and sensory attributes.
Understanding these chemical compounds in spices allows us to appreciate the scientific depth of the Indian kitchen, transforming it into a “natural pharmacy” that can reverse adverse physiological disorders.
/ta
Table of Contents
1. The Golden Alchemy of Turmeric (Haldi)
Turmeric (Curcuma longa L.) is perhaps the most iconic component of the chemistry of Indian spices, valued equally as a food additive and a therapeutic agent.
Its vibrant core ranges from yellow to bright orange-yellow, depending on the concentration of its primary bioactive compounds in kitchen spices.
Curcumin: The Hero Polyphenol
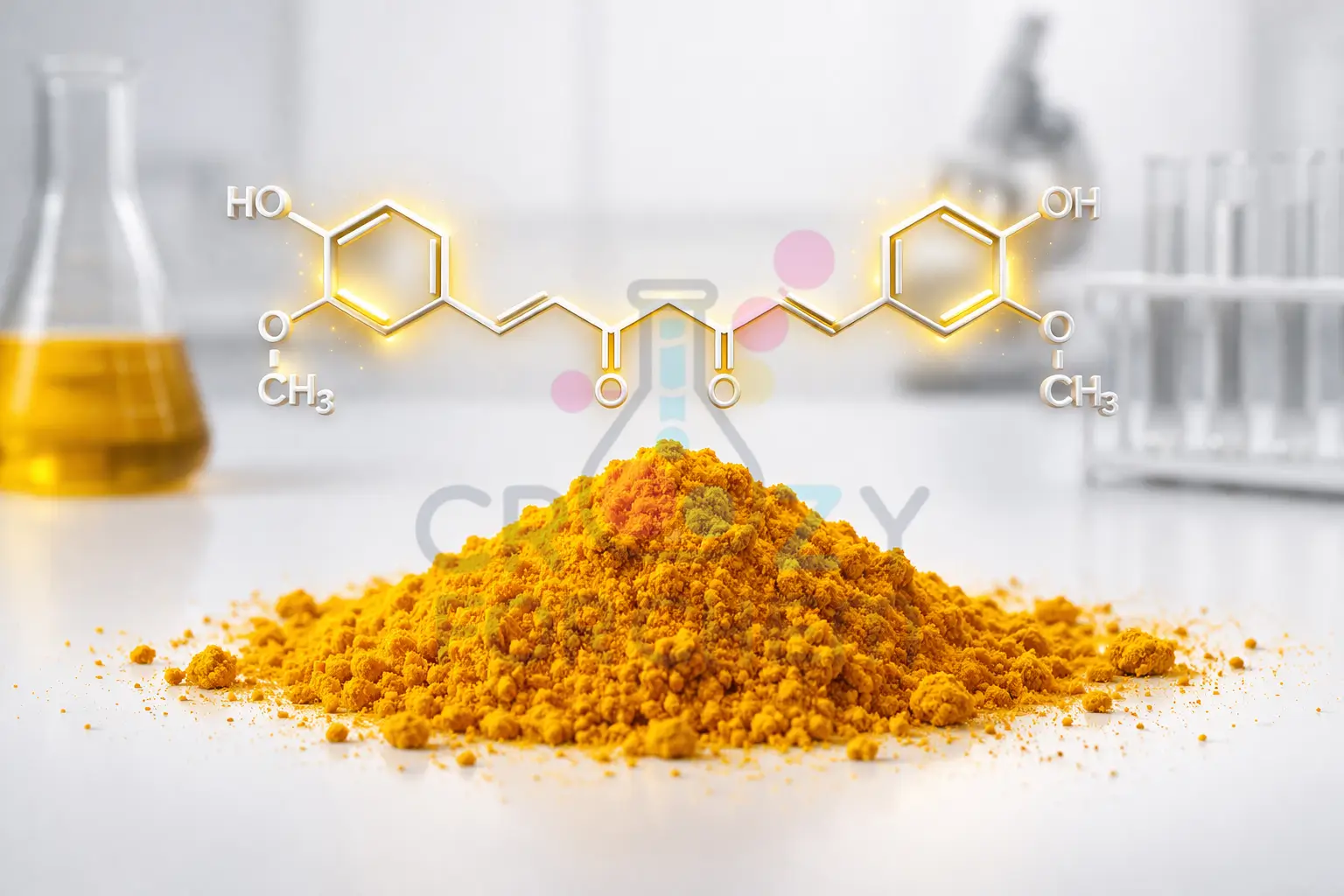
The primary bioactive principle in turmeric is curcumin (diferuloylmethane), which is insoluble in water but highly soluble in ethanol and acetone.
Curcumin typically constitutes 2.5–6% of the rhizome and is renowned for its anti-inflammatory, antioxidant, anti-carcinogenic, and wound-healing properties.
Research indicates that curcumin blocks several enzymes required for the growth of tumours, making it a focus for future cancer treatments.
Variability and Geographical Impact
The concentration of chemical compounds in spices is not uniform; it is highly dependent on location, agroclimatic conditions, and harvesting techniques.
For example, turmeric varieties like IISR Prathibha yield about 6% curcumin when grown in Kerala, but this recovery drops to 3–4% in Tamil Nadu or Andhra Pradesh and even lower in Madhya Pradesh.
The pH Sensitivity “Hack”
A fascinating chemical characteristic of turmeric is its role as a natural pH indicator. In acidic or neutral solutions, curcumin remains a stable yellow.
However, when it encounters an alkaline (basic) environment, such as common household soap or lime, the chemical structure rearranges, causing it to turn a brilliant red or brown.
This is the scientific reason why a turmeric-stained cloth turns red when scrubbed with detergent.
2. The Fiery Science of Chilli (Mirch)
The “heat” of Indian cuisine is a sensory experience governed by specific bioactive compounds in kitchen spices known as capsaicinoids.
Capsaicinoids: The Heat Molecules
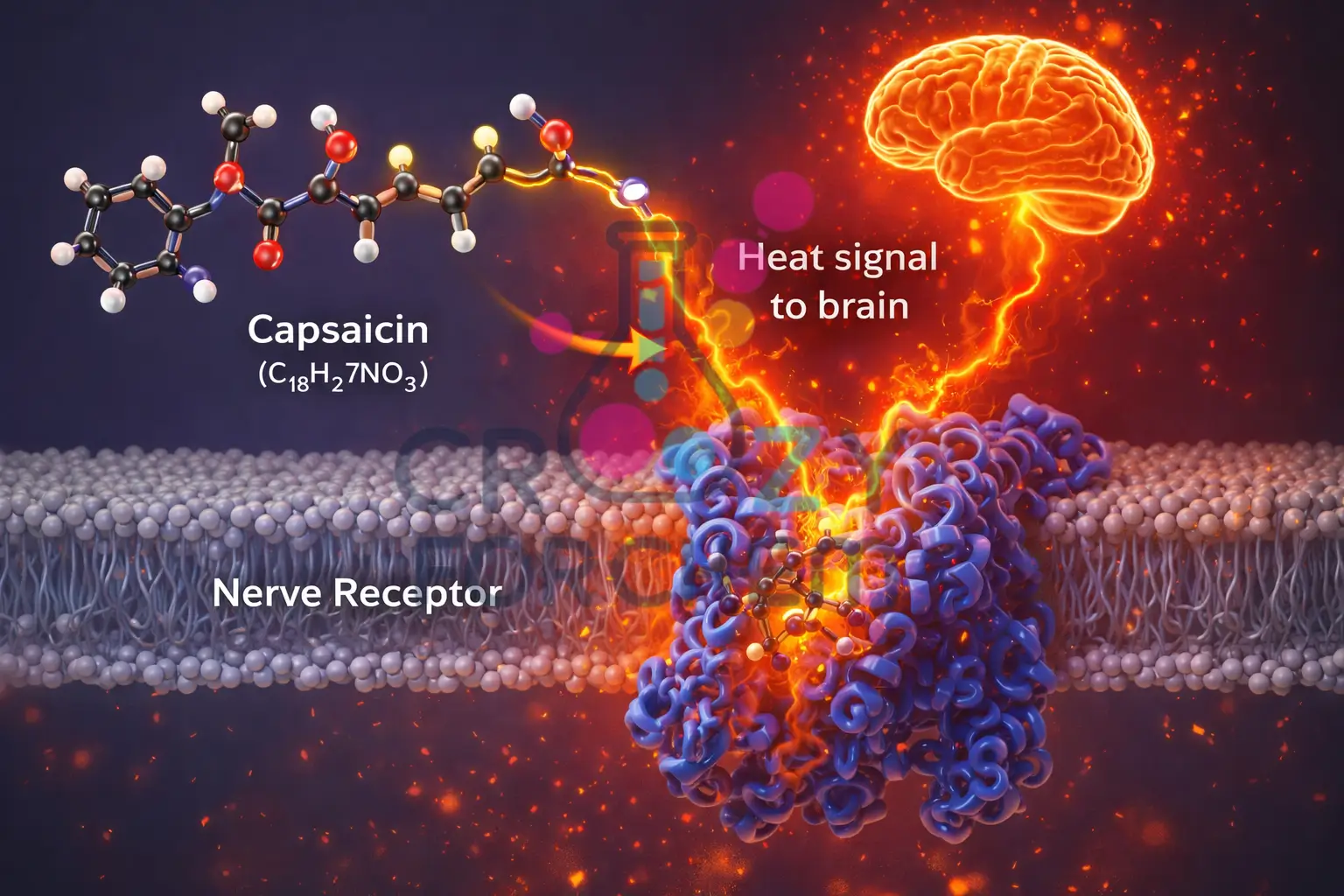
Pungency in chillies is produced by a group of seven homologous alkaloids found exclusively in the Capsicum genus.
The most prevalent is capsaicin (C₁₈H₂₇NO₃), a stable and powerful alkaloid that can be detected by human taste buds even in solutions of ten parts per million.
Capsaicin acts by binding to TRPV1 pain receptors in the mouth, tricking the brain into perceiving a sensation of burning heat, though no physical thermal damage occurs.
The Scoville Heat Unit (SHU) Scale
Chemistry allows us to quantify this “pain” through the Scoville scale, which measures the concentration of capsaicinoids.
Table 1: The Scoville Scale – Heat Levels of Common Varieties
| Chilli Variety | Scoville Heat Units (SHU) | Heat Level |
|---|---|---|
| Bell Pepper | 0 – 100 | Non-pungent |
| Jalapeño | 2,500 – 5,000 | Mildly Hot |
| Cayenne / Tabasco | 30,000 – 50,000 | Medium Hot |
| Habanero | 200,000 – 350,000 | Very Hot |
| Nagarhari (Assam) | 855,000 | Incendiary |
| Pure Capsaicin | 16,000,000 | Chemical Limit |
Carotenoids and Red Colour
The visual appeal of chillies comes from carotenoids, primarily capsanthin and capsorubin, which account for 65–80% of the total colour.
These pigments are more than just dyes; they are antioxidants that can be stabilised when esterified with fatty acids, protecting the spice from enzymatic oxidation.
3. The Aromatic Profile of Cumin (Jeera)
Cumin (Cuminum cyminum L.) is a cornerstone of the Chemistry of Indian spices, known for its warm, persistent aroma.
Cuminaldehyde: The Signature Scent
The dominant aroma component in cumin is cuminaldehyde. Interestingly, cuminaldehyde is also a potent tyrosinase inhibitor, which means it can prevent the browning of certain foods by inhibiting oxidation.
The Impact of Roasting
The tradition of dry-roasting cumin is a deliberate chemical intervention.
When cumin is heated (ideally at 125°C for 10 minutes), it triggers the Maillard reaction, a complex interaction between amino acids and reducing sugars.
This transformation produces new heterocyclic flavour compounds called pyrazines, which provide the characteristic roasted, nutty flavour.
Roasted jeera benefits also include improved digestion, as these compounds stimulate the secretion of digestive enzymes, as thymol does in ajwain.
4. Kitchen Chemistry: Heat and Solubility
Mastering the Chemistry of Indian spices requires an understanding of how these molecules interact with heat and solvents like water or oil.
Fat-Soluble vs Water-Soluble (The Science of Tadka)

Many of the most potent bioactive compounds in kitchen spices, such as curcumin and capsaicin, are lipophilic (fat-soluble) and have extremely poor water solubility.
This is the scientific justification for the Indian culinary technique of tempering (Tadka). By heating spices in oil or ghee, we “unlock” these organic compounds, making them significantly more bioavailable for the body to absorb.
Bioavailability and Synergy

Synergy is a key principle in spice chemistry. While curcumin is poorly absorbed on its own, its bioavailability is increased by 2,000% when consumed with black pepper.
The piperine in black pepper blocks the enzymes that would otherwise break down curcumin before the body can use it.
Thermal Stability and Nutrient Loss
Not all chemical compounds in spices survive the heat of the pressure cooker:
- Heat-Sensitive: Vitamin C (ascorbic acid), found in high concentrations (up to 121mg/100g) in fresh green chillies, is easily destroyed by heat and long storage. To preserve it, fresh chillies should be added at the end of cooking [Outline].
- Heat-Stable: In contrast, the pungency of piperine in black pepper is relatively stable, though it can even lose 16–34% of its concentration during intense pressure cooking [Outline].
5. Beyond the Trio: Supporting Spices
The Chemistry of Indian spices is further enriched by other “power players” that act as bioenhancers and health tonics.
Black Pepper: The King of Spices
Black pepper accounts for 35% of the world trade in spices. Its active principle, piperine, is essential for modern medicine as it enhances the bioavailability of a wide range of diverse drugs by altering membrane lipid dynamics in the intestine.
Garlic: The Natural Antibiotic
Garlic contains more than 200 chemical compounds, the most vital being allicin. Allicin provides garlic’s antibiotic properties and its distinctive pungent odour.
In comparative studies, allicin has demonstrated the highest antimicrobial potency against pathogens like E. coli.
Cardamom: The Queen of Spices
Small cardamom is dominated by 1,8-cineole (which provides a camphorous, zesty note) and α-terpinyl acetate (which adds a mild spicy flavour).
It is valued as a carminative and is used to detoxify caffeine in excessive coffee drinkers.
6. Quality Control and Storage Chemistry
The efficacy of bioactive compounds in kitchen spices is highly dependent on how they are processed and stored.
- Essential Oils: The aroma of spices comes from volatile oils that evaporate when exposed to heat, light, and air. This is why fresh spices always smell stronger; their “chemistry” is still intact.
- Storage Limits: Rhizomes like turmeric and ginger can only be stored for a maximum of two years in airtight containers before significant quality loss occurs due to pests like the cigarette beetle.
- Grinding Techniques: Traditional pounding is often superior to high-speed grinding. High temperatures during commercial grinding can cause the loss of delicate volatile oils. Cryogenic grinding (using liquid nitrogen) is a modern solution that protects these medicinal properties.
Chemist’s Note: “In my industrial training, I’ve seen how heat during grinding can degrade volatile oils by up to 40%. For the best medicinal chemistry at home, always pound your spices fresh!”
7. Conclusion: The Natural Pharmacy
The Chemistry of Indian spices transforms our daily meals from simple sustenance into a complex delivery system for health-promoting molecules.
By understanding how chemical compounds in spices like curcumin, capsaicin, and cuminaldehyde interact with heat and fat, we can optimise both the flavour and the medicinal potential of our food.
Modern science continues to validate the traditional wisdom of combining these spices, proving that the Indian spice box is truly a masterpiece of organic chemistry.
Frequently Asked Questions
-
What is the primary chemical compound in turmeric?
The primary bioactive chemical compound in turmeric is curcumin (diferuloylmethane). Curcumin is a polyphenol responsible for turmeric’s vibrant yellow colour and its potent anti-inflammatory and antioxidant properties. In the chemistry of Indian spices, curcumin is unique because it acts as a natural pH indicator, turning red in alkaline environments.
-
Why is black pepper eaten with turmeric in chemistry?
Consuming black pepper with turmeric is a classic example of biochemical synergy. Turmeric’s active compound, curcumin, has low bioavailability and is quickly metabolised by the liver. However, the piperine found in black pepper inhibits the metabolic pathway (glucuronidation) that eliminates curcumin, increasing its absorption in the human body by up to 2,000%.
-
Which chemical makes chillies taste hot?
The “heat” or pungency of chillies is caused by a group of alkaloids called capsaicinoids, the most dominant being capsaicin (C₁₈H₂₇NO₃). Capsaicin does not cause a physical burn; instead, it binds to TRPV1 receptors (vanilloid receptors) on the tongue, tricking the brain into sensing heat and pain.
-
How does the Scoville scale measure spice levels?
The Scoville Heat Unit (SHU) scale is the chemical standard for measuring the concentration of capsaicinoids in peppers. Originally a subjective taste test, it is now measured using High-Performance Liquid Chromatography (HPLC). On this scale, a bell pepper ranks at 0 SHU, while pure capsaicin reaches 16,000,000 SHU.
-
Why is “Tadka” (tempering) scientifically important?
In the chemistry of Indian spices, many bioactive molecules like curcumin (turmeric) and capsaicin (chilli) are lipophilic, meaning they are fat-soluble rather than water-soluble. Heating these spices in oil or ghee (Tadka) “unlocks” these organic compounds, allowing them to dissolve into the cooking fat, which makes them easier for the human digestive system to absorb.
-
What is the main aroma compound in cumin (Jeera)?
The signature earthy and warm scent of cumin is primarily due to a volatile organic compound called cuminaldehyde. When cumin seeds are roasted, the Maillard reaction occurs between the seeds’ amino acids and reducing sugars, producing new heterocyclic compounds called pyrazines, which give roasted Jeera its distinct nutty flavour.
-
Does boiling spices destroy their medicinal properties?
Some chemical compounds in spices are heat-sensitive. For example, Vitamin C in fresh green chillies is easily destroyed by high temperatures and long cooking times. However, other compounds like piperine in black pepper and curcumin in turmeric are relatively heat-stable, although they can still degrade slightly during intense pressure cooking.
